15/04/2026
Ammonia (NH3) is a ubiquitous molecule in chemistry, playing a vital role in everything from biological processes to industrial applications. Its seemingly simple structure, a central nitrogen atom bonded to three hydrogen atoms, belies a fascinating electron configuration that dictates its reactivity and behaviour, particularly when it acts as a ligand in coordination chemistry. Understanding the number of unpaired electrons and the availability of vacant orbitals in ammonia is key to grasping concepts like hybridisation and the formation of complex ions. This article delves into the electronic makeup of ammonia, clarifying common queries and shedding light on its significance.
The Electron Configuration of Nitrogen
To understand ammonia, we must first examine its central atom, nitrogen. Nitrogen resides in Group 15 of the periodic table, meaning it has an atomic number of 7. Its electron configuration is therefore 1s22s22p3. This tells us that the nitrogen atom has a total of 7 electrons distributed across its orbitals. The outermost shell, the n=2 shell, contains 2 electrons in the 2s orbital and 3 electrons in the 2p orbitals. These 2p orbitals (2px, 2py, and 2pz) are degenerate, meaning they have the same energy level, and according to Hund's rule, each of these 2p orbitals will be occupied by a single electron before any orbital is doubly occupied. This gives nitrogen three unpaired electrons in its ground state.
Bonding in Ammonia (NH3)
When nitrogen forms ammonia with three hydrogen atoms, it undergoes hybridisation. To form three sigma bonds with the hydrogen atoms, the nitrogen atom needs three hybrid orbitals. It achieves this by hybridising its one 2s orbital and all three 2p orbitals. However, this is not quite right. Nitrogen has 5 valence electrons (2s22p3). To form three single bonds with hydrogen, it needs to share electrons. Each hydrogen atom contributes one electron, and the nitrogen atom contributes one electron to each of the three N-H bonds. This uses three of nitrogen's valence electrons. The remaining two valence electrons on the nitrogen atom form a lone pair.
The hybridisation process in ammonia involves one 2s orbital and three 2p orbitals. This would result in four sp3 hybrid orbitals. Nitrogen uses three of these sp3 hybrid orbitals to form sigma bonds with the 1s orbitals of the three hydrogen atoms. The remaining sp3 hybrid orbital is occupied by the lone pair of electrons. Each of these sp3 hybrid orbitals has a specific electron occupancy:
| Hybrid Orbital | Electron Occupancy |
|---|---|
| sp3 (Bonding) | 1 electron |
| sp3 (Bonding) | 1 electron |
| sp3 (Bonding) | 1 electron |
| sp3 (Lone Pair) | 2 electrons |
Crucially, in the completed ammonia molecule, all valence electrons of the nitrogen atom are paired. The three electrons that form the sigma bonds are paired with the electrons from the hydrogen atoms, and the lone pair consists of two electrons occupying a single hybrid orbital, which are also paired.
Unpaired Electrons in NH3
Based on the bonding description above, we can definitively state that ammonia (NH3) has zero unpaired electrons. All the valence electrons of the nitrogen atom are involved in bonding or reside as a paired lone pair. This is a fundamental characteristic of ammonia that influences its behaviour as a Lewis base, readily donating its lone pair of electrons to form coordinate covalent bonds.
Vacant Orbitals in NH3
The question of vacant orbitals in ammonia requires careful consideration of the valence shell electrons and the hybridisation process. As described, nitrogen in ammonia utilises its 2s and 2p orbitals for hybridisation. The hybridisation process creates four sp3 hybrid orbitals. Three of these are used for sigma bonding, and one contains the lone pair. Therefore, within the valence shell that participated in hybridisation (the n=2 shell), there are no vacant hybrid orbitals available for further bonding directly from the nitrogen atom in the ammonia molecule itself.
However, the context in which this question often arises is in coordination chemistry, specifically when ammonia acts as a ligand. For instance, in the complex [Co(NH3)6]3+, the central cobalt ion (Co3+) accepts electron pairs from six ammonia ligands. For ammonia to act as a ligand, it needs to donate its lone pair of electrons. This donation occurs from the lone pair residing in one of the sp3 hybrid orbitals of the nitrogen atom. The act of donating this electron pair does not inherently create a vacant orbital *within* the ammonia molecule itself. The donation is from an *occupied* orbital.
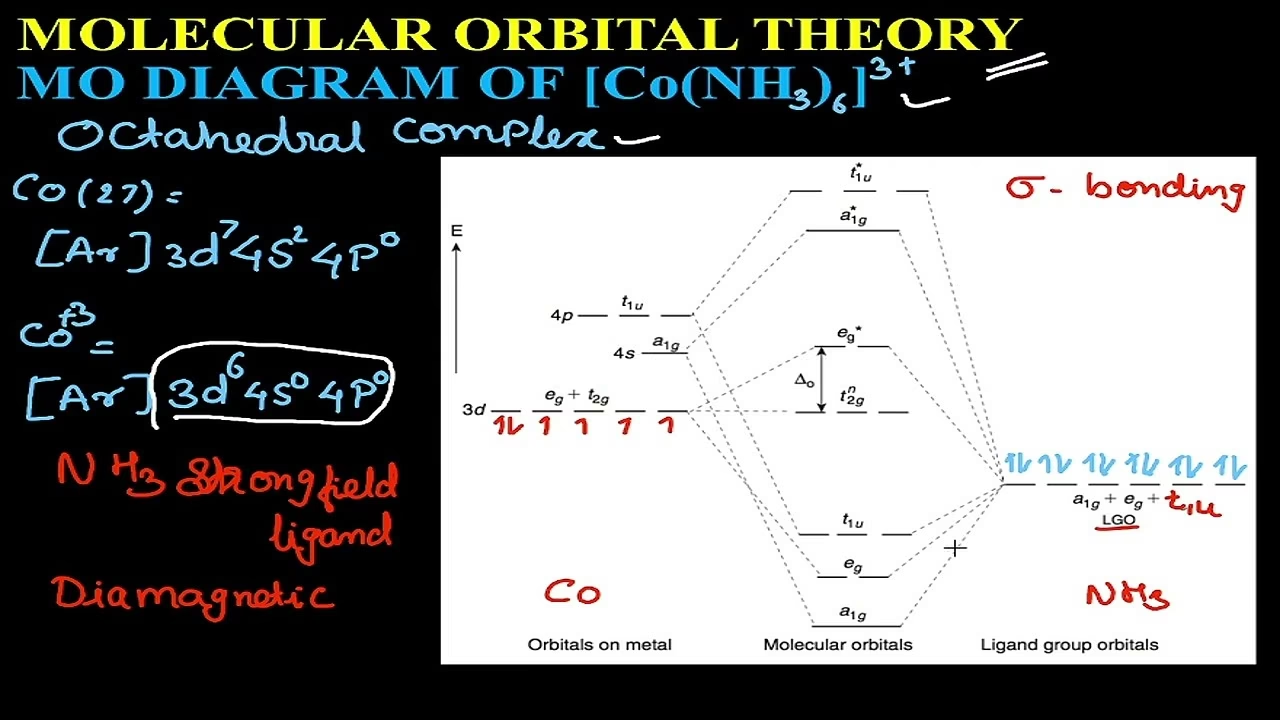
The concept of vacant orbitals becomes more pertinent when discussing the metal centre in coordination complexes. In the case of [Co(NH3)6]3+, the Co3+ ion has an electronic configuration that allows for vacant orbitals which accept the electron pairs from the ammonia ligands. The electronic configuration of Co3+ is [Ar] 3d6 4s0 4p0. To achieve an octahedral coordination, the cobalt ion undergoes d2sp3 hybridisation, utilising two vacant 3d orbitals, one vacant 4s orbital, and three vacant 4p orbitals to form six hybrid orbitals. These six hybrid orbitals then accept the lone pairs from the six ammonia molecules.
Ammonia as a Ligand and the MO Diagram
When ammonia acts as a ligand, it donates its lone pair from a filled sp3 hybrid orbital. The interaction between the metal ion and the ligand creates molecular orbitals. For a complex like [Co(NH3)6]3+, a Crystal Field Theory (CFT) or Molecular Orbital (MO) diagram is used to describe the splitting of the d-orbitals of the central metal ion and their interaction with the ligand orbitals. In the context of CFT, ammonia is considered a strong field ligand. This means it causes a large splitting of the d-orbitals of the metal ion. Due to its strength, ammonia can induce spin pairing in the metal ion's d-electrons if there are unpaired electrons present. In the case of Co3+, which has a d6 configuration, the six electrons in the d-orbitals will arrange themselves to minimise unpaired electrons when coordinated with a strong field ligand like ammonia, resulting in a low-spin complex.
The MO diagram for such a complex is complex, involving the interaction of the metal's d, s, and p atomic orbitals with the relevant orbitals of the ligand. For ammonia, the primary interaction involves the lone pair on the nitrogen atom. The MO diagram would show the formation of bonding, non-bonding, and antibonding molecular orbitals. The lone pair of ammonia typically occupies a non-bonding molecular orbital or contributes to bonding molecular orbitals depending on the specific model used.
The statement "Since NH3 is a strong ligand, due to spin pairing effect, All the four unpaired electrons in 3d orbital are paired giving two vacant 3d orbitals" is a bit misleading when referring to ammonia itself. Ammonia, as a molecule, does not have unpaired electrons in its ground state. The spin pairing effect is observed in the central metal ion (Co3+ in this case) when it is coordinated by strong field ligands like ammonia. The Co3+ ion has a d6 configuration. In a weak field environment, these six electrons would occupy the five 3d orbitals with one electron in each of the lower energy orbitals before pairing. However, with a strong field ligand like NH3, the crystal field splitting is large enough that it is energetically favourable for the electrons to pair up in the lower energy d-orbitals, resulting in a configuration with two unpaired electrons in the higher energy set of d-orbitals. This is not correct for Co3+ with NH3. Co3+ is d6. With strong field ligands like NH3, it is low spin. The configuration is t2g^6 eg^0. All electrons are paired.
The phrase "giving two vacant 3d orbitals" in the provided context likely refers to the consequence of spin pairing within the metal ion. When the d-orbitals of Co3+ become filled in a low-spin configuration (t2g6), the eg orbitals (which are dz2 and dx2-y2) remain vacant. These vacant eg orbitals, along with the vacant 4s and 4p orbitals, are then available for hybridisation to form the six coordinate bonds in the octahedral complex. However, it's crucial to remember that these vacant orbitals belong to the metal ion, not the ammonia molecule.
Summary of Ammonia's Electron Status
To reiterate the key points:
- Ammonia (NH3) has zero unpaired electrons in its neutral molecule. All valence electrons on the nitrogen atom are paired, either in sigma bonds or as a lone pair.
- Ammonia, in its isolated molecular form, does not possess vacant valence orbitals that are readily available for direct bonding in the way a metal ion might. The hybrid orbitals formed are occupied by bonding electrons or the lone pair.
- The concept of vacant orbitals becomes relevant when discussing the central metal ion in coordination complexes where ammonia acts as a ligand. The metal ion provides the vacant orbitals for accepting the electron pair donation from ammonia's lone pair.
Frequently Asked Questions
Does NH3 have any unpaired electrons?
No, the neutral ammonia molecule (NH3) has no unpaired electrons. The nitrogen atom has a lone pair of electrons and forms three single bonds with hydrogen atoms, with all electrons being paired.
How many vacant orbitals does NH3 have for bonding?
In the context of the isolated NH3 molecule, there are no vacant valence orbitals available for further bonding. All hybrid orbitals of the nitrogen atom are occupied.
What is the role of ammonia's lone pair?
The lone pair of electrons on the nitrogen atom in ammonia makes it a Lewis base. This lone pair is readily donated to form coordinate covalent bonds with Lewis acids, such as metal ions in coordination complexes.
Why is ammonia considered a strong field ligand?
Ammonia is considered a strong field ligand because it causes a significant splitting of the d-orbitals of the central metal ion. This strong interaction can lead to spin pairing in the metal ion's d-electrons, resulting in low-spin complexes.
What is the electronic configuration of Co3+ in [Co(NH3)6]3+?
The Co3+ ion has a d6 electronic configuration. In the presence of strong field ligands like ammonia, it forms a low-spin complex with the configuration t2g6 eg0, meaning all six d-electrons are paired in the lower-energy t2g orbitals, leaving the eg orbitals vacant.
Understanding the electronic structure of molecules like ammonia is fundamental to comprehending chemical bonding and the behaviour of substances in various chemical environments. The absence of unpaired electrons and the presence of a lone pair are key features that define ammonia's reactivity and its crucial role as a ligand in coordination chemistry.
If you want to read more articles similar to Ammonia's Electron Mystery, you can visit the Automotive category.

![What is a MO diagram for the [Co(NH3)6]3+ complex?](https://willandservicecentre.co.uk/wp-content/uploads/mot-diagram-of-co-nh3-6.avif)