13/11/2001
Olive oil, particularly extra virgin olive oil (EVOO), is prized for its flavour, health benefits, and culinary versatility. However, like all fats and oils, it is susceptible to oxidation, a process that can degrade its quality, alter its flavour profile, and diminish its nutritional value. Understanding how to measure this oxidation is crucial for producers, consumers, and chefs alike. This article delves into the methods used to assess olive oil oxidation, drawing on scientific insights into how different cultivars respond to challenging conditions.
What is Olive Oil Oxidation?
Oxidation in olive oil is a chemical reaction that occurs when the oil comes into contact with oxygen. This process can be accelerated by factors such as heat, light, and the presence of certain metals. The primary targets of oxidation are the unsaturated fatty acids, particularly the double bonds within the fatty acid chains. When these bonds react with oxygen, they can form a cascade of compounds, including hydroperoxides, aldehydes, ketones, and other volatile and non-volatile breakdown products. These compounds are responsible for the rancid or "off" flavours and aromas often associated with degraded oil.
Key Indicators of Olive Oil Oxidation
Scientists and quality control professionals employ several key parameters to quantify the extent of oxidation in olive oil. These measurements provide a snapshot of the oil's condition and its susceptibility to further degradation. The most commonly monitored indicators include:
1. Peroxide Value (PV)
The peroxide value is perhaps the most widely used measure of primary oxidation. It quantifies the amount of hydroperoxides present in the oil. Hydroperoxides are the initial products formed when oxygen attacks the unsaturated fatty acids. A higher peroxide value indicates that the oil has undergone more primary oxidation. This is typically measured using a titration method, where the hydroperoxides in a known amount of oil react with potassium iodide, releasing iodine. The amount of iodine released is then titrated with a standard solution of sodium thiosulphate. The result is usually expressed in milliequivalents of active oxygen per kilogram of oil (meq O₂/kg).
Table 1: Peroxide Value Ranges for Olive Oil Quality
| Quality Grade | Peroxide Value (meq O₂/kg) |
|---|---|
| Extra Virgin Olive Oil (EVOO) - Fresh | ≤ 20 |
| Extra Virgin Olive Oil (EVOO) - Acceptable | 20.1 - 25 |
| Virgin Olive Oil | ≤ 20 |
| Refined Olive Oil | ≤ 10 |
2. Conjugated Dienes and Trienes
As oxidation progresses, hydroperoxides can further break down and rearrange, leading to the formation of conjugated systems of double bonds. Conjugated dienes and conjugated trienes are secondary oxidation products that absorb ultraviolet (UV) light at specific wavelengths. Their presence and concentration can be measured using spectrophotometry. An increase in conjugated dienes, particularly those absorbing around 232 nm, indicates initial oxidative degradation. The presence of conjugated trienes, absorbing around 270 nm, suggests a more advanced stage of oxidation.
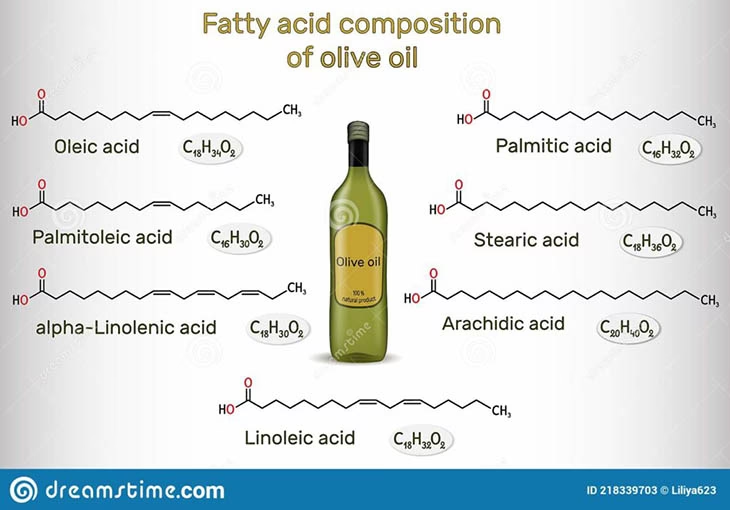
3. Fatty Acid Composition
Olive oil is primarily composed of triglycerides, which are esters of glycerol and fatty acids. The most abundant fatty acid in olive oil is oleic acid, a monounsaturated fatty acid. Other important fatty acids include linoleic acid (polyunsaturated) and palmitic acid (saturated). Oxidation primarily affects the unsaturated fatty acids due to the presence of their reactive double bonds. Monitoring changes in the fatty acid composition over time can provide insights into the oxidative stability of the oil. A significant decrease in polyunsaturated fatty acids like linoleic acid, or an increase in saturated fatty acids relative to unsaturated ones, can be indicative of oxidative stress.
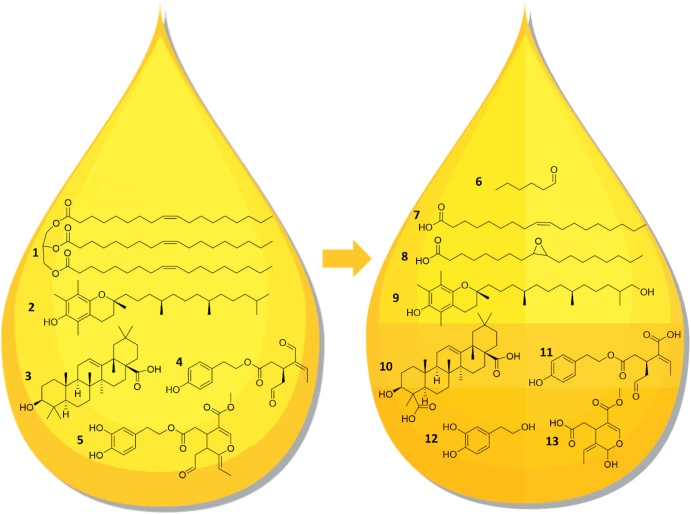
4. Minor Compound Content
EVOO is rich in various minor compounds that contribute significantly to its health benefits and stability. These include:
- Tocopherols: Primarily alpha-tocopherol, which is a potent antioxidant.
- Polyphenols: A diverse group of compounds, including phenolic acids, secoiridoids (like oleocanthal and oleuropein derivatives), lignans, and flavones. These are powerful antioxidants that protect the oil from oxidation.
- Sterols: Such as beta-sitosterol, campesterol, and stigmasterol.
- Squalene: A hydrocarbon that also possesses antioxidant properties.
The degradation rate of these minor compounds under oxidative conditions is a critical indicator of the oil's quality and shelf life. For instance, studies have shown that tocopherols and polyphenols are among the first compounds to be affected by heat and oxidation, acting as sacrificial antioxidants.
The Impact of Heat on Oxidation
Elevated temperatures, as experienced during processing or cooking, can significantly accelerate the oxidation of olive oil. A study involving heating two monovarietal EVOOs (Arbequina and Picual) at 180°C for 36 hours provides valuable insights into how different cultivars respond to thermal stress. The study monitored changes in oil quality parameters and the content of minor compounds.
Cultivar-Specific Responses to Heat
The findings revealed that while both cultivars experienced oxidation, their responses in terms of minor compound degradation differed:
- Tocopherols: Alpha-tocopherol loss was more pronounced in the Arbequina oil compared to the Picual oil. This suggests that Arbequina's alpha-tocopherol may be more susceptible to degradation under these specific heating conditions.
- Polyphenols: Total polyphenol content loss was greater in the Picual oil. Specifically, hydroxytyrosol and its secoiridoid derivatives (3,4-DHPEA-EDA and 3,4-DHPEA-EA) showed a significant decrease in Picual oil. Lignan content, however, decreased less dramatically. In contrast, for Arbequina oil, these specific phenolic compounds remained relatively stable, with a tendency for tyrosol and its derivatives (p-HPEA-EDA and p-HPEA-EA) to decrease. Flavone content also decreased, with a more significant reduction observed in Arbequina oil.
Despite these differences, the study concluded that even under severe heating conditions, VOO generally maintained most of its minor compounds, thereby retaining a significant portion of its nutritional properties. This highlights the inherent stability of high-quality olive oil.
Stable Compounds
Certain components of olive oil demonstrated remarkable stability against oxidation and heat. These included:
- Oleic acid
- Sterols
- Squalene
- Triterpenic alcohols (erythrodiol and uvaol)
- Triterpenic acids (oleanolic and maslinic)
These compounds are less susceptible to oxidative breakdown, contributing to the overall robustness of the oil.
Measuring Oxidation: A Practical Approach
For consumers and culinary professionals, directly measuring the complex chemical parameters might not be feasible. However, understanding these indicators helps in making informed choices:
- Look for Freshness: Purchase olive oil from reputable sources with clear production and best-before dates.
- Proper Storage: Store olive oil in a cool, dark place in a tightly sealed, preferably dark glass or stainless steel container. Avoid exposure to heat and light.
- Sensory Evaluation: While not a quantitative measurement, the aroma and taste of olive oil are excellent indicators of its quality. Fresh EVOO should have fruity, herbaceous, or grassy notes and a pleasant peppery finish. Rancid or stale notes (like crayons, putty, or stale nuts) suggest oxidation.
- Colour: While colour can vary depending on the cultivar and ripeness of the olives, it is not a reliable indicator of oxidation.
Frequently Asked Questions (FAQs)
Q1: What is the most important indicator of olive oil oxidation?
The peroxide value is considered a primary indicator of initial oxidation. However, a comprehensive assessment also includes secondary oxidation products like conjugated dienes and trienes, as well as the degradation of beneficial minor compounds.
Q2: Does olive oil go bad?
Yes, olive oil can go bad due to oxidation and other degradation processes. Its shelf life depends on factors like its quality, storage conditions, and exposure to heat, light, and air.
Q3: How can I prevent olive oil from oxidizing?
To prevent oxidation, store olive oil in a cool, dark place in an airtight container. Avoid prolonged exposure to heat and light. Using the oil within a reasonable timeframe after opening also helps.
Q4: Are all olive oils equally susceptible to oxidation?
No, different olive cultivars have varying levels of antioxidants (like polyphenols and tocopherols), which influence their oxidative stability. Oils with higher polyphenol content generally exhibit better resistance to oxidation.
Q5: Can I still use olive oil that has oxidized?
While oxidized olive oil is generally safe to consume in small amounts, it will have a significantly diminished flavour and aroma, and its beneficial health properties will be reduced. It is best to discard oil that has a strong rancid smell or taste.
Conclusion
Measuring olive oil oxidation involves a suite of chemical analyses, primarily focusing on peroxide value, conjugated dienes/trienes, fatty acid composition, and the integrity of minor antioxidant compounds like tocopherols and polyphenols. While heat and time are natural accelerators of oxidation, understanding the specific responses of different olive cultivars, such as Arbequina and Picual, provides valuable insights into their relative stability. By paying attention to quality indicators and proper storage practices, consumers can ensure they are enjoying the full flavour and health benefits of this exceptional oil.
If you want to read more articles similar to Olive Oil Oxidation: A Deep Dive, you can visit the Automotive category.