02/02/2005
When delving into the intricate world of chemical bonding, traditional models like the Valence Shell Electron Pair Repulsion (VSEPR) theory and the Valence Bond theory offer valuable insights. They accurately predict many bond properties and molecular shapes by portraying electrons as localised 'balloons' of high density. However, these theories sometimes fall short in fully explaining the nuanced behaviour of certain molecules, such as the paramagnetism of oxygen or the precise electronic spectra of compounds.
This is where Molecular Orbital (MO) theory emerges as a more powerful and extensive approach. It fundamentally describes electrons not as localised entities, but as delocalised moieties spread across adjacent atoms, incorporating the wave character of electrons. MO diagrams, the visual representation of this theory, are indispensable tools. They predict crucial physical and chemical properties of a molecule, including its shape, bond energy, bond length, bond angle, and even its electronic spectra and magnetic properties. The objective of this guide is to provide you with the fundamental steps required to construct molecular orbital diagrams for simple homonuclear and heteronuclear diatomic molecules, principles which can then be extrapolated to more complex polyatomic systems.
- Understanding Molecular Orbitals and Their Diagrams
- The Essential Steps to Constructing an MO Diagram
- Step 1: Determine the Total Number of Valence Electrons
- Step 2: Draw and Arrange the Atomic Orbitals (AOs)
- Step 3: Construct the Molecular Orbitals (MOs) and Determine Their Energy Ordering
- Step 4: Fill Electrons into the Molecular Orbitals
- Step 5: Interpret the MO Diagram: Predict Molecular Properties
- Comparative Table: MO Energy Order
- Worked Example: H2 Molecule
- Beyond Diatomics: A Glimpse into Polyatomic MO Diagrams
- Frequently Asked Questions (FAQs)
Understanding Molecular Orbitals and Their Diagrams
At the heart of MO theory lies the concept of a molecular orbital (MO). Unlike atomic orbitals (AOs) which describe the probability of finding an electron around a single atom, an MO describes the region where an electron is most likely to be found within a molecule. An MO is formally defined as the combination of atomic orbitals from the constituent atoms.
A molecular orbital diagram visually depicts the energy levels of these molecular orbitals relative to the atomic orbitals from which they are formed. Typically, the atomic orbital energy levels are shown on the sides, with the combined molecular orbital energy levels in the centre. Energy levels increase from bottom to top, and lines (often dashed diagonal lines) connect the MO levels to their constituent AO levels, indicating their parentage. Degenerate energy levels (orbitals of the same energy) are frequently depicted next to each other.
Electron filling in MO diagrams adheres to the same fundamental principles as atomic orbital filling: the Aufbau Principle (filling from lowest to highest energy), the Pauli Exclusion Principle (each orbital can hold a maximum of two electrons with opposite spins), and Hund's Rule (for degenerate orbitals, electrons fill singly before pairing up). Small vertical arrows represent electrons, with their direction indicating spin.
For simplicity, MO diagrams often omit the shapes of the AOs or MOs themselves, focusing solely on their energy levels and electron occupation. For diatomic molecules, an MO diagram effectively illustrates the energetics of the bond between the two atoms. For polyatomic molecules, a diagram might focus on one or more bonds of interest, simplifying the overall representation. Furthermore, inner orbital AOs and their electrons are often omitted from diagrams, as valence electrons are primarily responsible for bonding.
Key Concepts in Molecular Orbital Theory
Before diving into construction, let's clarify some essential terminology:
- Homonuclear Diatomics: These are molecules composed of two identical atoms, such as H2, N2, O2, and F2.
- Heteronuclear Diatomics: These molecules consist of two non-identical atoms, like CO, NO, HF, and LiF.
- Bonding and Antibonding Orbitals: When atomic orbitals combine, they can do so in two primary ways:
- Bonding Orbitals: These result from the constructive interference of atomic orbitals, where regions of high electron probability merge. They are lower in energy than the original atomic orbitals, leading to increased stability and bond formation.
- Antibonding Orbitals: These arise from the destructive interference of atomic orbitals, where regions with dense electron probabilities do not merge, often creating a node (a region of zero electron probability) between the nuclei. They are higher in energy than the original atomic orbitals, leading to destabilisation of the molecule. Antibonding orbitals are typically denoted with an asterisk (*).
- Phases and Nodes: In the context of electron waves, phases are designated either (+) or (-) relative to their wave 'up' or 'down' displacements. It's crucial to remember that these signs do NOT symbolise charges. A node occurs if the phase signs change from (+) to (-) or vice versa. Nodes are regions where the probability of finding an electron is zero.
- Sigma (σ) and Pi (π) Bonds:
- Sigma (σ) Bonds: These are 'end-to-end' bonds formed from the direct, head-on overlap of symmetric atomic orbitals (e.g., s-s, s-p, p-p along the internuclear axis). They are cylindrically symmetrical around the internuclear axis.
- Pi (π) Bonds: These are formed from the 'sideways' overlap of parallel p-orbitals. A pi bond consists of two regions of overlap, one above and one below the internuclear axis.
The Essential Steps to Constructing an MO Diagram
Constructing an MO diagram systematically involves several steps. Following these ensures accuracy and a comprehensive understanding of the molecule's electronic structure.
Step 1: Determine the Total Number of Valence Electrons
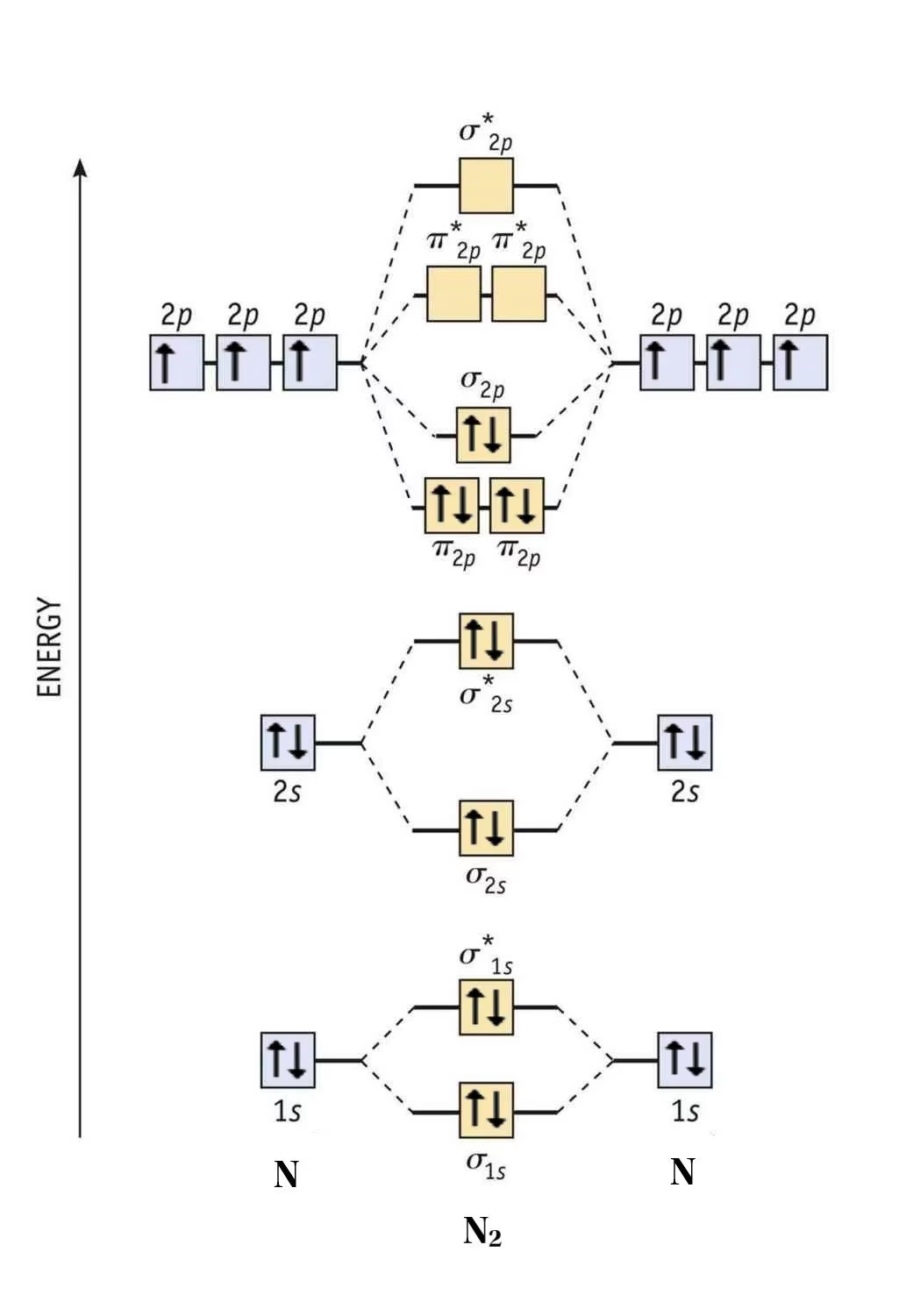
This is your starting point. For a molecule, sum the valence electrons of all constituent atoms. Remember that valence electrons are those in the outermost shell, participating in bonding. For diatomic molecules, since there are two atoms, the total number of valence electrons will be the sum of the valence electrons from each atom. For instance, in N2, each nitrogen atom has 5 valence electrons (2s22p3), so N2 has a total of 10 valence electrons.
Step 2: Draw and Arrange the Atomic Orbitals (AOs)
On either side of your diagram, draw the relevant atomic orbitals for each atom. For second-period elements, these will typically be the 2s and 2p orbitals. The 1s orbitals are usually considered inner-shell and often omitted for simplicity, as they form non-bonding or weakly bonding MOs that don't significantly impact the valence properties. Ensure the AOs are drawn at their approximate relative energy levels. For homonuclear diatomics, the AOs on both sides will be at the same energy. For heteronuclear diatomics, the AOs of the more electronegative atom will generally be lower in energy.

Step 3: Construct the Molecular Orbitals (MOs) and Determine Their Energy Ordering
This is arguably the most critical step. Atomic orbitals combine to form an equal number of molecular orbitals. For example, two 2s AOs combine to form one σ2s (bonding) and one σ*2s (antibonding) MO. Similarly, the three 2p AOs from each atom combine to form six MOs: σ2p, two degenerate π2p (bonding), σ*2p, and two degenerate π*2p (antibonding).
A crucial point here is the energy ordering of the 2p-derived MOs, which varies depending on the atoms involved due to s-p orbital mixing:
- For Lighter Diatomics (B2, C2, N2): Due to significant s-p mixing, the π2p bonding orbitals are lower in energy than the σ2p bonding orbital. The energy order is: σ2s < σ*2s < π2p < σ2p < π*2p < σ*2p.
- For Heavier Diatomics (O2, F2, Ne2): S-p mixing is less pronounced, so the σ2p bonding orbital is lower in energy than the π2p bonding orbitals. The energy order is: σ2s < σ*2s < σ2p < π2p < π*2p < σ*2p.
This difference is fundamental for correctly predicting properties like paramagnetism (e.g., for O2).
Step 4: Fill Electrons into the Molecular Orbitals
Once the MOs are drawn and their energy ordering established, fill them with the total number of valence electrons determined in Step 1. Apply the three fundamental rules:
- Aufbau Principle: Fill electrons into the lowest energy MOs first, moving upwards.
- Pauli Exclusion Principle: Each molecular orbital can hold a maximum of two electrons, and these electrons must have opposite spins (represented by arrows pointing up and down).
- Hund's Rule: When filling degenerate orbitals (like the two π2p or π*2p orbitals), electrons will occupy each orbital singly with parallel spins before any orbital is doubly occupied.
Step 5: Interpret the MO Diagram: Predict Molecular Properties
With the MO diagram filled, you can now deduce several important molecular properties:
- Molecular Stability: A molecule is stable if the number of electrons in bonding orbitals (Nb) is greater than the number of electrons in antibonding orbitals (Na). If Nb ≤ Na, the molecule is generally unstable or does not exist.
- Bond Order: This quantifies the number of chemical bonds between two atoms. It is calculated as half the difference between the number of bonding and antibonding electrons:
Bond Order = 1/2 (Nb - Na)
A bond order of 1, 2, or 3 typically corresponds to a single, double, or triple bond, respectively. A positive bond order indicates a stable molecule. - Bond Length: Bond order is inversely proportional to bond length. Higher bond orders imply stronger bonds and thus shorter bond lengths.
- Magnetic Properties:
- Diamagnetic: If all electrons in the MO diagram are paired, the molecule is diamagnetic, meaning it is weakly repelled by a magnetic field.
- Paramagnetic: If the molecule contains one or more unpaired electrons, it is paramagnetic, meaning it is attracted to a magnetic field. This is a key prediction where MO theory often surpasses Valence Bond theory (e.g., O2).
Comparative Table: MO Energy Order
Understanding the difference in MO energy ordering is crucial for accurate predictions:
| Type of Diatomic | Examples | MO Energy Order (2s & 2p derived) | Key Characteristic |
|---|---|---|---|
| Lighter Homonuclear Diatomics | B2, C2, N2 | σ2s < σ*2s < π2p < σ2p < π*2p < σ*2p | π2p is lower than σ2p due to s-p mixing |
| Heavier Homonuclear Diatomics | O2, F2, Ne2 | σ2s < σ*2s < σ2p < π2p < π*2p < σ*2p | σ2p is lower than π2p; less s-p mixing |
Worked Example: H2 Molecule
Let's apply these steps to the simplest homonuclear diatomic, H2:
- Total Valence Electrons: Each hydrogen atom has 1 valence electron (1s1). So, H2 has 1 + 1 = 2 valence electrons.
- Atomic Orbitals: We consider the 1s orbitals for each hydrogen atom.
- Molecular Orbitals: The two 1s AOs combine to form one σ1s (bonding) and one σ*1s (antibonding) MO. The σ1s is lower in energy than the σ*1s.
- Fill Electrons: We have 2 valence electrons to fill. Both electrons go into the lowest energy orbital, the σ1s bonding orbital, with opposite spins. The electronic configuration for H2 is σ1s2.
- Interpret Diagram:
- Nb = 2 (electrons in σ1s)
- Na = 0 (no electrons in σ*1s)
- Stability: Nb (2) > Na (0), so H2 is a stable molecule.
- Bond Order: 1/2 (2 - 0) = 1. This indicates a single bond between the two hydrogen atoms, consistent with its known structure.
- Magnetic Properties: All electrons are paired in the σ1s orbital. Therefore, the H2 molecule is diamagnetic.
Beyond Diatomics: A Glimpse into Polyatomic MO Diagrams
While this guide focuses on diatomic molecules, the fundamental principles of MO theory extend to polyatomic molecules. However, their diagrams become significantly more complex, often involving the combination of central atom AOs with group orbitals (combinations of AOs from surrounding atoms). The core idea remains: AOs combine to form MOs, which are then filled according to the Aufbau principle, Pauli exclusion principle, and Hund's rule, allowing for predictions of molecular properties. The complexity increases due to a larger number of AOs to consider and more intricate symmetry requirements for their combination.
Frequently Asked Questions (FAQs)
Here are some common questions regarding MO diagrams and theory:
Why is Molecular Orbital Theory considered superior to Valence Bond Theory or VSEPR?
MO theory offers a more complete picture of electron behaviour. While Valence Bond theory and VSEPR are excellent for predicting basic geometry and localised bonds, they struggle with phenomena like paramagnetism (e.g., O2, which VB theory predicts as diamagnetic) and delocalised bonding (e.g., benzene). MO theory naturally accounts for electron delocalisation and provides insights into electronic transitions (spectra), making it a more powerful and accurate model for understanding molecular properties.
What is the significance of bond order in MO theory?
Bond order is a direct indicator of bond strength and stability. A higher bond order means more electrons are contributing to bonding, resulting in a stronger, shorter, and more stable bond. A bond order of zero or less indicates that a stable molecule will not form between the atoms.
How do I determine if a molecule is paramagnetic using an MO diagram?
After filling all the valence electrons into the molecular orbitals according to Hund's rule, simply inspect the diagram. If any molecular orbital contains a single, unpaired electron, the molecule is paramagnetic. If all electrons are paired up in their respective orbitals, the molecule is diamagnetic.
Are inner shell electrons included in MO diagrams?
Typically, for simplicity and because they do not participate significantly in bonding, inner shell electrons and their corresponding atomic and molecular orbitals are omitted from MO diagrams. Focus is usually placed on the valence electrons and the MOs formed from their valence atomic orbitals.
Mastering the construction and interpretation of MO diagrams provides a profound understanding of chemical bonding and molecular behaviour, moving beyond simpler models to unlock the true electronic nature of compounds. This foundational knowledge is key to predicting and explaining a wide array of chemical phenomena.
If you want to read more articles similar to Building MO Diagrams: A Step-by-Step Guide, you can visit the Automotive category.
