19/03/2014
Fuel cells stand as a beacon of clean and efficient energy conversion, transforming chemical energy, typically from hydrogen, into electrical energy with remarkable efficacy and zero harmful emissions. This inherent environmental friendliness, coupled with growing concerns about primary energy supply, environmental protection, and energy sustainability, has propelled fuel cells into the spotlight in recent years. Among the diverse array of fuel cell technologies, the Polymer Electrolyte Membrane Fuel Cell (PEMFC) has emerged as a particularly promising candidate for automotive applications. Its advantages, including low noise operation, lower operating temperatures, and high power density, make it an attractive alternative to conventional internal combustion engines. Furthermore, PEMFCs are suitable for residential power generation, where both heat and power can be harnessed simultaneously, and even for portable electronics due to their high energy density.
A typical PEMFC comprises several key components: bipolar plates, gas diffusion layers (GDLs), catalyst layers (CLs), and the polymer electrolyte membrane (PEM). Hydrogen is supplied to the anode, while air or oxygen is fed to the cathode. The reactant gases traverse the GDLs, facilitating electrochemical reactions within the CLs. The sole by-product of this process is water, underscoring the environmentally benign nature of fuel cell technology.
- The Critical Role of Water Management in PEMFCs
- Humidification Strategies for PEMFCs
- Water Transport Mechanisms within a PEMFC
- Addressing Water Imbalance: Flooding vs. Drying Out
- Water Removal from PEM Fuel Cells
- Alternative Water Management Techniques
- Hydrogen Injection in Diesel Engines: A Related Concept
- Conclusion: The Importance of Water Balance
The Critical Role of Water Management in PEMFCs
Despite their significant advantages, the widespread adoption of fuel cells is still hampered by technical challenges, chief among them being water management. The performance and longevity of a fuel cell are intrinsically linked to the delicate balance of water within its structure. The polymer electrolyte membrane, the heart of the PEMFC, requires a specific level of hydration to conduct protons efficiently. Insufficient humidification, or operation in a dry state, can accelerate membrane degradation through radical formation and dehydration. Conversely, an excess of water can lead to a phenomenon known as water flooding. This occurs when water accumulates in the porous electrode structures, impeding the transport of reactant gases to the catalyst sites. Therefore, optimising the amount and distribution of water is paramount for achieving high proton conductivity, ensuring membrane durability, and facilitating reactant flow.
Humidification Strategies for PEMFCs
To address the complexities of water management, various techniques have been developed to humidify and control water levels within PEMFCs. These can be broadly categorised into internal and external humidification methods.
Internal Humidification Methods
Internal humidification strategies aim to manage water within the fuel cell stack itself. These methods can be further classified:
- Physical Methods: These often involve modifying the physical structure of the fuel cell components, such as adding microporous layers, optimising the pore structure of the GDL, or designing novel flow field structures. These physical modifications can influence how water is distributed and transported within the cell.
- Chemical Methods: While less commonly detailed in general overviews, chemical methods could potentially involve the use of specific additives or catalysts that influence water production or management, though physical methods are more prevalent in current research for direct water injection applications.
External Humidification Methods
External humidification involves supplying water to the fuel cell system from an external source. This is often necessary, especially when the water generated internally is insufficient to maintain optimal membrane hydration, particularly under high current densities. Common external methods include:
- Gas Bubbling Humidification: Reactant gases are passed through a water bath or bubbled through water to saturate them with moisture before entering the fuel cell.
- Direct Water (Liquid/Vapor) Injection: This method involves directly injecting liquid water or water vapour into the reactant gas streams. This is where the question of why direct water injection is used becomes most relevant. Direct injection offers precise control over the amount of water introduced, allowing for tailored humidification based on operating conditions. It can be more space-efficient than bubbling systems, making it attractive for applications where size and weight are critical, such as automotive. The finely atomised water droplets or vapour can effectively humidify the incoming gases, ensuring the membrane remains adequately hydrated without introducing excessive liquid water that could lead to flooding. This method can be particularly beneficial for rapidly adjusting humidity levels to meet changing power demands.
- Enthalpy Wheel Humidifiers: These devices use a rotating wheel to transfer heat and moisture between air streams, thereby humidifying the reactant gases.
- Membrane Humidification: This involves using a separate membrane to humidify the reactant gases, often employing a water reservoir.
- Exhaust Gas Recirculation: In some systems, a portion of the exhaust gas, which contains water vapour, is recirculated back into the system to help humidify the incoming reactants.
Water Transport Mechanisms within a PEMFC
Understanding how water moves within a PEMFC is crucial for effective management. Water is transported through several pathways:
- Gas Channels: Water can be carried along with the reactant gases through the flow channels.
- Membrane and Electrodes: Water can also diffuse through the membrane and the porous electrodes.
Two primary transport processes occur across the polymer membrane:
- Electro-osmotic Drag: As protons migrate from the anode to the cathode, they carry water molecules with them. The number of water molecules transported per proton is quantified by the electro-osmotic drag coefficient. This process contributes to water accumulation on the cathode side.
- Back Diffusion: A concentration gradient of water across the membrane can drive the diffusion of water from the cathode back towards the anode. This is a crucial mechanism for retaining water within the membrane, especially when internal humidification is insufficient.
The accumulation of water at the cathode is a result of both electro-osmotic drag and the direct production of water from the electrochemical reaction. Managing this accumulated water is key. Options include transporting it to the flow channels via the gas diffusion backing, evaporating it using heaters, or allowing it to diffuse back through the membrane towards the anode.

Addressing Water Imbalance: Flooding vs. Drying Out
The core challenge in PEMFC water management lies in preventing both flooding and drying out. The water content within the polymer membrane directly impacts its proton conductivity and the activation overpotentials. Insufficient hydration leads to decreased protonic conductivity and increased cell resistance, hindering performance. Conversely, excessive water can flood the electrodes and gas diffusion backings, blocking reactant pathways and reducing efficiency.
The delicate balance is influenced by several operational parameters:
- Current density
- Temperature
- Reactant flow rates
- Pressures
- Humidification levels
- Cell design
- Component materials
Water production at the cathode is directly proportional to the current density. Effective water removal strategies, such as optimising the gas diffusion layer, flow channel design, or employing heaters, are essential to mitigate water buildup.
Water Removal from PEM Fuel Cells
Water can be efficiently removed from a PEM fuel cell primarily through the reactant streams. Undersaturated reactant gases flowing through the flow channels can pick up liquid water. The rate of water removal and the proportion of liquid versus gaseous water are highly dependent on operating conditions such as temperature, pressure, and flow rates, as well as the cell's design. These parameters dictate the water vapour content of the gases and the rate of evaporation.
When the water generated at the cathode is insufficient to maintain adequate membrane hydration, particularly under high current demands where electro-osmotic drag might exceed back diffusion, humidified reactant gases become necessary. This is often exacerbated when using thicker polymer membranes, which require more water to remain hydrated. The humidification of the anode can be managed through hydrogen humidification, while cathode water management is more complex due to the combined effects of reaction water and electro-osmotic drag.
Gas diffusion layers play a vital role in facilitating water removal from the electrodes. Their porosity, thickness, and hydrophobicity significantly influence the water concentration gradient. Similarly, the geometry of the flow channels – their width, length, depth, and spacing – affects reactant flow velocity, diffusion rates, and the movement of liquid water within the cell.

Alternative Water Management Techniques
For specific applications, particularly small, portable ones, external gas humidifier subsystems might be undesirable due to added weight, size, complexity, and parasitic power consumption. Several alternative methods exist to control water within the fuel cell:
- Direct Water Humidification of the MEA: Directly applying water to the Membrane Electrode Assembly (MEA) can ensure consistent hydration.
- Thinner Membranes: Utilising thinner membranes can enhance water back diffusion from the cathode to the anode, aiding in internal water management.
- Internal Membrane Humidification: This might involve embedding hydrophilic catalyst particles or materials within the membrane to help retain moisture.
- Improvement of Gas Diffusion Layers: Designing GDLs with optimised porosity and controlled hydrophobicity can significantly improve water transport and removal.
In compact, portable fuel cell systems, a dead-ended bipolar design can be advantageous. In such configurations, the electro-osmotic drag and water back diffusion can be balanced, preventing water accumulation in the anode compartment.
Water management at the cathode is a complex interplay of temperature, cathode flow rate, channel structure, air flow, and the properties of the gas diffusion backing. While natural convection offers a simple solution for small, portable fuel cells, forced convection, often using a small fan or blower, can enhance overall fuel cell efficiency but complicates water management.
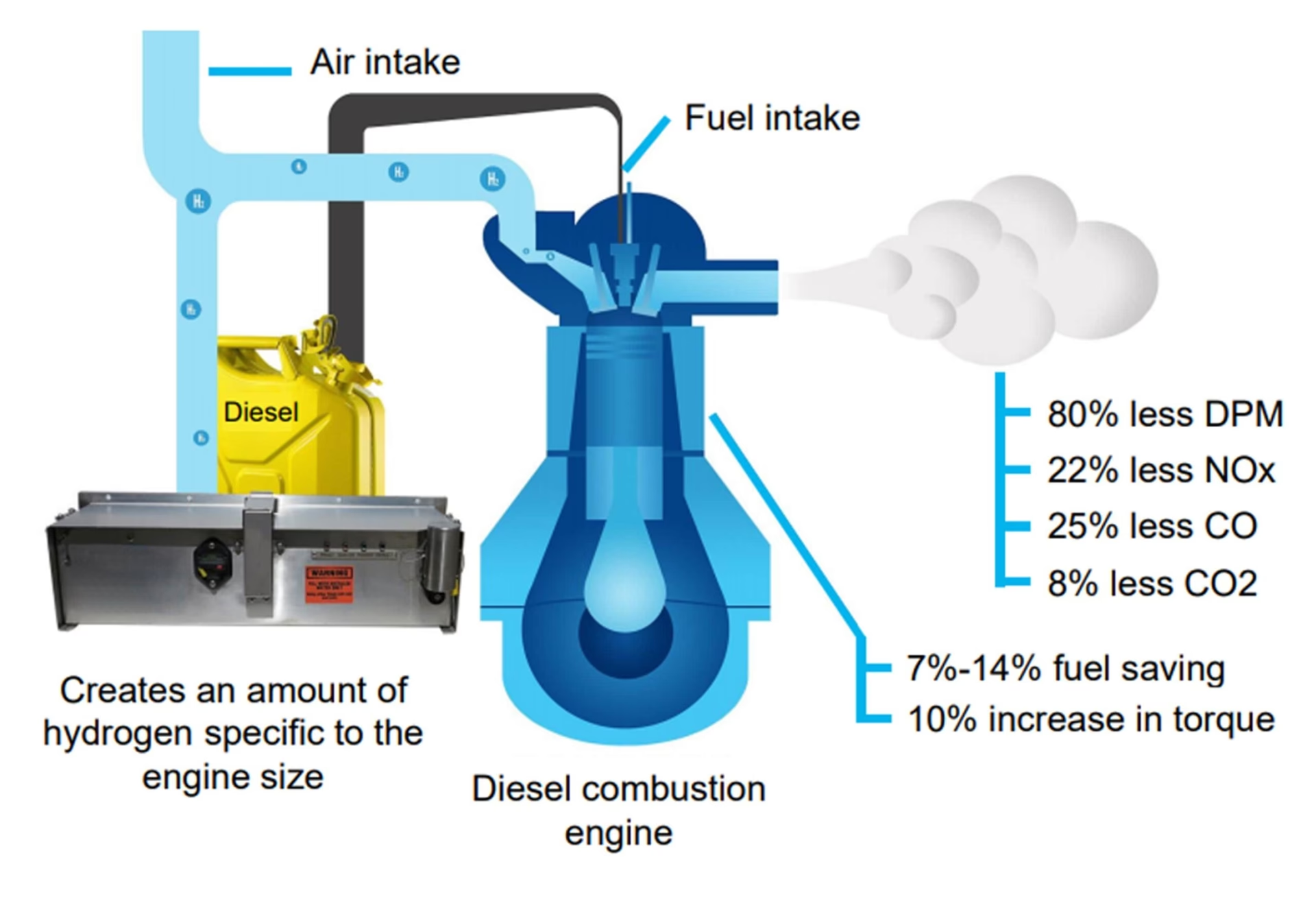
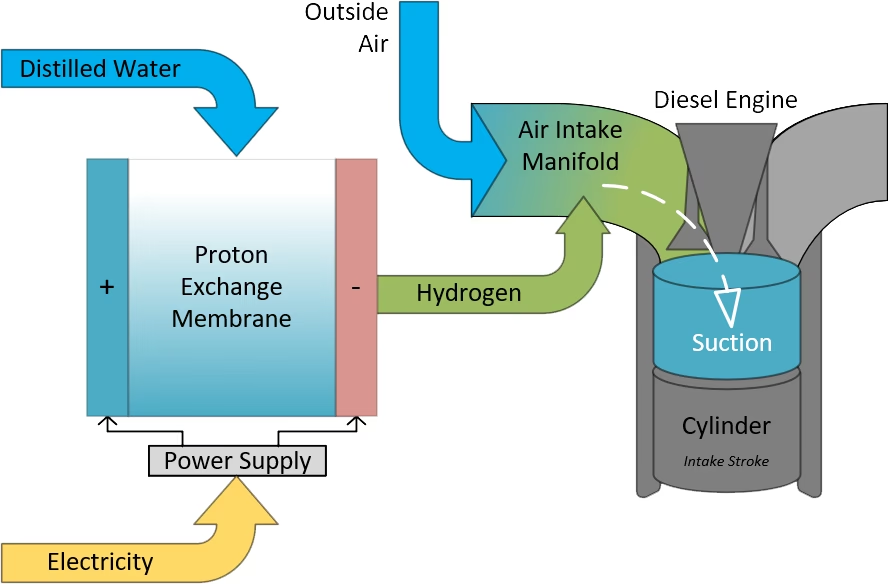
While distinct from fuel cell technology, the concept of hydrogen injection in diesel engines highlights another application of hydrogen in internal combustion engines. In these systems, hydrogen, often produced on-demand from deionised water, is introduced into the diesel engine's combustion process. This hydrogen enrichment can lead to lower emissions, increased power output, and reduced fuel consumption. Sophisticated electronics manage the hydrogen flow responsively to engine demand, optimising performance. Independent testing has indicated that hydrogen addition can significantly reduce harmful emissions, by up to 80%, and decrease fuel consumption by up to 14%. Such systems operate only when the engine is running, using only deionised water as the source for hydrogen generation.
Conclusion: The Importance of Water Balance
In conclusion, achieving the correct water balance is a fundamental design consideration for any fuel cell stack. Meticulous attention to water management can substantially increase the efficiency and power output of the fuel cell, whilst also ensuring the longevity of the stack. Key design parameters that must be carefully considered include the overall stack dimensions, nominal power requirements, flow field design, GDL porosity and hydrophobicity, the chosen humidification strategy, membrane thickness, and the integration of any external plant components. Direct water injection, when employed as an external humidification strategy, offers a precise and often space-efficient method to ensure optimal membrane hydration, thereby contributing significantly to the overall performance and reliability of PEM fuel cells.
If you want to read more articles similar to Direct Water Injection in Fuel Cells Explained, you can visit the Automotive category.