25/06/2003
When you light a fire, whether it's a bonfire in your garden, the fuel combusting in your car engine, or a simple candle illuminating a room, have you ever paused to consider the fundamental nature of what's actually happening? Is the material merely changing its form, or is something far more profound occurring? This question, seemingly simple, delves into the core principles of chemistry, differentiating between physical and chemical changes. The answer is unequivocal: burning is, without a doubt, a chemical change. This involves a complex chemical reaction, typically with oxygen, leading to the creation of entirely new substances and the significant release of energy in the form of heat and light. The original substance undergoes a complete transformation, altering its chemical composition and properties.
Understanding this distinction is crucial, not just for academics but for anyone with a keen interest in how the world around us works, especially when dealing with the mechanics and maintenance of vehicles where combustion is a daily occurrence. Let's delve deeper into why burning falls squarely into the category of a chemical change and explore the fascinating evidence that proves it.
- Burning: A Definitive Chemical Change
- Why Burning Is Not a Physical Change
- Key Indicators of a Chemical Change
- Chemical vs. Physical Changes: A Comparative Overview
- Reversibility: Chemical Changes Are Generally Harder to Reverse
- Beyond Burning: Other Chemical Transformations
- Separating Mixtures: A Physical Approach
- Frequently Asked Questions About Chemical Changes
- Q1: Is frying (or cooking) an egg a chemical change?
- Q2: Is cooking in general a physical or chemical change?
- Q3: Why is digestion of food a chemical change?
- Q4: Is milk souring a physical or chemical change?
- Q5: Is baking a cake a physical or chemical change?
- Q6: Are chemical changes always harder to reverse than physical changes?
- Conclusion
Burning: A Definitive Chemical Change
Burning is universally classified as a chemical change because it involves a chemical reaction that results in the formation of new substances with properties distinctly different from the original materials. This process is scientifically known as combustion, a rapid reaction between a substance (the fuel) and an oxidant, usually oxygen from the air, which produces heat and light.
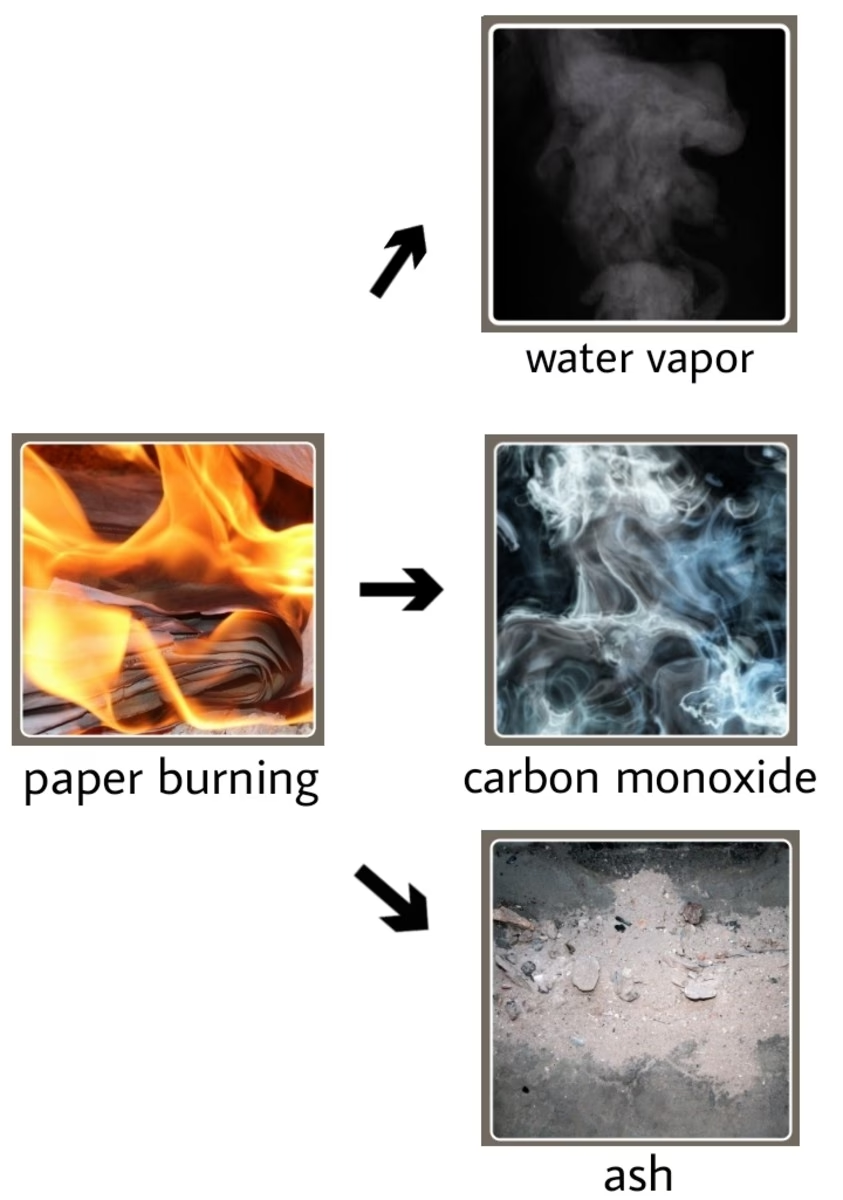
During combustion, the atoms within the reacting substances are rearranged. Chemical bonds in the original reactants are broken, and new chemical bonds are formed to create the products. For instance, when a hydrocarbon like wood or petrol burns, it reacts with oxygen. The carbon atoms in the fuel combine with oxygen to form carbon dioxide (CO₂), and the hydrogen atoms combine with oxygen to form water vapour (H₂O). These products – carbon dioxide and water – are entirely new compounds with different chemical formulas and properties compared to the original fuel and oxygen.
Consider the example of a log burning in a fireplace. Before burning, you have solid wood and gaseous oxygen. After burning, you are left with ash (primarily inorganic compounds), carbon dioxide gas, and water vapour, alongside the visible heat and light. The wood has not just changed its state; it has fundamentally changed its identity. You cannot simply cool the ash and gases to retrieve the original log. This irreversibility, or at least extreme difficulty in reversal, is a strong indicator of a chemical change.
The Role of Energy in Chemical Changes
Chemical changes like burning often involve significant energy transformations. Combustion is an exothermic reaction, meaning it releases energy, typically as heat and light, into the surroundings. This energy release is a direct consequence of the difference in energy stored in the chemical bonds of the reactants versus the products. The formation of new, more stable bonds in the products (like CO₂ and H₂O) releases excess energy, which we perceive as heat and light. This energy change is another defining characteristic that sets chemical changes apart from physical ones.
Why Burning Is Not a Physical Change
To fully grasp why burning is a chemical change, it's equally important to understand why it is definitively not a physical change. A physical change alters the physical properties of a substance, such as its shape, size, or state (solid, liquid, or gas), but crucially, it does not alter its chemical composition or identity. The molecules themselves remain unchanged.
Think about melting ice. When ice (solid water) melts, it turns into liquid water. Its state changes, but it is still water (H₂O). You can refreeze it back into ice, and it will still be H₂O. Similarly, boiling water turns liquid water into steam (gaseous water), but again, the chemical formula remains H₂O. Dissolving sugar in water is another physical change; the sugar molecules disperse throughout the water, but they don't transform into a new substance. You can evaporate the water, and the sugar will be left behind, still chemically sugar.
In contrast, burning involves the breaking and forming of chemical bonds, leading to the creation of entirely new substances. The distinct chemical properties of the byproducts of burning (carbon dioxide, water vapour, ash) are a clear testament to this transformation. You cannot simply reverse the process and get the original fuel back, highlighting the fundamental difference between a physical alteration and a chemical transformation.
Key Indicators of a Chemical Change
Since we can't directly observe atoms breaking and forming bonds, chemists rely on observable evidence to determine if a chemical change has occurred. These indicators are crucial for identifying chemical reactions in everyday life, from cooking to industrial processes. Here are the primary signs:
- Temperature Changes: A noticeable increase (exothermic reaction, like burning) or decrease (endothermic reaction) in temperature without external heating or cooling. The heat and light produced during combustion are prime examples.
- Light Given Off: The emission of light, often seen as a flame or glow, is a common sign of a chemical reaction, particularly combustion.
- Unexpected Colour Changes: The formation of a substance with a new, different colour, rather than just the mixing of original colours. For instance, a silver nail turning reddish-brown as it rusts.
- Bubbles are Formed (Gas Production): The appearance of gas bubbles indicates the formation of a new gaseous substance, provided the substance is not simply boiling. For example, mixing baking soda and vinegar produces carbon dioxide bubbles.
- Different Smell or Taste: The production of new substances with distinct odours or tastes. (Caution: Never taste chemistry experiments!) The smell of burning wood is distinctly different from fresh wood.
- A Solid Forms (Precipitate): When two clear liquids are mixed and a solid, insoluble substance forms and settles out of the solution, it's called a precipitate. This solid is a new compound.
- Difficulty in Reversibility: While not always absolute, chemical changes are generally much harder to reverse than physical changes, often requiring another chemical reaction or significant energy input.
Chemical vs. Physical Changes: A Comparative Overview
To further clarify the distinction, let's look at a comparative table outlining the core differences between physical and chemical changes:
| Feature | Physical Change | Chemical Change |
|---|---|---|
| Composition | Remains the same | Changes; new substances formed |
| Bonds | No bonds broken or formed | Bonds broken and/or formed |
| Energy Change | Minimal (e.g., phase change energy) | Significant (e.g., heat, light released/absorbed) |
| Reversibility | Generally easily reversible | Often difficult or impossible to reverse |
| Properties | Physical properties altered (shape, state) | Chemical and physical properties altered |
| Examples | Melting ice, boiling water, dissolving sugar, cutting paper | Burning wood, rusting iron, cooking an egg, digestion |
Reversibility: Chemical Changes Are Generally Harder to Reverse
One of the most practical ways to differentiate between physical and chemical changes is to consider their reversibility. Physical changes are often readily reversible. For example, you can melt an ice cube and then refreeze the water back into an ice cube. You can dissolve salt in water and then evaporate the water to recover the salt. These processes simply involve a change in state or dispersion, not a change in the chemical identity of the substance.
Chemical changes, on the other hand, typically lead to new substances that cannot be easily converted back to their original forms. Once wood has burned into ash and gases, you cannot simply reassemble those components to reconstruct the original wood. Similarly, once iron has rusted, it cannot be simply 'un-rusted' back into pure iron without a further, often complex, chemical process. This difficulty in reversal stems from the fundamental alteration of molecular structures and the formation of new chemical bonds. While some chemical reactions can be reversed, they usually require specific conditions, significant energy input, or another chemical reaction to do so, unlike the simple physical processes that reverse physical changes.
Beyond Burning: Other Chemical Transformations
The concept of chemical change extends far beyond just combustion. Many everyday occurrences are, in fact, chemical transformations:
- Rusting of Iron: Iron reacting with oxygen and water to form iron oxide (rust). This is a slow oxidation process, similar in principle to burning but without the rapid heat and light.
- Fermentation of Sugar: Yeast converting sugars into alcohol and carbon dioxide. This process is vital in brewing and baking, producing entirely new compounds.
- Digestion of Food: Enzymes in our bodies breaking down complex food molecules into simpler nutrients that the body can absorb. This involves a series of intricate chemical reactions.
- Souring of Milk: Bacteria converting lactose (milk sugar) into lactic acid, changing the taste and texture of milk.
- Baking a Cake: Ingredients like flour, sugar, eggs, and baking powder undergo chemical reactions when heated, causing the cake to rise and its texture and flavour to develop.
Each of these examples demonstrates the formation of new substances with different properties, solidifying their classification as chemical changes.
Separating Mixtures: A Physical Approach
While chemical changes alter the identity of substances, physical changes are often exploited to separate mixtures without changing the individual components. This highlights another practical application of understanding these distinctions:
- Distillation: Used to separate liquids with different boiling points, such as alcohol from water, or to purify water from dissolved salts. The more volatile component evaporates and is then condensed back into a liquid, but its chemical identity remains unchanged.
- Filtration: Separates insoluble solids from liquids by passing the mixture through a filter medium. The solid particles are trapped, while the liquid (filtrate) passes through. Again, no chemical transformation occurs; the components are merely physically separated.
These methods are prime examples of how understanding physical properties allows us to manipulate substances without altering their fundamental chemical makeup, a stark contrast to the profound changes seen in combustion.
Frequently Asked Questions About Chemical Changes
Q1: Is frying (or cooking) an egg a chemical change?
Yes, frying an egg is a chemical change. The heat causes the proteins in the egg white and yolk to denature and coagulate, forming new chemical bonds and altering their structure, texture, and appearance permanently. You cannot revert a cooked egg back to its raw state.
Q2: Is cooking in general a physical or chemical change?
Cooking often involves both, but predominantly chemical changes. While some aspects like melting butter are physical, most cooking processes, such as baking, frying, or boiling vegetables, involve chemical reactions. These reactions alter the starches, proteins, and fats, changing flavour, texture, and digestibility, creating new substances not easily reversed.
Q3: Why is digestion of food a chemical change?
Digestion is a complex series of chemical changes. Enzymes in your digestive system break down large, complex food molecules (like carbohydrates, proteins, and fats) into smaller, simpler molecules (sugars, amino acids, fatty acids). This process involves breaking and forming chemical bonds to create new substances that the body can absorb and utilise.
Q4: Is milk souring a physical or chemical change?
Souring milk is a chemical change. Bacteria in the milk consume lactose (milk sugar) and convert it into lactic acid. This new substance (lactic acid) causes the milk to curdle, thicken, and develop a sour taste, fundamentally altering its chemical composition and properties.
Q5: Is baking a cake a physical or chemical change?
Baking a cake is primarily a chemical change. The heat from the oven causes ingredients like flour, eggs, sugar, and baking powder to react with each other. Proteins coagulate, starches gelatinise, and leavening agents produce gases that make the cake rise. These reactions create a new product with a different structure, taste, and texture that cannot be easily reversed to the original ingredients.
Q6: Are chemical changes always harder to reverse than physical changes?
Generally, yes. Chemical changes typically involve the rearrangement of atoms and the formation of new chemical bonds, making reversal much more challenging and often requiring another chemical reaction or significant energy input. Physical changes, conversely, only alter a substance's form or state without changing its chemical identity, making them much easier to reverse.
Conclusion
In summary, burning is a quintessential example of a chemical change. It's a powerful process of combustion that transforms initial substances into entirely new ones, releasing energy in the process. Recognising the indicators of chemical change—such as changes in temperature, light emission, colour shifts, or the formation of gases or precipitates—helps us understand the dynamic transformations happening all around us. From the engine of your car to the simple act of lighting a match, chemical changes are at the heart of countless phenomena, constantly reshaping the world we live in.
If you want to read more articles similar to Burning: A Chemical Transformation Explained, you can visit the Automotive category.