22/09/2004
In the early 20th century, the world faced a critical challenge: feeding a rapidly growing population. The key to unlocking agricultural productivity lay in nitrogen, a vital nutrient for plant growth. However, atmospheric nitrogen, abundant as it is, exists in a largely unreactive form, inaccessible to most organisms. This immense chemical barrier sparked a global race to develop methods for 'fixing' nitrogen, transforming it into usable compounds. Amidst this scientific fervour, two Norwegian visionaries, Kristian Birkeland and Sam Eyde, pioneered an ingenious industrial process that, for a time, led the charge in synthetic fertiliser production.
- The Birth of an Industrial Solution
- Unravelling the Ingenious Process: How it Worked
- Harnessing Norway's Natural Power: The Energy Demands
- A Footprint in History: Rjukan and Notodden
- The March of Progress: Why the Birkeland-Eyde Process Faded
- The Enduring Legacy
- Frequently Asked Questions (FAQs)
- Who invented the Birkeland-Eyde process?
- What was the primary purpose of the Birkeland-Eyde process?
- Why was the Birkeland-Eyde process so energy-intensive?
- What replaced the Birkeland-Eyde process?
- Where were the Birkeland-Eyde factories located?
- What is 'Norwegian saltpeter'?
- Was the Birkeland-Eyde process environmentally friendly?
The Birth of an Industrial Solution
Developed in 1903, the Birkeland-Eyde process represented a monumental leap in industrial chemistry. Its primary purpose was to convert atmospheric nitrogen (N₂) and oxygen (O₂) into nitric acid (HNO₃), a crucial precursor for manufacturing synthetic fertilisers. Before this, humanity relied heavily on natural sources like guano and Chilean saltpetre, which were finite and often located far from agricultural hubs. The ability to create nitrogen-based fertilisers synthetically promised to revolutionise food production and significantly reduce reliance on distant, dwindling resources.
Unravelling the Ingenious Process: How it Worked
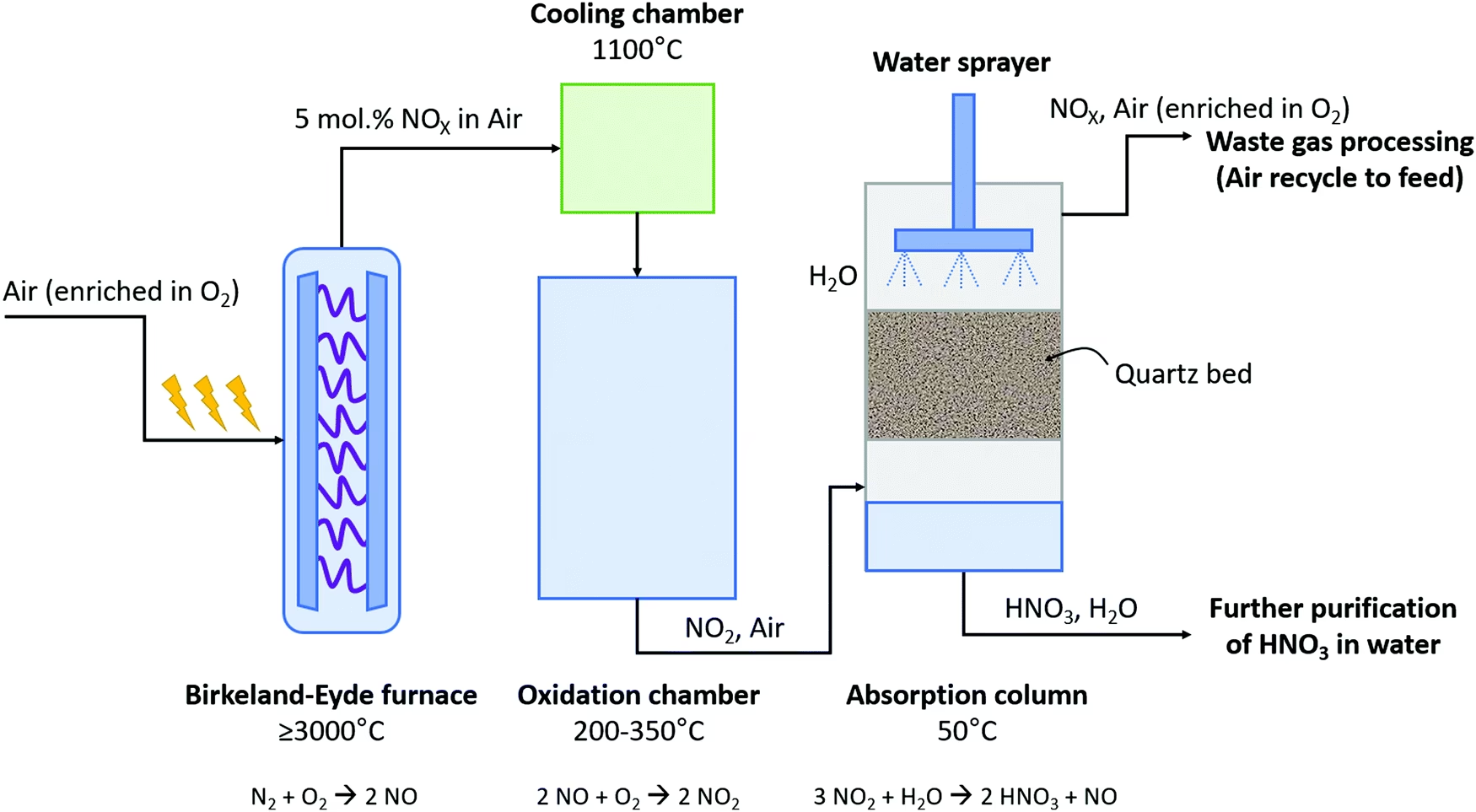
The Birkeland-Eyde process was a multi-step reaction, demanding immense energy but offering a groundbreaking solution. It drew inspiration from natural phenomena, specifically the way lightning naturally fixes nitrogen in the atmosphere.
Step 1: The Electric Arc Reactor – Forging Nitric Oxide
At the heart of the process was a specially designed electric arc furnace. Here, an electrical arc was formed between two coaxial, water-cooled copper tube electrodes. This arc was powered by a high voltage alternating current, typically 5 kV at 50 Hz. To enhance the reaction, a powerful static magnetic field, generated by a nearby electromagnet, was employed. This magnetic field, utilising the Lorentz force, spread the intense arc into a thin, disc-like plasma. This innovative setup was based on earlier experiments by Julius Plücker in 1861, who demonstrated how to create a disc of sparks with electromagnets, and later replicated by Walther Nernst and others.
Within this plasma disc, temperatures soared, often exceeding 3000 °C. Air, a readily available mixture of nitrogen and oxygen, was then blown directly through this superheated arc. At these extreme temperatures, a fraction of the atmospheric nitrogen (N₂) reacted with oxygen (O₂) to form nitric oxide (NO). This initial reaction was represented as: N₂ + O₂ → 2 NO. By meticulously controlling the energy supplied to the arc and the velocity of the air stream, the inventors managed to achieve yields of approximately 4–5% nitric oxide at 3000 °C, with lower yields at reduced temperatures. This step was extremely energy-intensive, requiring a constant and substantial power supply.
Step 2: Oxidation to Nitrogen Dioxide
The hot nitric oxide produced in the arc furnace was then carefully cooled. As it cooled and combined with more atmospheric oxygen, it underwent further oxidation to produce nitrogen dioxide (NO₂). This secondary reaction was crucial, as nitrogen dioxide is more reactive and suitable for the next stage of acid production. The speed of this conversion depended on the concentration of NO; for instance, at 1% NO, it would take about 180 seconds for 90% conversion, whereas at 6% NO, it would only take approximately 40 seconds. The chemical transformation here was: 2 NO + O₂ → 2 NO₂.
Step 3: Crafting Nitric Acid – The Absorption Challenge
The nitrogen dioxide gas was then dissolved in water to yield nitric acid (HNO₃). This seemingly simple step was, in fact, one of the most critical and complex aspects of the entire system, demanding sophisticated engineering for efficiency. The process involved a series of absorption towers, typically packed column or plate column designs, each standing an impressive four stories tall.
Within these towers, nitrogen dioxide was bubbled through water, often over non-reactive quartz fragments to maximise surface area for reaction. The reaction can be summarised as: 3 NO₂ + H₂O → 2 HNO₃ + NO. As the nitric acid reached a desired concentration, usually around 40–50%, it was transferred to granite storage containers. The liquid from the next water tower would then replace it, and this progressive movement continued down the line until the last water tower was replenished with fresh water.
Achieving high purity was a challenge. The nitric acid obtained through this method typically reached about 68% purity, as nitric oxide and water form an azeotropic mixture, making further purification by simple fractional distillation difficult. Furthermore, not all the oxides of nitrogen reacted. Approximately 20% remained unreacted, so the final towers in the series contained an alkaline solution of lime. This solution was used to convert the remaining oxides into calcium nitrate, also known as Norwegian saltpeter, which itself was a valuable fertiliser. Despite these efforts, a small percentage, roughly 2%, of the oxides of nitrogen were still released into the air.
Harnessing Norway's Natural Power: The Energy Demands
One of the defining characteristics of the Birkeland-Eyde process was its prodigious energy consumption. It required an astonishing 15 MWh (megawatt-hours) of electricity per ton of nitric acid produced, yielding approximately 60 grams per kWh. To put this into perspective, this was an incredibly inefficient process in terms of energy usage compared to later methods. This immense demand for electricity was a major limiting factor, but it also highlighted the ingenuity of its founders.
Fortuitously, Norway possessed abundant natural resources in the form of fast-flowing rivers, perfect for generating clean, inexpensive hydroelectric power. This synergy was no accident; the factories built for the Birkeland-Eyde process in Rjukan and Notodden were explicitly combined with the construction of large hydroelectric power facilities. This strategic placement allowed the process to operate economically, leveraging the cheap and plentiful electricity that was essential for its survival.
A Footprint in History: Rjukan and Notodden
The Norwegian towns of Rjukan and Notodden became industrial hubs for the Birkeland-Eyde process. These sites were chosen not only for their proximity to hydroelectric power but also for their logistical advantages. The factories, complete with their massive arc reactors and multi-story absorption towers, transformed the landscape and the local economy. For several decades, these facilities were at the forefront of the global fertiliser industry, a testament to Norwegian innovation and engineering prowess. One type of reactor used at Rjukan from 1912 to 1940 had a capacity of 3000 kW, showcasing the scale of these operations.

The March of Progress: Why the Birkeland-Eyde Process Faded
Despite its initial success and groundbreaking nature, the Birkeland-Eyde process was relatively short-lived as the dominant method for nitrogen fixation. Its inherent energy inefficiency proved to be its Achilles' heel. As scientific and industrial innovation continued, more efficient methods emerged, gradually rendering the Birkeland-Eyde process obsolete.
In the 1910s and 1920s, it was progressively replaced, primarily by a combination of the Haber process and the Ostwald process. The Haber process, developed by Fritz Haber and Carl Bosch, produces ammonia (NH₃) from molecular nitrogen (N₂) and hydrogen (H₂). This ammonia is then converted into nitric acid (HNO₃) via the Ostwald process. The Haber-Ostwald combination offered significantly greater energy efficiency and scalability, marking a new era in synthetic fertiliser production.
Birkeland-Eyde vs. Haber-Ostwald: A Comparative Look
To understand the shift in industrial preference, it's useful to compare the key attributes of these two revolutionary approaches to nitrogen fixation:
| Feature | Birkeland-Eyde Process | Haber-Ostwald Process |
|---|---|---|
| Primary Raw Materials | Air (N₂, O₂) and water | Air (N₂), natural gas/methane (for H₂), water |
| Main Intermediate Product | Nitric Oxide (NO) | Ammonia (NH₃) |
| Final Product (for fertiliser) | Nitric Acid (HNO₃) | Nitric Acid (HNO₃) from Ammonia |
| Energy Consumption | Extremely High (approx. 15 MWh/ton HNO₃) | Significantly Lower (more energy efficient) |
| Process Complexity | Multi-step, high-temperature arc | Multi-step, high-pressure synthesis of ammonia, then catalytic oxidation |
| Date of Dominance | Early 20th Century (c. 1903-1920s) | From 1910s onwards |
The Enduring Legacy
Despite its eventual replacement by the more efficient Haber-Ostwald process, the Birkeland-Eyde process holds a significant place in industrial history. It was a pioneering effort that demonstrated the viability of large-scale atmospheric nitrogen fixation. It directly addressed a global need, contributing immensely to agricultural output and preventing widespread famine in an era of rapid population growth. The process showcased remarkable ingenuity in engineering and chemistry, leveraging Norway's natural resources to create an entirely new industry. Its legacy lies not just in the fertiliser it produced, but in paving the way for future, even more efficient, industrial solutions to humanity's challenges.
Frequently Asked Questions (FAQs)
Who invented the Birkeland-Eyde process?
The Birkeland-Eyde process was developed by the Norwegian industrialist and scientist Kristian Birkeland, in collaboration with his business partner Sam Eyde, in 1903. Their work was built upon earlier experiments by individuals such as Henry Cavendish in 1784.
What was the primary purpose of the Birkeland-Eyde process?
The main purpose of the Birkeland-Eyde process was to fix atmospheric nitrogen to produce nitric acid. This nitric acid was then used as a key ingredient in the manufacture of synthetic fertilisers, which were crucial for enhancing agricultural productivity and feeding a growing global population.
Why was the Birkeland-Eyde process so energy-intensive?
The process was exceptionally energy-intensive because it relied on creating an extremely hot electric arc (over 3000 °C) to force atmospheric nitrogen and oxygen to react. This high-temperature environment required a continuous and substantial supply of electricity, consuming approximately 15 MWh per ton of nitric acid produced.
What replaced the Birkeland-Eyde process?
The Birkeland-Eyde process was gradually replaced in the 1910s and 1920s by a combination of the Haber process and the Ostwald process. The Haber process efficiently produces ammonia from nitrogen and hydrogen, and the Ostwald process then converts this ammonia into nitric acid, offering a far more energy-efficient solution.
Where were the Birkeland-Eyde factories located?
Factories based on the Birkeland-Eyde process were built in Rjukan and Notodden, Norway. These locations were strategically chosen due to the availability of large hydroelectric power facilities nearby, which were essential to meet the enormous electricity demands of the process.
What is 'Norwegian saltpeter'?
'Norwegian saltpeter' is another name for calcium nitrate. In the Birkeland-Eyde process, an alkaline solution of lime was used in the final absorption towers to convert unreacted nitrogen oxides into calcium nitrate, which was also utilised as a fertiliser.
Was the Birkeland-Eyde process environmentally friendly?
While it used clean hydroelectric power, the process itself released about 2% of unreacted nitrogen oxides into the air, which are atmospheric pollutants. Its high energy consumption, even if from renewable sources, made it less sustainable compared to later, more efficient processes. However, it was a significant step forward from relying solely on finite natural nitrate deposits.
If you want to read more articles similar to The Arc of Innovation: Birkeland-Eyde's Legacy, you can visit the Automotive category.
