04/07/2008
Understanding whether a chemical compound is 'aromatic' is a cornerstone of organic chemistry, profoundly impacting a molecule's stability and reactivity. Aromatic compounds possess unique characteristics that set them apart, making them exceptionally stable compared to their non-aromatic counterparts. This enhanced stability is crucial in countless chemical reactions and biological processes. But how exactly does one determine if a compound qualifies for this special designation? It all comes down to a set of specific criteria, often referred to as Hückel's Rules, which provide a clear pathway to identifying these fascinating structures.
- The Four Pillars of Aromaticity: Hückel's Criteria
- The Mystique of 4n+2 π Electrons: Hückel's Molecular Orbital Theory
- Applying the 4n+2 Rule: A Practical Guide
- Deciphering π Electrons: Where Do They Reside?
- Aromatic Ions: Charge No Barrier to Aromaticity
- Heterocyclic Aromatic Compounds: Expanding the Ring
- Aromatic, Anti-aromatic, or Non-aromatic? A Comparative Overview
- Frequently Asked Questions About Aromaticity
The Four Pillars of Aromaticity: Hückel's Criteria
To ascertain if a compound is aromatic, you must meticulously evaluate it against four fundamental criteria. If a molecule fails to satisfy even one of these conditions, it cannot be classified as aromatic. These rules are derived from sophisticated molecular orbital theory but can be applied quite practically.
- The Molecule Must Be Cyclic: The atoms must form a closed ring structure. Open-chain compounds, regardless of other features, can never be aromatic. This circular arrangement is fundamental to the continuous overlap of p-orbitals, which is essential for aromaticity.
- The Molecule Must Be Planar: All atoms within the ring must lie in the same plane. This coplanarity allows for the effective side-by-side overlap of p-orbitals, forming a continuous cloud of electron density above and below the ring. If the ring is distorted or twisted, this essential overlap is disrupted, preventing aromaticity. For smaller rings, planarity is often inherent if the other conditions are met, but larger rings can adopt non-planar conformations.
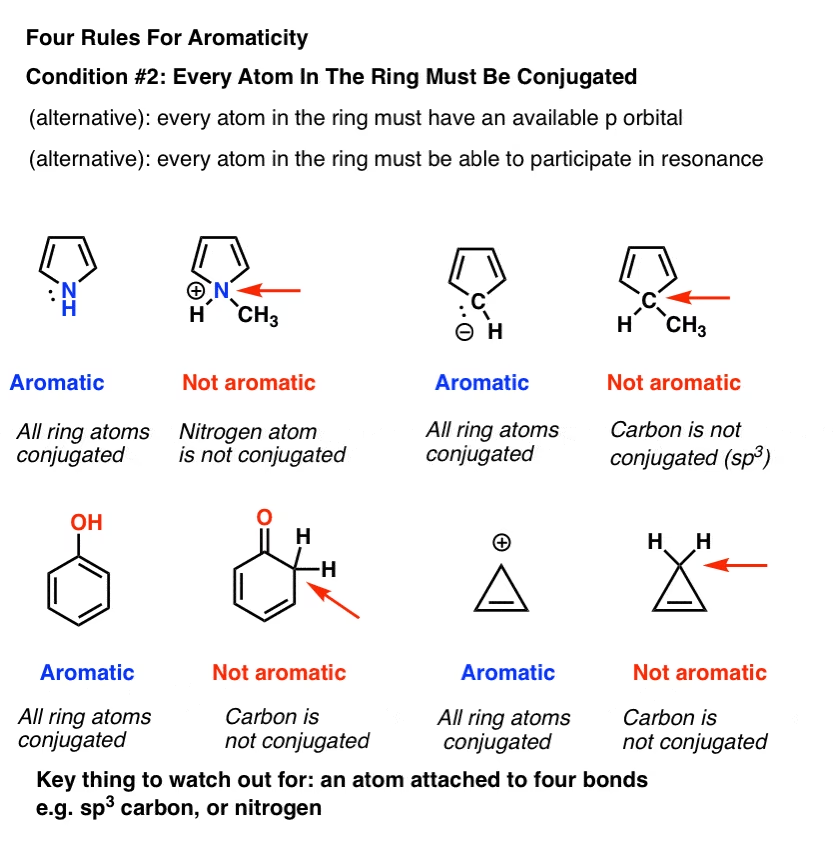
- The Molecule Must Be Fully Conjugated: There must be a continuous overlap of p-orbitals at every atom within the ring. This typically means that every atom in the ring must be sp2 or sp hybridised, each possessing an unhybridised p-orbital. This continuous system allows electrons to delocalise freely around the entire ring, a phenomenon central to aromatic stability. A break in this conjugation, such as an sp3 hybridised carbon within the ring, immediately disqualifies a molecule from being aromatic.
- The Molecule Must Possess (4n+2) π Electrons: This is perhaps the most famous and often misunderstood criterion, known as Hückel's Rule. The total number of pi (π) electrons in the cyclic, conjugated system must fit the formula 4n+2, where 'n' is any non-negative integer (0, 1, 2, 3, etc.). This rule dictates the specific number of electrons required for a stable, closed-shell electron configuration in the molecular orbitals of the ring.
The Mystique of 4n+2 π Electrons: Hückel's Molecular Orbital Theory
The 4n+2 rule isn't arbitrary; it stems directly from Hückel's Molecular Orbital Theory. This theory explains why certain cyclic, planar, and conjugated systems exhibit extraordinary stability. According to Hückel, a compound achieves particular stability when all its bonding molecular orbitals are completely filled with paired electrons, and no anti-bonding orbitals are occupied. This is precisely what happens in aromatic compounds.
In an aromatic system, the lowest energy molecular orbital is filled by 2 π electrons. Subsequent energy levels are then filled by 4 π electrons each. The 'n' in the 4n+2 rule represents the number of these subsequent energy levels that are fully occupied. This arrangement ensures that all bonding orbitals are filled, resulting in a highly stable, closed-shell electron configuration. Consider benzene, the quintessential aromatic compound. Benzene possesses 6 π electrons. The first 2 π electrons fill the lowest energy orbital, leaving 4 electrons. These 4 then fill the next energy level (where n=1), resulting in a total of 4(1)+2 = 6 π electrons. This perfect filling of bonding orbitals, with no electrons in anti-bonding orbitals, is why benzene is so incredibly stable.
Applying the 4n+2 Rule: A Practical Guide
To effectively apply Hückel's Rule, follow these straightforward steps:
- Count the π Electrons: Carefully determine the total number of π electrons participating in the cyclic, conjugated system. This is often the trickiest part, as we'll discuss shortly.
- Solve for 'n': Set the counted number of π electrons equal to 4n+2 and solve for 'n'.
- Verify 'n': If 'n' is 0 or any positive integer (1, 2, 3, ...), then the 4n+2 rule is satisfied. If 'n' is a fraction or a negative number, the rule is not met, and the compound is not aromatic (it might be anti-aromatic or non-aromatic).
Let's revisit benzene as an example. It has six π electrons:
4n + 2 = 6
4n = 4
n = 1
Since n=1, a positive integer, benzene perfectly adheres to Hückel's Rule, confirming its aromatic nature.
Deciphering π Electrons: Where Do They Reside?
Identifying which electrons qualify as π electrons is paramount. Essentially, π electrons are those that reside in unhybridised p-orbitals and are involved in π bonds or delocalised lone pairs. Here's how to identify them:
- Double and Triple Bonds: Each double bond (π bond) always contributes 2 π electrons. A triple bond contributes 4 π electrons (two π bonds).
- sp2 Hybridisation and p-Orbitals: Atoms that are sp2 hybridised each possess one unhybridised p-orbital. If every atom in a cyclic compound is sp2 hybridised, it means the molecule is fully conjugated, and the electrons within these p-orbitals are the π electrons.
- Identifying sp2 Hybridisation: A simple heuristic for identifying an sp2 hybridised atom is to look for an atom with three attached atoms and no lone pairs of electrons. For instance, in a cyclic hydrocarbon with alternating single and double bonds (like benzene), each carbon atom is typically attached to one hydrogen and two other carbons. This means each carbon is sp2 hybridised and contributes a p-orbital.
- Lone Pairs and Conjugation: Lone pairs of electrons can also contribute to the π system if they reside in a p-orbital and are part of the continuous conjugation. A crucial rule of thumb: If an atom has one or more lone pairs and is directly attached to an sp2 hybridised atom within the ring, it often becomes sp2 hybridised itself, allowing one of its lone pairs to occupy a p-orbital and participate in the aromatic system. However, only one lone pair from an atom can contribute to the aromaticity, as each sp2 atom only has one p-orbital available for the π system. Any additional lone pairs would reside in sp2 hybrid orbitals and would not be part of the π system.
Let's consider benzene again. It has three double bonds. Each double bond contributes 2 π electrons, so 3 double bonds x 2 electrons/bond = 6 π electrons. All carbons are sp2 hybridised and form a continuous conjugated system, confirming the count.
Aromatic Ions: Charge No Barrier to Aromaticity
Hückel's Rule is not exclusive to neutral molecules; it applies equally to ions. As long as the compound meets all four criteria, including the 4n+2 π electron count, its overall charge (positive or negative) does not preclude it from being aromatic. A classic example is the cyclopentadienyl anion.
The cyclopentadienyl anion is a five-membered ring with a negative charge. Let's examine its aromaticity:
- Cyclic: Yes, it's a ring.
- Planar: Yes, it's a small, symmetrical ring, typically planar.
- Fully Conjugated: Carbons 2 through 5 are sp2 hybridised (each has a double bond and is attached to three atoms). What about Carbon 1, which bears the negative charge and a lone pair? According to the rule mentioned earlier, if an atom with a lone pair is attached to an sp2 hybridised atom within the ring, it can become sp2 hybridised itself, with one lone pair occupying a p-orbital to complete the conjugation. Thus, Carbon 1 is also sp2 hybridised, and its lone pair occupies a p-orbital, making the ring fully conjugated.
- 4n+2 π Electrons: It has two double bonds (2 x 2 = 4 π electrons) plus the lone pair on Carbon 1 (2 π electrons). Total = 6 π electrons.
4n + 2 = 6
4n = 4
n = 1Since n=1, the cyclopentadienyl anion is indeed aromatic.
Heterocyclic Aromatic Compounds: Expanding the Ring
So far, our examples have focused on homocyclic rings composed solely of carbon atoms. However, aromaticity is not limited to carbon-only rings. Compounds incorporating one or more atoms other than carbon within the ring structure are called heterocyclic compounds, and many of them exhibit aromatic properties, provided they satisfy Hückel's criteria. Common heteroatoms include oxygen, nitrogen, and sulphur.
Consider furan, a five-membered heterocyclic ring containing an oxygen atom. Let's assess its aromaticity:
- Cyclic: Yes, it's a ring.
- Planar: Yes, it's a small, typically planar ring.
- Fully Conjugated: All four carbon atoms in furan are sp2 hybridised (each involved in a double bond or attached to three atoms within the ring). What about the oxygen atom? Oxygen has two lone pairs. Since it's attached to sp2 hybridised carbon atoms within the ring, one of its lone pairs can occupy a p-orbital to complete the cyclic conjugation. The other lone pair would reside in an sp2 hybrid orbital and not participate in the aromatic system. Thus, furan is fully conjugated.
- 4n+2 π Electrons: Furan has two double bonds (2 x 2 = 4 π electrons) plus one lone pair from the oxygen atom (2 π electrons). Total = 6 π electrons.
4n + 2 = 6
4n = 4
n = 1Since n=1, furan is a classic example of a heterocyclic aromatic compound.
Aromatic, Anti-aromatic, or Non-aromatic? A Comparative Overview
It's vital to distinguish between aromatic, anti-aromatic, and non-aromatic compounds, as their stabilities and reactivities differ dramatically. This table summarises the key distinctions:
| Characteristic | Aromatic | Anti-aromatic | Non-aromatic |
|---|---|---|---|
| Cyclic | Yes | Yes | No, or irrelevant |
| Planar | Yes | Yes | No |
| Fully Conjugated | Yes | Yes | No |
| π Electron Count | 4n+2 | 4n | Does not fit 4n+2 or 4n (or not conjugated) |
| Relative Stability | Highly Stable | Highly Unstable | Typical Stability |
Anti-aromatic compounds are particularly unstable due to having 4n π electrons in a cyclic, planar, fully conjugated system. Their electron configuration results in occupied anti-bonding molecular orbitals, leading to significant destabilisation. Non-aromatic compounds simply lack one or more of the first three criteria (cyclic, planar, fully conjugated) and thus do not experience the special stabilisation or destabilisation associated with aromatic or anti-aromatic systems.
Frequently Asked Questions About Aromaticity
- What if a molecule has multiple rings?
- For polycyclic compounds, aromaticity can apply to the entire fused system (e.g., naphthalene, anthracene) if the entire molecule is planar and fully conjugated and follows the 4n+2 rule for the total number of π electrons across the entire system. Sometimes, only individual rings might be considered aromatic, depending on the context and conjugation path.
- Can an sp3 hybridised atom be part of an aromatic ring?
- No. An sp3 hybridised atom lacks an unhybridised p-orbital, thereby breaking the continuous conjugation of the ring. This immediately makes the molecule non-aromatic, regardless of other factors.
- How do I know if a molecule is planar?
- For smaller rings (up to 7 or 8 members), if it's fully conjugated, it's often planar or can achieve planarity. Larger rings might twist to avoid steric hindrance or angle strain, thus breaking planarity and preventing aromaticity. This often requires more advanced analysis or knowledge of specific compound structures.
- Do all lone pairs contribute to π electrons?
- No. Only lone pairs that reside in a p-orbital and are part of the continuous, cyclic conjugation count as π electrons. An atom can only contribute one p-orbital to the π system. If an atom has multiple lone pairs (like oxygen in furan), only one will participate; the others will be in hybrid orbitals (e.g., sp2) and not be part of the π system.
- What is the significance of aromaticity?
- Aromatic compounds are exceptionally stable, which influences their reactivity. They often undergo substitution reactions rather than addition reactions, preserving their aromatic character. This stability is vital in many natural products, pharmaceuticals, and industrial chemicals, making aromaticity a cornerstone concept in chemistry.
Mastering the criteria for aromaticity is a fundamental skill in organic chemistry. By systematically checking for cyclic structure, planarity, full conjugation, and the critical 4n+2 π electron count, you can confidently identify these remarkably stable compounds. This understanding not only deepens your appreciation for molecular structure but also provides invaluable insights into chemical reactivity and the vast world of organic compounds.
If you want to read more articles similar to Aromaticity: Understanding Hückel's Rules, you can visit the Automotive category.
