08/02/2019
Understanding the Molecular Orbital Diagram of Hydrogen Fluoride (HF)
The formation of chemical bonds is a cornerstone of chemistry, explaining how atoms unite to create the molecules that surround us. While homonuclear diatomic molecules, like O2 or N2, involve the combination of identical atomic orbitals, heteronuclear diatomic molecules present a more nuanced picture. These molecules, composed of two different atoms, exhibit unique bonding characteristics due to the disparity in the energies of their constituent atomic orbitals. Hydrogen Fluoride (HF) serves as an excellent case study for exploring these principles, offering insights into polar covalent bonding.
The Nature of Heteronuclear Diatomic Molecules
In heteronuclear diatomic molecules, the atomic orbitals from the two different atoms combine to form molecular orbitals. However, unlike their homonuclear counterparts, these atomic orbitals possess unequal energies. This energy difference arises from the varying electronegativity of the constituent atoms. Electronegativity, a measure of an atom's ability to attract shared electrons in a chemical bond, plays a crucial role in determining the polarity of the resulting bond and the characteristics of the molecular orbitals.
For atomic orbitals to effectively combine and form molecular orbitals, they must meet two key criteria: compatible symmetry and similar energy. When atomic orbitals of the same symmetry exhibit significant energy differences, their combination becomes less favourable. A general rule of thumb suggests that if the energy difference between atomic orbitals exceeds approximately 10-14 eV, they will not combine significantly. This principle is vital when constructing molecular orbital diagrams.
A fundamental concept in molecular orbital theory is that the closer a molecular orbital's energy is to that of a particular atomic orbital, the greater the contribution of that atomic orbital to the molecular orbital. This relationship allows us to predict the 'character' of molecular orbitals – whether they are predominantly derived from one atom or the other.
Orbital Ionization Energies: The Key to Understanding
To accurately generate a molecular orbital diagram for a heteronuclear diatomic molecule like HF, a thorough understanding of the relative energies of electrons in different atomic orbitals is paramount. This information is typically obtained through orbital ionization energies, which represent the energy required to remove an electron from a specific atomic orbital. There are two primary approaches to obtaining this crucial data:
- Consulting Tables of Ionization Energies: Detailed tables, often found in chemistry textbooks or online resources, provide experimentally determined ionization energies for various atomic orbitals. These tables offer precise values that are essential for accurate diagram construction.
- Utilising Periodic Trends: In situations where direct data is unavailable, chemists can approximate relative orbital energies by considering periodic trends. Factors like electronegativity and ionization energy trends across the periodic table can provide valuable clues about the relative stability of electrons in different atomic orbitals. For instance, more electronegative atoms tend to have lower ionization energies for their valence electrons, indicating that these electrons are held more tightly and are at a lower energy level.
Constructing the HF Molecular Orbital Diagram
Let's now apply these principles to the specific case of Hydrogen Fluoride (HF). HF is a molecule formed between hydrogen (H) and fluorine (F). We need to consider the atomic orbitals of both hydrogen and fluorine and how they interact.
Atomic Orbitals of Hydrogen and Fluorine
Hydrogen (H): Hydrogen has a single electron in its 1s atomic orbital. The energy of this 1s orbital is relatively high compared to the core orbitals of fluorine.
Fluorine (F): Fluorine is in the second period and has the electron configuration 1s²2s²2p⁵. For bonding in HF, we are primarily concerned with the valence orbitals: the 2s and 2p orbitals. The fluorine 2s orbital is significantly lower in energy than the hydrogen 1s orbital due to fluorine's high electronegativity. The fluorine 2p orbitals (2px, 2py, 2pz) are also lower in energy than the hydrogen 1s orbital, but their interaction with the hydrogen 1s orbital is crucial for bond formation.
Orbital Energy Levels (Approximate Values)
To construct the diagram, we need approximate energy levels. While precise values can be found in tables, general trends are sufficient for understanding:
- Hydrogen 1s: Relatively high energy.
- Fluorine 2s: Significantly lower energy than H 1s.
- Fluorine 2p: Lower energy than H 1s, but higher than F 2s.
The electronegativity difference between fluorine and hydrogen dictates these energy differences. Fluorine is much more electronegative, pulling electron density towards itself.
Molecular Orbital Formation
The molecular orbital diagram is built by combining the atomic orbitals of H and F that have compatible symmetry and reasonably similar energies. The key interactions are:
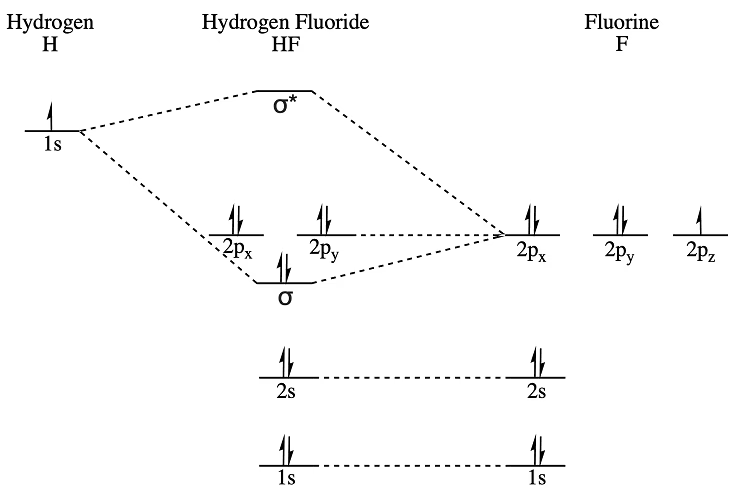
- F 2s and H 1s: These orbitals have the correct symmetry (sigma, σ) for interaction. However, due to the large energy difference between the fluorine 2s and hydrogen 1s orbitals, the interaction is weak. The F 2s orbital contributes more to the lower-energy bonding molecular orbital, and the H 1s contributes more to the higher-energy antibonding molecular orbital.
- F 2p and H 1s: The hydrogen 1s orbital can interact with the fluorine 2pz orbital (assuming the internuclear axis is along the z-axis). This interaction leads to the formation of a sigma (σ) bonding molecular orbital and a sigma-antibonding (σ*) molecular orbital. The F 2pz orbital is lower in energy, so it contributes more to the σ bonding orbital, while the H 1s contributes more to the σ* antibonding orbital. The F 2px and 2py orbitals are perpendicular to the internuclear axis and do not have sigma symmetry, so they do not form sigma bonds with the H 1s orbital. Instead, they form pi (π) molecular orbitals, but in HF, these are non-bonding or very weakly interacting with hydrogen's orbitals, often considered non-bonding for simplicity in introductory diagrams.
The HF Molecular Orbital Diagram Explained
The resulting molecular orbital diagram for HF typically shows the following levels, from lowest to highest energy:
- σ (from F 2s and H 1s): A low-energy bonding molecular orbital formed from the weaker interaction of F 2s and H 1s. This orbital is primarily localized on the fluorine atom.
- σ (from F 2pz and H 1s): A bonding molecular orbital formed from the stronger interaction of F 2pz and H 1s. This is the main bonding orbital responsible for holding the HF molecule together. It has significant character from both H 1s and F 2pz.
- Non-bonding π orbitals (from F 2px and F 2py): These orbitals are essentially the fluorine 2px and 2py orbitals that do not interact significantly with the hydrogen 1s orbital. They are considered non-bonding and are localized on the fluorine atom.
- σ* (from F 2pz and H 1s): An antibonding molecular orbital formed from the cancellation of the bonding σ orbital. It is higher in energy and has a node between the nuclei.
- σ* (from F 2s and H 1s): A higher-energy antibonding molecular orbital formed from the cancellation of the lower-energy σ orbital.
The valence electrons of HF are 1 (from H) + 7 (from F) = 8 electrons. These electrons fill the molecular orbitals according to the Aufbau principle, Hund's rule, and the Pauli exclusion principle. The filling order would be:
- The low-energy σ (F 2s/H 1s) orbital is filled with 2 electrons.
- The main bonding σ (F 2pz/H 1s) orbital is filled with 2 electrons.
- The two non-bonding π orbitals are filled with 4 electrons.
This leaves the antibonding orbitals (σ* and σ*) empty. The presence of electrons primarily in bonding and non-bonding orbitals, with the absence of electrons in antibonding orbitals, indicates a stable molecule.
Bond Polarity in HF
The molecular orbital diagram clearly illustrates why HF is a polar molecule. The bonding molecular orbital formed from the interaction of F 2pz and H 1s has a greater contribution from the more electronegative fluorine atom. Similarly, the non-bonding orbitals are localized on fluorine. This unequal distribution of electron density results in a partial negative charge (δ-) on the fluorine atom and a partial positive charge (δ+) on the hydrogen atom, creating a significant dipole moment.
Comparison of Bonding in HF and H2
Comparing the bonding in HF to that in H2 highlights the impact of electronegativity differences:
| Feature | H2 (Homonuclear) | HF (Heteronuclear) |
|---|---|---|
| Atomic Orbitals Involved | H 1s + H 1s | H 1s + F 2s, F 2p |
| Bond Type | Nonpolar Covalent | Polar Covalent |
| Molecular Orbitals Formed | σ (bonding), σ* (antibonding) | σ (bonding from F 2s), σ* (from F 2s); σ (bonding from F 2pz), σ* (from F 2pz); Non-bonding π |
| Electron Distribution | Symmetrical | Asymmetrical, towards Fluorine |
| Dipole Moment | Zero | Significant |
Frequently Asked Questions (FAQs)
Q1: What is the primary reason for the polarity of the HF bond?
A1: The significant difference in electronegativity between fluorine and hydrogen leads to an unequal sharing of electrons in the molecular orbitals, with electron density being pulled towards the more electronegative fluorine atom.
Q2: Which atomic orbitals are most important for the bonding in HF?
A2: The primary bonding interaction occurs between the hydrogen 1s atomic orbital and the fluorine 2pz atomic orbital, forming a sigma (σ) bonding molecular orbital.
Q3: Are there any non-bonding orbitals in HF?
A3: Yes, the fluorine 2px and 2py atomic orbitals do not participate in sigma bonding with hydrogen and are considered non-bonding molecular orbitals in the HF diagram.
Q4: How does the molecular orbital diagram explain the stability of HF?
A4: The diagram shows that the valence electrons fill bonding and non-bonding molecular orbitals, while the higher-energy antibonding orbitals remain empty. This net filling of bonding orbitals indicates a stable molecule.
Q5: Why is the interaction between H 1s and F 2s weaker than between H 1s and F 2p?
A5: The significant energy difference between the hydrogen 1s orbital and the fluorine 2s orbital makes their interaction less effective compared to the closer energy match between the hydrogen 1s and fluorine 2p orbitals.
Conclusion
The molecular orbital diagram for Hydrogen Fluoride provides a sophisticated explanation for its polar covalent bond. By considering the interactions between the atomic orbitals of hydrogen and fluorine, and accounting for their differing energy levels and symmetries, we can understand the formation of stable bonding and non-bonding molecular orbitals. This theoretical framework not only explains the observed polarity of the HF molecule but also serves as a powerful tool for understanding the chemical bonding in a wide range of heteronuclear diatomic molecules.
If you want to read more articles similar to Unveiling the HF Molecular Orbital Diagram, you can visit the Automotive category.
