10/04/2019
- The Rise of Polymersomes in Advanced Biomedical Applications
- Precision Engineering: Controlling Polymersome Size for Optimal Efficacy
- Tailoring the Membrane: Physicochemical Properties for Enhanced Performance
- Shaping the Future: Polymersome Morphology and Targeted Delivery
- Surface Functionalisation: The Art of Targeted Delivery
- Stimuli-Responsive Polymersomes: Smart Release on Demand
- Conclusion: Polymersomes as a Versatile Platform
The Rise of Polymersomes in Advanced Biomedical Applications
In the ever-evolving landscape of medical science, the quest for more effective and targeted drug delivery systems is paramount. Traditional methods, while foundational, often face limitations such as systemic toxicity, poor bioavailability, and inefficient targeting. Enter polymersomes – sophisticated, self-assembled nanostructures that are rapidly emerging as a powerful platform for a myriad of biomedical applications, with a particular focus on revolutionising cancer therapy. These engineered vesicles, crafted from amphiphilic copolymers, offer a remarkable degree of control over their structure, properties, and functionality, making them ideal candidates for carrying delicate therapeutic proteins, nucleic acids, and even imaging agents directly to diseased sites.
The concept of using self-assembling amphiphilic polymers to create vesicle structures, known as polymersomes, offers a versatile alternative to lipid-based carriers like liposomes. While liposomes have seen success, with commercial examples like Doxil® demonstrating preferential accumulation at disease sites and reduced off-target effects, they face challenges. A significant hurdle is their interaction with the immune system, often leading to rapid clearance from circulation due to opsonisation by serum proteins. To mitigate this, liposomes are frequently modified with poly(ethylene glycol) (PEG) to create a steric shield. Polymersomes, however, offer inherent advantages. Their design, composed of a hydrophobic bilayer membrane and a hydrophilic core, allows for the encapsulation of a broad spectrum of molecules. Crucially, the physicochemical properties of polymersomes, including size, shape, membrane rigidity, and responsiveness to stimuli, can be meticulously tuned by altering the hydrophilic-to-hydrophobic ratio of the constituent copolymers. This tunability not only enhances their colloidal and structural stability compared to liposomes but also opens up unprecedented possibilities for controlled release and targeted delivery, positioning polymersomes as a next-generation nanomedicine.
Precision Engineering: Controlling Polymersome Size for Optimal Efficacy
The journey of a nanocarrier within the human body is intricately linked to its size. For polymersomes, this is a critical design parameter that dictates their biodistribution, cellular uptake, and therapeutic efficacy. The human microvasculature, particularly within healthy organs, has narrow, non-fenestrated capillaries, effectively filtering out particles larger than approximately 12 nm. In stark contrast, the tumour microvasculature is notoriously leaky, characterised by larger gaps between endothelial cells (380-780 nm) and impaired lymphatic drainage. This physiological difference presents a prime opportunity for nanocarriers. Polymersomes designed to be smaller than these tumour vasculature gaps can exploit the enhanced permeability and retention (EPR) effect, leading to their passive accumulation within the tumour site.
However, size control is not solely about tumour targeting. For effective intracellular delivery, where many drug targets reside within subcellular compartments, polymersomes need to be efficiently internalised by cells. Endocytosis, a process where cells engulf particles, is significantly influenced by nanoparticle size. Studies suggest that spherical nanoparticles with diameters around 50-60 nm are optimal for facilitating cellular uptake. The self-assembly process of amphiphilic polymers, driven by factors like polymer concentration and mixing rates, fundamentally dictates the resulting polymersome size. Furthermore, techniques such as:
- Filtration: Cross-flow filtration and differential centrifugation can separate polymersomes into size-defined populations, allowing for the isolation of specific size ranges (e.g., 22 nm to 240 nm). This has been crucial in studies correlating polymersome size with cellular uptake, demonstrating how size influences interaction with specific cellular receptors.
- Extrusion/Sonication: Applying shear forces through extrusion or acoustic energy via sonication can effectively reduce polymersome diameters to below 100 nm, yielding particles with uniform size distribution. The effectiveness of sonication is, however, dependent on the membrane's water content and rigidity.
- Polymer Chemistry: The inherent chemical structure of the amphiphilic copolymers, including block length and the volume fraction of hydrophilic blocks, directly influences the final polymersome diameter. Longer hydrophobic blocks or a higher proportion of hydrophilic segments generally lead to larger vesicles.
The precise control over polymersome size is not merely an academic exercise; it translates directly into enhanced therapeutic outcomes. For instance, smaller polymersomes (around 90 nm) have shown longer circulation times compared to larger ones (120-250 nm), which are cleared more rapidly from the bloodstream. This extended circulation time is vital for allowing the nanocarriers to reach their intended targets.
Tailoring the Membrane: Physicochemical Properties for Enhanced Performance
Beyond size, the physicochemical properties of the polymersome membrane play a pivotal role in their interaction with biological systems. These properties, including surface charge and permeability, govern cellular uptake, endosomal escape, and the spatial orientation of encapsulated cargo. By engineering these characteristics, researchers aim to maximise both diagnostic and therapeutic efficacy.
The surface charge of polymersomes significantly influences their interaction with the negatively charged surface of mammalian cell membranes (typically around -20 mV zeta potential). Asymmetrical charge distributions within the polymersome membrane have been shown to improve cellular endocytosis and subsequent endosomal escape. This asymmetry can be achieved by using triblock copolymers with distinct hydrophilic blocks. For example, polymersomes with asymmetrical charge distributions have demonstrated faster cellular uptake compared to their symmetrical counterparts. In some cases, highly positively charged polymersomes can be rapidly internalised by cells due to strong electrostatic interactions with cell surface proteoglycans. However, this rapid uptake can sometimes lead to unintended cellular toxicity.
Conversely, increasing the proportion of hydrophilic blocks, such as PEG, on the polymersome surface can create steric repulsion, slowing down cellular uptake but also reducing toxicity. This ability to modulate surface charge allows for a delicate balance between efficient delivery and minimal adverse effects. Furthermore, mimicking the negative surface charge of red blood cells can alter biodistribution patterns, with negatively charged polymersomes showing different accumulation profiles in organs compared to neutral ones. The potential for positively charged polymersomes to act as antimicrobial agents, by disrupting bacterial membranes, also highlights the versatility of surface charge manipulation.
Permeability: The Gatekeeper of Cargo Release
The permeability of the polymersome membrane is a critical factor controlling the rate at which molecules can traverse it. This permeability is primarily influenced by the nature of the polymer forming the hydrophobic block, which dictates membrane fluidity and thickness. The glass transition temperature (Tg) of the polymer is a key indicator of membrane fluidity; membranes above their Tg are more fluid and allow faster molecular movement. Polymers with more hydrophilic membrane compositions, such as those based on poly(butylene oxide), have shown higher permeability than those with aliphatic lipid membranes. Conversely, polymers with higher molecular weights or increased membrane thickness, as observed with poly(ethylene glycol)-block-poly(1,2-butadiene) systems, tend to reduce molecular movement across the membrane.
Asymmetry in membrane permeability can also be engineered to achieve directional solute transport. By mixing polymersomes with different permeability characteristics, researchers have created structures with specialised "buds" that facilitate controlled release. This has been demonstrated in systems designed for propulsion, where the controlled release of reaction products from a permeable bud drives the polymersome forward, enhancing its transport across biological barriers like the blood-brain barrier.
The solubility and diffusivity of the cargo molecule within the hydrophobic membrane also play a role. Charged molecules, for instance, experience a larger energy barrier to dissolve in the hydrophobic membrane, slowing their transport compared to neutral molecules of similar size. The size of the diffusing molecule itself can also impact the rate of movement, with larger molecules potentially inducing conformational changes in the polymer chains to facilitate their passage.
Furthermore, the permeability of polymersomes can be modulated in situ by incorporating ionisable functional groups into the hydrophobic block. For example, polymersomes containing piperidine groups can become more permeable in acidic environments, such as the tumour microenvironment, leading to triggered drug release. This pH-sensitivity is a powerful tool for localised drug delivery, minimising systemic exposure.
Structural Influence on Imaging Contrast
The architecture of polymersomes can also significantly impact their performance in medical imaging. For Magnetic Resonance Imaging (MRI), contrast agents like gadolinium are used to enhance image clarity. However, when encapsulated within traditional liposomes, the slow diffusion of water across the lipid bilayer can limit the relaxivity of gadolinium. Polymersomes with porous membranes have been developed to overcome this limitation, increasing water flux and thereby enhancing the relaxivity of encapsulated contrast agents. These porous structures, achieved through methods like cross-linking followed by surfactant extraction, can lead to a dramatic amplification of MRI signal intensity compared to non-porous counterparts.
The spatial arrangement of contrast agents within the polymersome is also crucial. Immobilising chelated gadolinium on the polymersome surface, for instance, can reduce the distance to surrounding water molecules, improving relaxivity. Similarly, the arrangement of superparamagnetic iron oxide nanoparticles (SPIONs) within polymersomes can influence their relaxivity. Radial arrangements at the core-shell interface have shown higher relaxivity rates than homogenous incorporation, with specific arrangements of larger SPIONs leading to decreased polymersome diameter and significantly enhanced relaxivity.
Fluorescence Imaging Applications
In fluorescence imaging, polymersomes encapsulating near-infrared (NIR) fluorophores are used for tracking cell migration and biodistribution. The efficiency of these systems depends on factors like fluorophore structure, their average distance within the membrane, and the bilayer thickness. Polymersomes with thicker hydrophobic bilayers can lead to higher excited-state deactivation rates, affecting fluorescence. Optimising the number of fluorophores and their concentration within the polymersome is key to achieving high signal sensitivity and photostability, enabling deep-tissue imaging and long-term tracking of cells.
Shaping the Future: Polymersome Morphology and Targeted Delivery
The shape of polymersomes is not merely an aesthetic feature; it profoundly influences their transport and interaction with biological systems. While spherical polymersomes are the most common due to their thermodynamic stability, non-spherical morphologies like ellipsoids, tubules, and stomatocytes offer unique advantages.
Ellipsoidal and Tubular Polymersomes: Enhanced Adhesion
Ellipsoidal and tubular polymersomes, with their increased surface area compared to spheres, offer enhanced binding efficacy to target cells. This larger surface area allows for a greater density of surface-bound targeting ligands, such as peptides or antibodies, leading to stronger cellular adhesion. Ellipsoidal polymersomes, for example, have shown improved diffusivity in blood-mimicking solutions and superior adhesion to cells overexpressing specific integrins under flow conditions. Tubular polymersomes can be formed through methods like film rehydration or chemical modification of spherical precursors, and their increased surface area can present more targeting moieties, boosting adhesion affinity.
However, the increased surface area of non-spherical shapes can also impact cellular uptake kinetics. While offering greater adhesion, tubular polymersomes have sometimes exhibited slower internalisation compared to spherical counterparts, potentially due to higher energy requirements for endocytosis. This suggests that the optimal shape is application-dependent, balancing adhesion and uptake efficiency.
Stomatocytes: Tailoring for Specific Applications
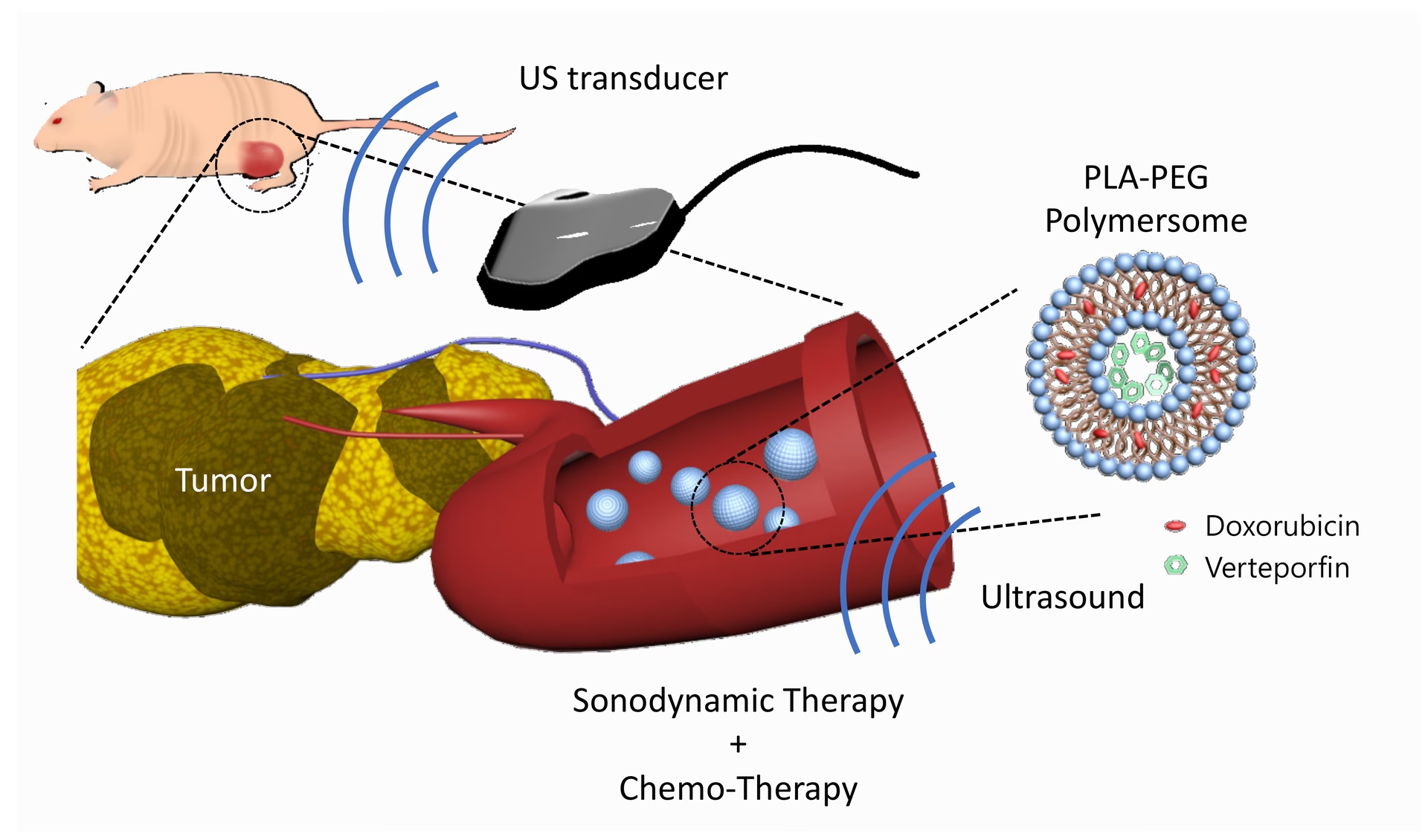
Stomatocytes, or bowl-shaped polymersomes, can be transformed from spherical precursors through controlled dialysis against specific solvent mixtures. This shape change can alter membrane permeability and internal volume, making them suitable for specific applications. For instance, stomatocytes have been used as nanocontainers for platinum nanoparticles, enabling autonomous movement through the decomposition of hydrogen peroxide. Biodegradable stomatocytes have also been developed, offering controlled release of drugs like doxorubicin in response to acidic environments.
Surface Functionalisation: The Art of Targeted Delivery
To ensure polymersomes reach their intended targets and exert their therapeutic effects, their surfaces can be functionalised with specific homing ligands. These ligands, which can include small molecule metabolites, peptides, aptamers, or antibodies, bind to receptors that are overexpressed on diseased cells or tissues, facilitating targeted delivery.
Small Molecule Metabolites (e.g., Folic Acid)
Folate receptors are significantly upregulated in many types of cancer. Conjugating folic acid to polymersomes enhances their accumulation in folate receptor-positive cancer cells through receptor-mediated endocytosis. This targeted approach has demonstrated a significant increase in cellular uptake and a reduction in tumour volume in preclinical models, making folic acid-functionalised polymersomes a promising strategy for cancer chemotherapy.
Peptide Conjugation
Peptides can be designed to target specific receptors, including those on the blood-brain barrier (BBB). Peptides like Angiopep-2, which targets the low-density lipoprotein receptor-related protein 1 (LRP1), have been conjugated to polymersomes to significantly enhance their brain penetration. Similarly, peptides targeting integrins have shown improved delivery to lung cancer cells. The Tat peptide, known for its cell-penetrating ability, has also been used to enhance the uptake of polymersomes by dendritic cells, crucial for cancer immunotherapy.
Aptamer and Antibody Functionalisation
Aptamers, short single-stranded DNA or RNA molecules, can be engineered to bind specific targets with high affinity. EpCAM RNA aptamers, for instance, have been used to target lung cancer cells, improving the delivery of encapsulated drugs. Antibodies, such as Trastuzumab, which targets the HER2 receptor overexpressed in breast cancer, can also be conjugated to polymersomes. These antibody-conjugated polymersomes demonstrate specific targeting of HER2-positive cancer cells and have shown promise in both imaging and therapeutic applications. Similarly, anti-ICAM-1 antibodies can target inflammatory sites.
Stimuli-Responsive Polymersomes: Smart Release on Demand
One of the key advantages of polymersomes is their potential to be engineered as stimuli-responsive systems. This means they can undergo structural changes or release their cargo in response to specific internal or external triggers, such as pH, redox potential, enzymes, ultrasound, or magnetic fields. This "smart" release mechanism significantly improves therapeutic precision and reduces systemic toxicity.
Internal Stimuli Responsiveness
- pH-Responsive Polymersomes: Tumour microenvironments are often characterised by lower pH (6.5-6.9) compared to normal tissues (7.4). Polymersomes incorporating pH-sensitive linkages or ionisable groups can exploit this difference. In acidic conditions, these polymersomes can destabilise, leading to the release of encapsulated drugs. This pH-triggered release is also effective within the acidic environment of endosomes and lysosomes (pH 4.5-6.0) after cellular uptake, facilitating intracellular drug delivery.
- Redox-Responsive Polymersomes: The concentration of glutathione, a biological reducing agent, is significantly higher in tumour cells and intracellular compartments like the cytosol. Polymersomes incorporating disulfide bonds, which are cleaved by glutathione, can therefore release their cargo specifically within these reducing environments. This has been demonstrated to enhance the nuclear delivery of drugs like doxorubicin.
- Enzyme-Responsive Polymersomes: Certain enzymes, such as hyaluronidase, are overexpressed in specific tissues or by pathogens. Polymersomes engineered with enzyme-cleavable linkages can be designed to release their cargo only in the presence of these specific enzymes. This strategy has been used to target bacteria or to facilitate drug release within lysosomes.
External Stimuli Control
- Focused Ultrasound: Focused ultrasound can be used to trigger the collapse of polymer bubbles or disrupt polymersome structures, leading to the release of encapsulated drugs. This technique has been employed to enhance the penetration of therapeutics into deep-seated tumours, particularly in hypoxic regions, by using monocytes as carriers for ultrasound-sensitive polymersomes.
- Magnetic Field: Magnetic fields can induce local heating via superparamagnetic iron oxide nanoparticles (SPIONs) embedded within polymersomes. This localised heating can trigger drug release or the collapse of polymer bubbles, offering another non-invasive method for controlled delivery.
Conclusion: Polymersomes as a Versatile Platform
Polymersomes represent a significant advancement in nanomedicine. Their inherent tunability in size, shape, membrane properties, and responsiveness to stimuli makes them an exceptionally versatile platform for drug delivery, imaging, and diagnostics. The ability to precisely engineer these nanocarriers allows for targeted delivery, enhanced cellular uptake, controlled release, and reduced systemic toxicity, particularly in challenging diseases like cancer. As research continues to refine their design and application, polymersomes are poised to play an increasingly vital role in the future of precision medicine, offering novel solutions for unmet clinical needs.
Frequently Asked Questions (FAQs)
1. What are polymersomes and how are they different from liposomes?
Polymersomes are self-assembled vesicular structures formed from amphiphilic block copolymers. Unlike liposomes, which are made from lipids, polymersomes offer greater structural and colloidal stability, and their properties (size, shape, permeability, etc.) can be more readily tuned by modifying the polymer chemistry.
2. How do polymersomes help in cancer therapy?
Polymersomes can be engineered to target cancer cells specifically, deliver potent anti-cancer drugs directly to tumours, and release these drugs in a controlled manner, thereby minimising side effects on healthy tissues. Their ability to exploit the EPR effect for passive tumour targeting and to be functionalised with targeting ligands (like folic acid or antibodies) for active targeting makes them highly valuable in cancer treatment.
3. Can polymersomes deliver genetic material like mRNA or DNA?
Yes, polymersomes can be designed to encapsulate and deliver nucleic acids, such as mRNA for vaccines or gene therapy, and DNA for gene delivery. Their protective structure shields the genetic material from degradation, and their surface can be modified to facilitate cellular uptake and endosomal escape, which are critical steps for effective gene delivery.
4. What does "stimuli-responsive" mean in the context of polymersomes?
Stimuli-responsive polymersomes are designed to change their structure or release their encapsulated cargo in response to specific internal or external triggers. These triggers can include changes in pH (e.g., in tumour microenvironments or endosomes), the presence of specific enzymes, or external stimuli like focused ultrasound or magnetic fields. This allows for highly localised and controlled drug release.
5. What are the main advantages of using polymersomes in drug delivery?
The key advantages include:
- High stability: Greater resistance to degradation and aggregation compared to liposomes.
- Tunable properties: Control over size, shape, membrane thickness, and permeability.
- Versatile cargo loading: Ability to encapsulate both hydrophilic (in the core) and hydrophobic (in the membrane) molecules, as well as larger biomolecules like proteins and nucleic acids.
- Targeted delivery: Can be functionalised with ligands to specifically target diseased cells or tissues.
- Stimuli-responsiveness: Enable controlled and localised drug release.
If you want to read more articles similar to Polymersomes: The Future of Drug Delivery?, you can visit the Automotive category.