24/02/2011
- The Molecular Orbital Diagram of Nitrogen: A Deep Dive
- Understanding Atomic Orbitals and the Basis of MO Theory
- Constructing the Molecular Orbital Diagram for N₂
- The Complete MO Diagram for N₂
- Visual Representation (Conceptual Description)
- Calculating Bond Order
- Magnetic Properties of N₂
- Comparison with Other Diatomic Molecules
- Why is Nitrogen So Stable and Unreactive?
- Frequently Asked Questions (FAQs)
- Conclusion
The Molecular Orbital Diagram of Nitrogen: A Deep Dive
The nitrogen molecule, N₂, is a cornerstone of atmospheric chemistry and a fundamental building block in countless organic and inorganic compounds. Its remarkable stability, a direct consequence of its electronic configuration, is often best understood through the lens of molecular orbital (MO) theory. A molecular orbital diagram for nitrogen offers a powerful visual representation of how atomic orbitals combine to form molecular orbitals, and crucially, how electrons occupy these new energy levels. This diagram is not merely an academic exercise; it provides profound insights into the nature of the N-N triple bond, the molecule's diamagnetic nature, and its relatively low reactivity.
Understanding Atomic Orbitals and the Basis of MO Theory
Before we delve into the specifics of nitrogen, it's essential to recall the basics of atomic orbitals. For nitrogen (atomic number 7), the electronic configuration is 1s²2s²2p³. This means each nitrogen atom possesses electrons in the 1s, 2s, and 2p subshells. According to molecular orbital theory, when two atoms approach each other to form a molecule, their atomic orbitals (AOs) can combine to form new molecular orbitals (MOs). These MOs are delocalised over the entire molecule, unlike AOs which are localised on individual atoms. The number of MOs formed is always equal to the number of AOs that combine. These MOs can be either:
- Bonding Molecular Orbitals (BMOs): These are formed by the constructive interference of atomic orbitals. They have lower energy than the parent AOs and lead to increased electron density between the nuclei, thus strengthening the bond.
- Antibonding Molecular Orbitals (ABMOs): These are formed by the destructive interference of atomic orbitals. They have higher energy than the parent AOs and have a node (a region of zero electron density) between the nuclei, weakening the bond.
The filling of these MOs with electrons follows the same principles as filling AOs: the Aufbau principle, Hund's rule, and the Pauli exclusion principle.
Constructing the Molecular Orbital Diagram for N₂
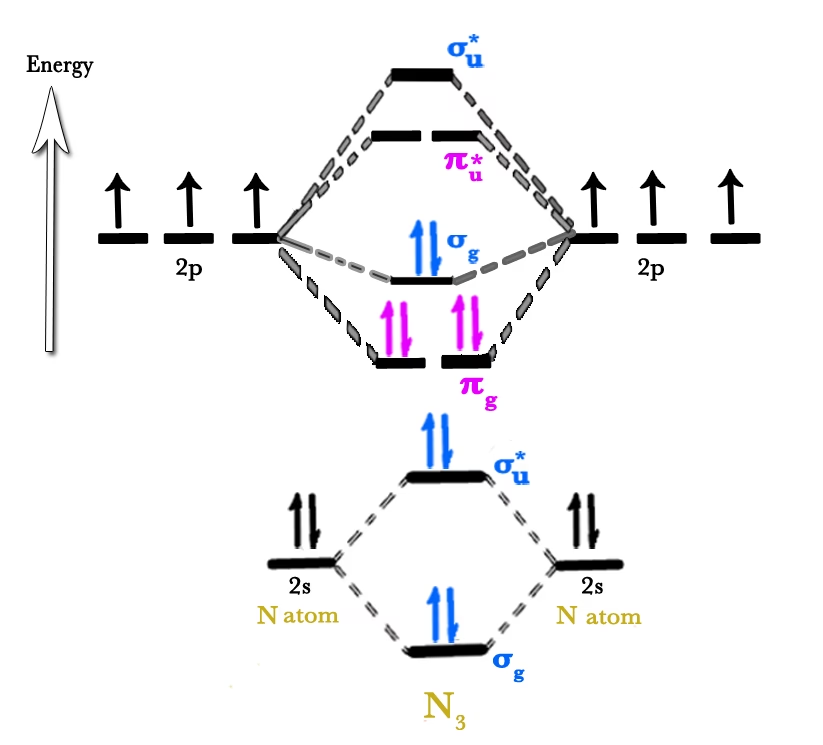
To construct the MO diagram for N₂, we consider the combination of atomic orbitals from two nitrogen atoms. We will focus on the valence orbitals, as the 1s orbitals are core electrons and their contribution to bonding is minimal. The valence atomic orbitals of each nitrogen atom are 2s and 2p.
1. Combination of 2s Atomic Orbitals
The two 2s atomic orbitals, one from each nitrogen atom, combine to form two molecular orbitals:
- σ₂s (bonding): This is formed by the in-phase overlap of the two 2s AOs. It is lower in energy than the 2s AOs and has increased electron density between the nuclei.
- σ₂s* (antibonding): This is formed by the out-of-phase overlap of the two 2s AOs. It is higher in energy than the 2s AOs and has a nodal plane between the nuclei.
2. Combination of 2p Atomic Orbitals
The combination of the 2p atomic orbitals is slightly more complex due to the possibility of head-on (sigma) and side-on (pi) overlap. For diatomic molecules of second-period elements up to and including nitrogen, there is significant s-p mixing, which results in the σ₂p orbital being higher in energy than the π₂p orbitals. This is a crucial point for understanding the N₂ molecule.
The 2p orbitals can combine in two ways:
- Head-on overlap: The two 2pz orbitals (assuming the internuclear axis is the z-axis) overlap end-to-end to form sigma molecular orbitals.
- Side-on overlap: The two 2px and the two 2py orbitals overlap side-by-side to form pi molecular orbitals.
Due to s-p mixing, the energy ordering of the molecular orbitals derived from the 2p atomic orbitals for N₂ is:
- π₂px and π₂py (bonding): These are degenerate (have the same energy) bonding orbitals formed from the side-on overlap of the 2px and 2py AOs.
- σ₂pz (bonding): This bonding orbital is formed from the head-on overlap of the 2pz AOs. In N₂, this orbital is actually at a higher energy than the π₂p orbitals due to s-p mixing.
- σ₂pz* (antibonding): This antibonding orbital arises from the out-of-phase overlap of the 2pz AOs.
- π₂px* and π₂py* (antibonding): These are degenerate antibonding orbitals formed from the out-of-phase side-on overlap of the 2px and 2py AOs.
The Complete MO Diagram for N₂
Combining these, the molecular orbital diagram for N₂ shows the following energy ordering from lowest to highest:
σ₂s < σ₂s* < π₂px = π₂py < σ₂pz < π₂px* = π₂py* < σ₂pz*
Each nitrogen atom contributes 5 valence electrons (2 from 2s and 3 from 2p), for a total of 10 valence electrons in the N₂ molecule. Let's fill these MOs according to the Aufbau principle, Hund's rule, and the Pauli exclusion principle:
- σ₂s: 2 electrons
- σ₂s*: 2 electrons
- π₂px: 2 electrons
- π₂py: 2 electrons
- σ₂pz: 2 electrons
This accounts for all 10 valence electrons. The remaining antibonding orbitals (π₂px*, π₂py*, and σ₂pz*) are empty.
Visual Representation (Conceptual Description)
Imagine two columns on the sides representing the atomic orbitals of each nitrogen atom (2s and 2p orbitals). In the centre, these combine to form the molecular orbitals. The 2s AOs combine to form the lower-energy σ₂s and higher-energy σ₂s* MOs. The 2p AOs combine to form the degenerate π₂p bonding orbitals, then the σ₂pz bonding orbital (higher in energy than π₂p), followed by the degenerate π₂p* antibonding orbitals, and finally the highest energy σ₂pz* antibonding orbital. Electrons are then filled into these MOs from the bottom up.
Calculating Bond Order
A key piece of information derived from the MO diagram is the bond order, which indicates the strength and stability of the bond. It is calculated as:
Bond Order = ½ (Number of electrons in bonding MOs - Number of electrons in antibonding MOs)
For N₂:
- Bonding electrons: 2 (in σ₂s) + 2 (in π₂px) + 2 (in π₂py) + 2 (in σ₂pz) = 8
- Antibonding electrons: 2 (in σ₂s*) = 2
- Bond Order = ½ (8 - 2) = ½ (6) = 3
A bond order of 3 confirms the presence of a triple bond between the two nitrogen atoms, which is consistent with experimental observations and explains the high bond dissociation energy of N₂.
Magnetic Properties of N₂
The MO diagram also helps predict the magnetic properties of a molecule. Molecules with unpaired electrons are paramagnetic (attracted to a magnetic field), while molecules with all electrons paired are diamagnetic (weakly repelled by a magnetic field).
In the N₂ molecule, all 10 valence electrons are paired in the molecular orbitals (two in σ₂s, two in σ₂s*, two in π₂px, two in π₂py, and two in σ₂pz). Therefore, N₂ is diamagnetic, which is experimentally verified.
Comparison with Other Diatomic Molecules
It's instructive to compare the MO diagram of N₂ with those of other second-period diatomic molecules. The ordering of MOs can change, significantly impacting bond order and magnetic properties. For instance, O₂ and F₂ have a different energy ordering where the σ₂pz orbital is lower in energy than the π₂p orbitals due to less s-p mixing. This leads to unpaired electrons in O₂, making it paramagnetic.
| Molecule | Valence Electron Configuration (MOs) | Bond Order | Magnetic Property |
|---|---|---|---|
| Li₂ | (σ₂s)² (σ₂s*)¹ | 1 | Paramagnetic |
| Be₂ | (σ₂s)² (σ₂s*)² | 0 | Diamagnetic |
| B₂ | (σ₂s)² (σ₂s*)² (π₂px)¹ (π₂py)¹ | 1 | Paramagnetic |
| C₂ | (σ₂s)² (σ₂s*)² (π₂px)² (π₂py)² | 2 | Diamagnetic |
| N₂ | (σ₂s)² (σ₂s*)² (π₂px)² (π₂py)² (σ₂pz)² | 3 | Diamagnetic |
| O₂ | (σ₂s)² (σ₂s*)² (σ₂pz)² (π₂px)² (π₂py)² (π₂px*)¹ (π₂py*)¹ | 2 | Paramagnetic |
| F₂ | (σ₂s)² (σ₂s*)² (σ₂pz)² (π₂px)² (π₂py)² (π₂px*)² (π₂py*)² | 1 | Diamagnetic |
Why is Nitrogen So Stable and Unreactive?
The molecular orbital diagram for nitrogen provides a clear explanation for its exceptional stability and low reactivity. The triple bond (bond order of 3) requires a significant amount of energy to break. The bond dissociation energy for N₂ is approximately 945 kJ/mol, one of the highest for any diatomic molecule. This strong triple bond means that breaking it requires substantial activation energy, making reactions involving the direct breaking of the N-N bond difficult under normal conditions. This is why nitrogen gas (N₂) is so inert and why processes like nitrogen fixation (converting N₂ into usable nitrogen compounds) are industrially important and require harsh conditions.
Frequently Asked Questions (FAQs)
1. What is the role of s-p mixing in the N₂ MO diagram?
S-p mixing is the interaction between the 2s and 2p atomic orbitals. In molecules like N₂, this interaction causes the σ₂pz molecular orbital to be pushed to a higher energy level than the π₂p molecular orbitals. This specific ordering is crucial for explaining the electronic configuration and diamagnetic nature of N₂.
2. How many valence electrons does a nitrogen molecule have?
Each nitrogen atom has 5 valence electrons (2s²2p³). Therefore, a diatomic nitrogen molecule (N₂) has a total of 10 valence electrons.
3. What is the bond order of N₂?
The bond order of N₂ is 3, indicating a strong triple covalent bond between the two nitrogen atoms.
4. Is N₂ paramagnetic or diamagnetic?
N₂ is diamagnetic because all of its electrons in the molecular orbitals are paired.
5. Why is nitrogen gas considered unreactive?
The high bond order (triple bond) of N₂ means that a large amount of energy is required to break the N-N bond, making it very stable and unreactive under normal conditions.
Conclusion
The molecular orbital diagram of nitrogen is a powerful tool that illuminates the fundamental nature of the N₂ molecule. It explains the existence of a strong triple bond, the molecule's diamagnetic character, and its characteristic low reactivity. By understanding how atomic orbitals combine to form molecular orbitals, we gain a deeper appreciation for the stability and chemical behaviour of one of Earth's most abundant gases. The principles illustrated by the N₂ MO diagram are foundational for comprehending chemical bonding across a wide range of molecules.
If you want to read more articles similar to Nitrogen's Molecular Orbital Diagram Explained, you can visit the Automotive category.