13/03/2023
While my usual beat involves the fascinating mechanics beneath the bonnet of a motor vehicle, the principles of energy, structure, and interaction extend far beyond the tangible world of engines and gears. Today, we're taking a deep dive into the microscopic realm of molecular orbital theory, specifically addressing a rather specific, yet profoundly important, parameter: 't' in a molecular orbital (MO) diagram. This concept is fundamental to understanding the electronic structure and properties of transition metal complexes, which, while not directly related to your car's engine, underpin many modern technologies and materials.
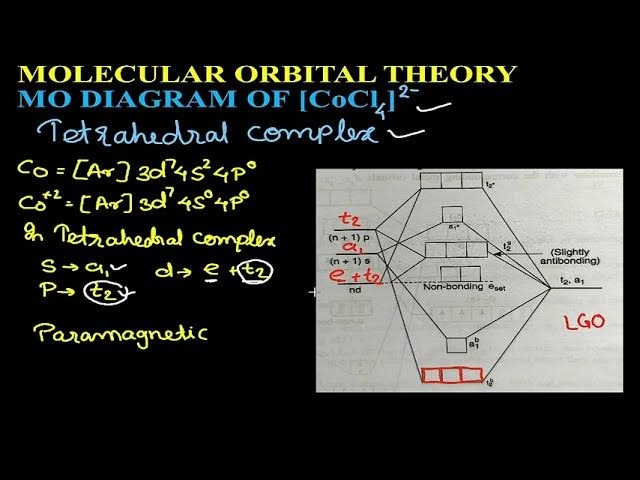
Molecular Orbital (MO) theory is a sophisticated approach to describing the bonding in molecules. Unlike simpler theories that focus on localised bonds between two atoms, MO theory considers the molecule as a whole, allowing electrons to occupy orbitals that span the entire molecular structure. These molecular orbitals are formed by combining atomic orbitals from the constituent atoms. The result is a set of new orbitals – some lower in energy (bonding orbitals), some higher (antibonding orbitals), and sometimes some remaining at roughly the same energy (non-bonding orbitals).
The Core of Molecular Orbital Theory
At its heart, MO theory posits that when atoms come together to form a molecule, their atomic orbitals (AOs) combine to produce an equal number of molecular orbitals (MOs). These MOs can be broadly categorised:
- Bonding Orbitals: These are lower in energy than the original atomic orbitals and concentrate electron density between the nuclei, leading to a stable bond.
- Antibonding Orbitals: Higher in energy, these orbitals have a node (region of zero electron density) between the nuclei and destabilise the bond. They are often denoted with an asterisk (*).
- Non-bonding Orbitals: These orbitals have energies similar to their parent atomic orbitals and do not significantly contribute to bonding or antibonding interactions.
Electrons then fill these molecular orbitals following the same rules as atomic orbitals: the Aufbau principle (filling from lowest energy up), Hund's rule (maximising unpaired spins), and the Pauli exclusion principle (maximum two electrons per orbital with opposite spins).
Introducing Crystal Field Theory (CFT)
To truly grasp 't', we must first understand a related concept: Crystal Field Theory (CFT). CFT is a model that describes the breaking of degeneracies of electron orbital energies, specifically the d-orbitals of transition metal ions, due to the electrostatic interaction with surrounding ligands. Ligands are ions or molecules that bond to a central metal atom, typically transition metals.
In an isolated transition metal ion, the five d-orbitals (dxy, dxz, dyz, dz², dx²-y²) are degenerate, meaning they have the same energy. However, when ligands approach the metal ion, their negative charge (or the negative end of their dipole) creates an electrostatic field that interacts with the d-electrons. This interaction causes the d-orbitals to split into different energy levels. The pattern of this splitting depends on the geometry of the complex. For octahedral complexes, the five d-orbitals split into two sets:
- t2g set: Consisting of the dxy, dxz, and dyz orbitals. These orbitals point between the axes and experience less repulsion from ligands approaching along the axes.
- eg set: Consisting of the dz² and dx²-y² orbitals. These orbitals point directly along the axes and experience greater repulsion from ligands.
The energy difference between the t2g and eg sets in an octahedral complex is known as the crystal field splitting energy, often denoted as Δo (delta-oh) or 10Dq. This parameter is crucial because it dictates many properties of the complex, such as its colour, magnetic properties, and reactivity.
The Convergence: Ligand Field Theory and the Meaning of 't'
While CFT is useful, it's a purely electrostatic model and doesn't explicitly account for covalent bonding. Ligand Field Theory (LFT) is a more comprehensive approach that combines elements of both CFT and MO theory, providing a more accurate description of bonding in transition metal complexes. It acknowledges that there is significant covalent character in the metal-ligand bonds, not just electrostatic interactions.
Within the framework of LFT, when we construct the molecular orbital diagram for a transition metal complex (such as an octahedral one), the d-orbitals of the metal ion interact with the orbitals of the ligands. This interaction gives rise to various molecular orbitals. For octahedral complexes, the relevant interactions lead to:
- Sigma (σ) bonding orbitals (lowest energy)
- Non-bonding orbitals (derived primarily from metal d-orbitals that don't participate in strong sigma bonding)
- Sigma (σ) antibonding orbitals (highest energy)
Crucially, some of the metal's d-orbitals participate in σ-bonding (forming eg bonding and eg* antibonding orbitals with ligand σ-orbitals), while others (the t2g set) are often considered non-bonding or participate in π-bonding/antibonding interactions depending on the ligand.
Now, let's address 't'. In the context of a molecular orbital diagram for an octahedral transition metal complex, the central portion of the diagram often resembles the d-orbital splitting seen in crystal field theory. Here, 't' corresponds to the energy separation between the e(nb) and the t2* orbitals.
- e(nb): This refers to the non-bonding 'e' set of molecular orbitals. These are derived from the metal's dz² and dx²-y² orbitals that participate in sigma interactions with ligand orbitals. After forming bonding and antibonding sigma MOs, the remaining 'e' orbitals, if they are primarily metal d-character and not strongly involved in bonding, can be considered non-bonding or slightly antibonding. However, in the context where 't' is defined, e(nb) often refers to the higher energy set of d-orbitals that are *not* the primary antibonding orbitals formed from sigma interactions. This notation can be a bit confusing as 'e' usually implies antibonding in CFT. It is more common to see the split referred to as between t2g and eg* where eg* are the antibonding orbitals derived from metal d-orbitals.
- t2*: This refers to the antibonding t2 set of molecular orbitals. In the context of sigma-only bonding, the t2g set of d-orbitals (dxy, dxz, dyz) are non-bonding. However, when ligands possess pi (π) orbitals, these t2g metal orbitals can interact with ligand π-orbitals to form π-bonding and π-antibonding (t2*) molecular orbitals.
Therefore, 't' represents a specific energy gap within the MO diagram, particularly relevant when considering the influence of pi-interactions. It signifies the energy difference between a non-bonding or weakly interacting 'e' set (often derived from metal d-orbitals) and an antibonding 't2*' set. This separation is analogous to, and derived from, the crystal field splitting energy (Δo or 10Dq) that is central to CFT. The term 't' highlights that the splitting observed in CFT is a direct consequence of the formation of molecular orbitals when metal and ligand orbitals interact.
Why is 't' Important?
The magnitude of 't' (or Δo) has profound implications for the properties of transition metal complexes:
- Colour: Many transition metal complexes are vibrantly coloured. This colour arises from the absorption of specific wavelengths of visible light, which promotes electrons from lower energy (e.g., t2g) to higher energy (e.g., eg*) d-orbitals. The energy of this transition corresponds directly to 't'. A larger 't' means absorption of higher energy (shorter wavelength) light, leading to a different observed colour.
- Magnetic Properties: The spin state (high spin vs. low spin) of a complex depends on the balance between the energy required to pair electrons in an orbital (pairing energy, P) and the splitting energy 't'. If 't' is large, electrons will pair up in lower energy orbitals (low spin). If 't' is small, electrons will occupy higher energy orbitals before pairing (high spin). This dictates whether a complex is paramagnetic (unpaired electrons) or diamagnetic (all paired electrons).
- Stability and Reactivity: The distribution of electrons in the molecular orbitals, influenced by 't', affects the overall stability of the complex. Complexes with filled bonding orbitals and empty antibonding orbitals tend to be more stable. The reactivity of complexes, particularly in ligand exchange reactions, is also influenced by the availability of vacant orbitals and the energy of occupied ones.
Comparing CFT and MO Theory (LFT)
It's helpful to view 't' in the context of how CFT and MO theory complement each other:
| Feature | Crystal Field Theory (CFT) | Molecular Orbital Theory (MO Theory/LFT) |
|---|---|---|
| Nature of Interaction | Purely electrostatic (ligands as point charges). | Covalent and electrostatic interactions (orbital overlap). |
| Origin of Splitting | Repulsion between ligand electrons and metal d-electrons. | Formation of bonding and antibonding molecular orbitals from metal and ligand atomic orbitals. |
| Scope | Focuses primarily on d-orbital splitting and its consequences. | Considers all valence orbitals of metal and ligand, providing a complete bonding picture. |
| Predictive Power | Good for predicting colour, magnetism qualitatively. | More quantitatively accurate for energy levels, bond strengths, and electronic transitions. |
| Key Parameter | Δo (or 10Dq), crystal field splitting energy. | Energy separation between specific MOs, including 't' (often related to Δo). |
| Limitations | Does not explain covalent bonding or ligand contributions to MOs. | More complex to apply, requiring knowledge of group theory and orbital symmetry. |
Factors Influencing 't' (and Δo)
The magnitude of 't' is not constant and depends on several factors, which are also reflected in the spectrochemical series of ligands:
- Nature of the Ligand: Ligands vary in their ability to cause d-orbital splitting. Strong-field ligands (e.g., CN-, CO) cause a large 't', while weak-field ligands (e.g., I-, Br-) cause a small 't'. This is due to their different abilities to donate electron density (sigma donation) and accept electron density (pi back-bonding) with the metal.
- Charge on the Metal Ion: As the positive charge on the metal ion increases, the metal orbitals contract, leading to stronger interactions with ligands and thus a larger 't'. For example, Fe3+ complexes generally have a larger 't' than Fe2+ complexes.
- Principal Quantum Number (n) of the Metal Ion: For a given ligand and oxidation state, 't' increases down a group in the periodic table (e.g., 3d < 4d < 5d metals). This is because 4d and 5d orbitals are more diffuse and can overlap more effectively with ligand orbitals, leading to stronger interactions.
- Geometry of the Complex: The splitting pattern and magnitude of 't' are highly dependent on the coordination geometry (e.g., octahedral, tetrahedral, square planar).
Frequently Asked Questions About 't' and MO Diagrams
Is 't' always the term used for this energy separation?
Not always. While 't' is used in some specific contexts to denote this separation between e(nb) and t2* orbitals in MO diagrams, the more commonly encountered term for the overall d-orbital splitting energy, particularly in the context of crystal field theory and ligand field theory, is Δo (delta-oh for octahedral) or Δt (delta-tee for tetrahedral), or sometimes 10Dq. The specific notation 't' often arises when explicitly discussing the interaction of metal t2g orbitals with ligand π-orbitals leading to t2* antibonding orbitals.
What's the difference between t2g and t2*?
The distinction lies in their context within the theories. t2g refers to a set of three degenerate d-orbitals (dxy, dxz, dyz) in the metal atom itself, which are lowered in energy due to ligand field effects in CFT, or become non-bonding/weakly π-bonding in LFT. t2* (t-two-star) refers to an antibonding molecular orbital formed when the metal's t2g orbitals interact with ligand π-orbitals. This interaction causes the original t2g energy level to split into a lower energy π-bonding MO and a higher energy π-antibonding MO (t2*). So, t2g is an atomic orbital set, while t2* is a molecular orbital. The energy separation 't' is between a higher energy 'e' set (often non-bonding or weakly antibonding) and this t2* antibonding MO.
How does 't' relate to the colour of a complex?
The colour of a transition metal complex arises from electronic transitions between the split d-orbitals. Specifically, it's often a d-d transition, where an electron absorbs light energy to jump from a lower energy orbital (like t2g or a bonding MO) to a higher energy orbital (like eg* or t2*). The energy difference between these orbitals is directly related to 't' (or Δo). The absorbed energy corresponds to a specific colour of light. The colour we perceive is the complementary colour of the light that was absorbed. A larger 't' means a larger energy gap, requiring higher energy (shorter wavelength) light for the transition, thus shifting the perceived colour.
What are non-bonding orbitals, and why are they important?
Non-bonding orbitals are molecular orbitals that do not significantly contribute to either bonding or antibonding interactions. Their energy is roughly the same as the atomic orbitals from which they are primarily derived. In transition metal complexes, certain d-orbitals of the metal might become non-bonding if they do not have the correct symmetry to overlap effectively with ligand orbitals, or if the overlap is very weak. These orbitals are important because they can still be occupied by electrons, and their energy levels are crucial in determining the overall electronic configuration, spin state, and spectroscopic properties of the complex, even if they don't directly form strong bonds.
Can 't' be measured experimentally?
The energy separation denoted by 't' (or more broadly, Δo) can be determined experimentally through techniques like UV-Visible Spectroscopy. When a complex absorbs light, the wavelength of maximum absorption (λmax) corresponds to the energy required for an electron to jump from a lower energy d-orbital set to a higher energy set. This energy (E = hc/λmax) directly relates to the splitting parameter 't'. Magnetic susceptibility measurements can also provide insights into the spin state, which is dependent on the magnitude of 't' relative to the pairing energy.
Conclusion
The parameter 't' in a molecular orbital diagram for transition metal complexes represents a crucial energy separation, often linking the non-bonding 'e' orbitals with the antibonding 't2*' orbitals. This concept is a direct result of the sophisticated interplay between crystal field theory and molecular orbital theory, culminating in ligand field theory. Understanding 't' is fundamental to explaining the vibrant colours, diverse magnetic behaviours, and varied reactivities of these fascinating compounds. While seemingly a niche chemical concept, the principles it embodies are critical to fields ranging from catalysis and material science to biological systems, showcasing how deep insights into molecular structure can unlock a world of possibilities.
If you want to read more articles similar to Understanding 't' in Molecular Orbital Diagrams, you can visit the Automotive category.
