08/07/2013
- Understanding Crude Oil: The SARA Analysis Explained
- What Does SARA Stand For?
- The SARA Oil Testing Process
- SARA Analysis for Crude Oil Processing
- Crude Oil Stability and the Colloidal Instability Index (CII)
- Considerations for Heavy Oils and Distillates
- SARA Analysis: A Comparative Overview
- Frequently Asked Questions (FAQs)
- Conclusion
Understanding Crude Oil: The SARA Analysis Explained
Crude oil is a complex mixture of hydrocarbons, and its precise composition dictates how it will behave during refining and processing. To optimise these operations and ensure efficient resource utilisation, a thorough understanding of the crude oil's characteristics is paramount. One of the most widely recognised and effective methods for characterising crude oil is the SARA analysis. This sophisticated technique breaks down the crude into four key hydrocarbon classes: Saturates, Aromatics, Resins, and Asphaltenes. By understanding the relative proportions of these components, engineers and chemists can make informed decisions regarding refining strategies, predict potential processing issues, and even assess the stability of the crude oil itself.
What Does SARA Stand For?
SARA is an acronym representing the four main hydrocarbon classes analysed in crude oil:
- Saturates: These are paraffinic and naphthenic hydrocarbons, generally considered the most desirable components for refining into fuels like gasoline and diesel. They are relatively stable and have lower boiling points.
- Aromatics: Comprising cyclic hydrocarbons with at least one benzene ring, aromatics are valuable for producing high-octane gasoline components and chemical feedstocks. However, they can contribute to fouling and coke formation in processing units.
- Resins: Often referred to as 'polar compounds', resins are larger, more complex molecules that contain heteroatoms like nitrogen, sulphur, and oxygen. They tend to be sticky and can act as dispersants for asphaltenes.
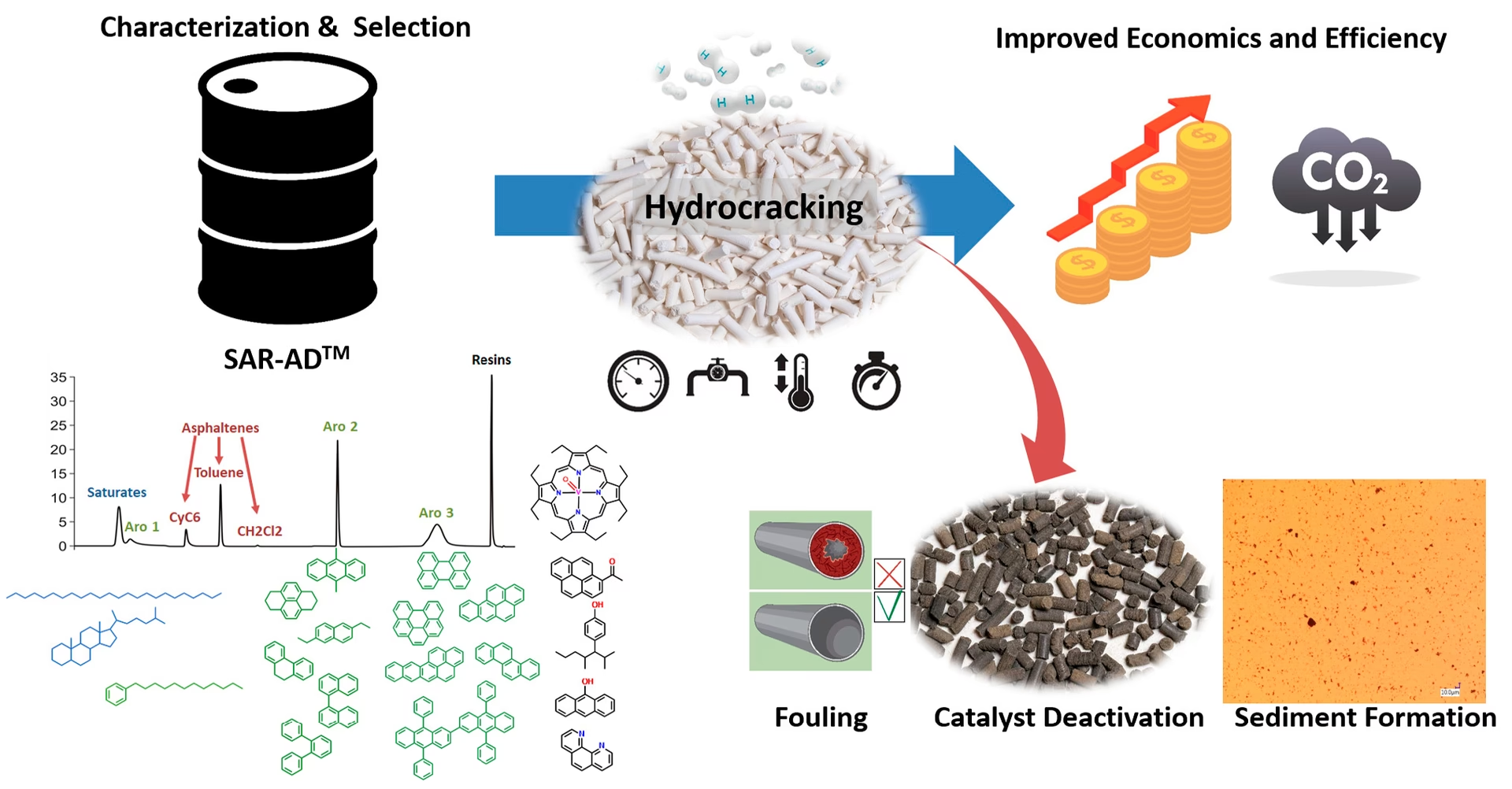
- Asphaltenes: These are the heaviest and most polar components of crude oil. They are typically defined as the fraction insoluble in n-pentane or n-heptane but soluble in aromatic solvents like toluene. Asphaltenes are notorious for their tendency to precipitate and cause operational problems such as pipeline plugging and catalyst deactivation.
The SARA Oil Testing Process
SARA oil testing is a detailed analytical process designed to quantify the amount of each of these four hydrocarbon classes present in a crude oil sample. The methodology typically involves a series of separation and analytical techniques. Here's a breakdown of the common approaches:
Separation Techniques
The core of SARA analysis lies in effectively separating these different hydrocarbon classes. Various chromatographic methods are employed:
- Adsorption Chromatography: This is a widely used technique where the crude oil sample is passed through a column packed with a polar adsorbent material, such as silica or a silica/alumina mixture. Different hydrocarbon classes will interact with the adsorbent to varying degrees, allowing for their sequential elution (washing out) using different solvents.
- Solvent Precipitation: For asphaltenes, a common preliminary step involves adding a non-polar solvent (like n-pentane or n-heptane) to the crude oil. Asphaltenes, being insoluble in these solvents, precipitate out and can be separated by filtration.
Elution and Detection
Once separated, the fractions are further analysed:
- Saturates: These are typically eluted first from the adsorption column using a paraffinic solvent, such as pentane or heptane.
- Aromatics: Following the saturates, aromatics are eluted using either paraffinic or moderately polar solvents, such as toluene or dichloromethane (DCM).
- Resins: Resins are more polar than aromatics and are eluted using more polar solvents like pyridine, toluene, or methanol. The definition of resins can vary, sometimes being classified by their solubility in specific solvent mixtures (e.g., heptane soluble, pentane insoluble).
- Asphaltenes: While often precipitated initially, if the analysis follows a column chromatography route, asphaltenes would be the final fraction eluted with a highly polar solvent.
The eluted fractions are then quantified, often gravimetrically (by weighing the residue after solvent evaporation) or using other detection methods.
Alternative and Advanced Methods
While traditional methods are effective, advancements have led to alternative and more sophisticated techniques:
- Iatroscan TLC-FID: For heavy oils with minimal volatile components (light-ends), the Iatroscan Thin-Layer Chromatography with Flame Ionisation Detection (TLC-FID) is a standardised method (IP-469). This technique also uses adsorption chromatography to determine all four SARA components. It's important to note that Iatroscan asphaltene values may differ numerically compared to gravimetric methods like IP-143, typically being around 80% of the IP-143 result.
- Flash Chromatography with ELSD: Flash chromatography coupled with Evaporative Light Scattering Detection (ELSD) has proven to be an efficient way to both separate and quantify SARA components. The ability to automate solvent switching during the analysis further enhances the resolution of the SARA fractions.
- High-Performance Liquid Chromatography (HPLC): Preparative HPLC (as described in IP-368) can also be used as an alternative or complementary technique for separating SARA components.
- Clay-Gel Method (ASTM D-2007): This method is also considered appropriate for SARA analysis, particularly when assessing certain properties.
SARA Analysis for Crude Oil Processing
The primary driver behind performing SARA analysis is to gain critical insights into how a particular crude oil will behave during refining and processing. The distribution of saturates, aromatics, resins, and asphaltenes has a direct impact on:
- Yields of Desired Products: Crude oils with a higher proportion of saturates generally yield more valuable fuels like gasoline and diesel.
- Processing Unit Fouling and Coking: High levels of resins and asphaltenes can lead to increased fouling and coke formation in refinery equipment, such as distillation columns and catalytic crackers. This reduces efficiency, increases downtime for cleaning, and can lead to equipment damage.
- Catalyst Deactivation: Asphaltenes and some resins can deposit onto refinery catalysts, reducing their activity and lifespan.
- Feedstock Suitability: SARA analysis helps determine if a crude oil is suitable for a particular refinery configuration or processing unit. For instance, heavy crudes rich in asphaltenes may require specialised upgrading processes.
- Emulsification Tendencies: The presence of polar compounds like resins and asphaltenes can influence the tendency of crude oil to form stable emulsions with water, complicating separation processes.
Crude Oil Stability and the Colloidal Instability Index (CII)
Beyond processing, SARA analysis is crucial for understanding crude oil stability, particularly concerning the potential for asphaltene precipitation. Asphaltenes are often considered to be in a colloidal suspension, stabilised by the presence of resins. When the resin-to-asphaltene ratio is too low, or when the crude is subjected to changes in pressure, temperature, or encountering incompatible solvents (like those used in enhanced oil recovery or water injection), asphaltenes can flocculate and precipitate. This precipitation can lead to:
- Reservoir Souring: Precipitation can clog pore throats, reducing oil flow.
- Pipeline Blockages: Precipitated asphaltenes can accumulate in pipelines, restricting flow and potentially causing blockages.
- Equipment Fouling: Similar to processing, asphaltene precipitation can foul surface equipment.
To quantify this instability, the Colloidal Instability Index (CII) is often computed from SARA values. The CII provides a numerical measure of the crude oil's tendency to precipitate asphaltenes, allowing for proactive measures to be taken. Different methods for SARA quantification, such as IP-143 and ASTM D893-69, are used to obtain the SARA values needed to calculate the CII.
Considerations for Heavy Oils and Distillates
The SARA analysis methodology needs to be adapted depending on the type of crude oil or petroleum product being analysed:
- Heavy Crude Oils: These oils typically have fewer volatile components (light-ends). They can often be analysed directly using SARA methods. This includes vacuum distillates, atmospheric and vacuum residues, bitumens, and asphalts. Minimising material loss from volatiles is key.
- Conventional Crude Oils: For conventional crudes with a significant proportion of light-ends, it is common practice to 'top' the sample (remove components boiling above 300°C) before conducting the SARA analysis. This prevents losses of volatile components during the separation steps, allowing the lighter distillate fraction to be analysed by other suitable methods.
- Distillates: For distillates (e.g., those <300°C), SARA analysis can still be applied, though the interpretation and specific methods might differ slightly due to the absence of heavier fractions like asphaltenes.
SARA Analysis: A Comparative Overview
Different standards and methods exist for SARA analysis, each with its nuances. Understanding these differences is important for consistent and comparable results:
| Method/Standard | Primary Technique(s) | Notes |
|---|---|---|
| IP-143 | Solvent Precipitation (Pentane/Heptane Insolubles) for Asphaltenes, Gravimetric | A common gravimetric method for asphaltenes. |
| IP-469 | Iatroscan TLC-FID | Suitable for heavy oils with low volatiles. Asphaltene results typically ~80% of IP-143. |
| IP-368 | Preparative HPLC | Advanced chromatographic separation. |
| ASTM D-2007 | Clay-Gel Adsorption Chromatography | Alternative method for SARA analysis. |
| General Adsorption Chromatography | Column Chromatography (Silica/Alumina) | Separates fractions based on polarity using various solvents. |
Frequently Asked Questions (FAQs)
Q1: Why is SARA analysis important for crude oil?
SARA analysis is crucial because it breaks down crude oil into its fundamental hydrocarbon classes, providing vital information for refining processes, predicting operational issues like fouling and precipitation, and assessing the overall stability of the crude.
Q2: What are the main differences between Saturates and Aromatics?
Saturates are paraffinic and naphthenic hydrocarbons, generally easier to refine into fuels. Aromatics are cyclic hydrocarbons containing benzene rings, valuable for high-octane components but can contribute to coking.
Q3: Can SARA analysis be used for all types of crude oil?
Yes, but the specific methodology might need adjustments. Heavy oils with low volatiles can be analysed directly, while conventional crudes with significant light-ends might require pre-treatment like topping.
Q4: What is the significance of Asphaltenes in SARA analysis?
Asphaltenes are the heaviest, most polar components and are prone to precipitation, causing operational problems. Their quantity and behaviour are critical indicators of crude oil stability and potential processing challenges.
Q5: How does SARA analysis relate to crude oil stability?
SARA analysis, particularly the ratio of resins to asphaltenes, helps determine the colloidal stability of the crude oil. A low ratio can indicate a higher risk of asphaltene precipitation, often quantified by the Colloidal Instability Index (CII).
Conclusion
The SARA analysis is an indispensable tool in the petroleum industry. It provides a detailed fingerprint of crude oil composition, enabling refiners, engineers, and geoscientists to make data-driven decisions. Whether it's optimising refining yields, preventing costly operational disruptions, or understanding the fundamental stability of a hydrocarbon resource, a comprehensive SARA analysis offers the clarity needed to navigate the complexities of crude oil characterisation and processing.
If you want to read more articles similar to Crude Oil Characterisation: The SARA Method, you can visit the Automotive category.