17/03/2008
When we delve into the intricate world of chemical bonding, simpler models like Lewis structures and Valence Bond Theory often provide excellent starting points. They help us visualise how atoms connect and share electrons to form molecules. However, these models sometimes fall short, struggling to explain certain phenomena or the behaviour of more complex molecules. For instance, the familiar oxygen molecule, O₂, poses a significant challenge: while its Lewis structure suggests all electrons are paired, experimental evidence reveals it is attracted to magnetic fields, a property known as paramagnetism. This is where Molecular Orbital (MO) Theory emerges as a more sophisticated and powerful framework, offering a deeper, quantum mechanical understanding of how atoms bond and electrons behave within a molecule.
MO Theory provides a global, delocalised perspective on chemical bonding. Unlike Valence Bond Theory, which often localises electrons between specific atoms, MO Theory posits that electrons in a molecule reside in molecular orbitals that are spread out over the entire molecule. Imagine electrons not confined to a single atom or a bond between two atoms, but rather free to move under the influence of all the nuclei in the molecule. This approach allows for a more accurate description of electron distribution and energy levels, aligning more closely with experimental observations, particularly for molecules that exhibit unusual magnetic properties or extensive electron delocalisation.
- The Core Principle: Linear Combination of Atomic Orbitals (LCAO)
- Understanding Bond Order: A Measure of Stability
- Magnetism Explained: The Oxygen Anomaly Revisited
- Beyond Simple Bonds: Delocalisation and Extended Systems
- MO Theory vs. Valence Bond Theory: A Brief Comparison
- Frequently Asked Questions About MO Theory
The Core Principle: Linear Combination of Atomic Orbitals (LCAO)
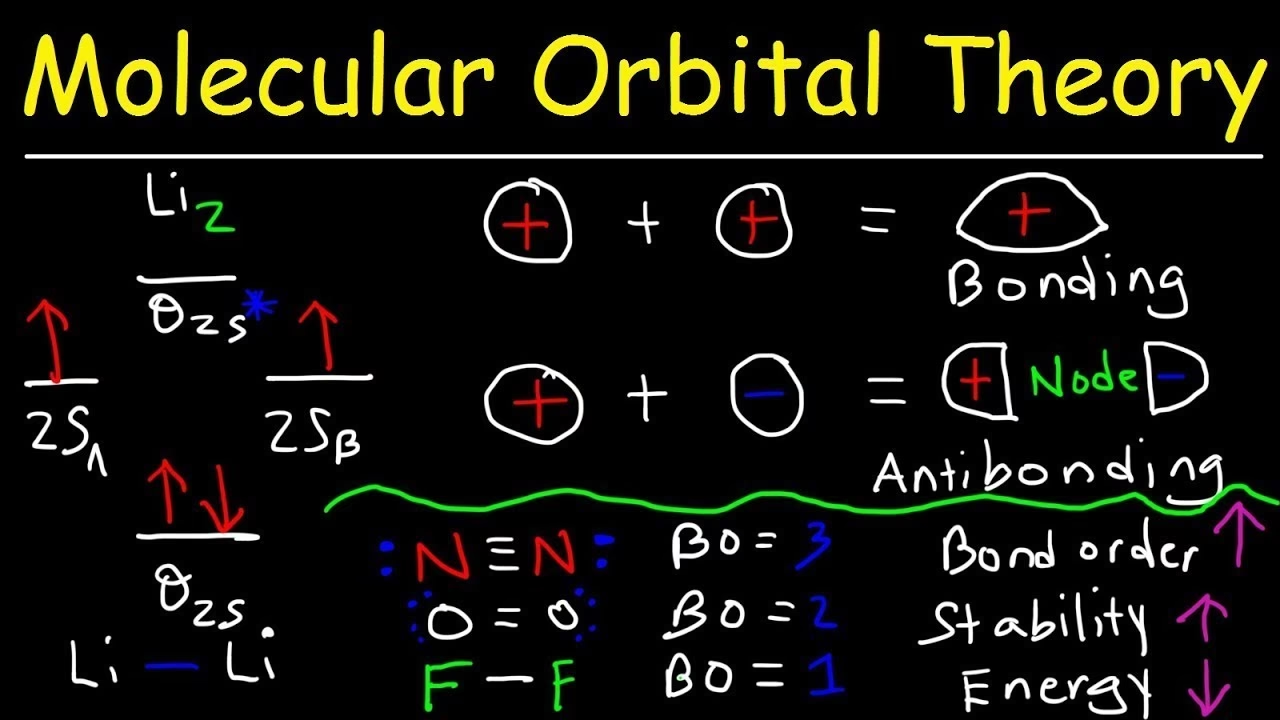
At the heart of MO Theory is the Linear Combination of Atomic Orbitals (LCAO) method. This fundamental principle states that when atoms come together to form a molecule, their atomic orbitals (AOs) combine or overlap to create new molecular orbitals (MOs). This combination isn't just a simple merging; it's a mathematical process based on quantum mechanics, where atomic wave functions are added or subtracted. The resulting molecular orbitals describe the probable location and energy of electrons within the entire molecule.
When two atomic orbitals combine, they typically form two molecular orbitals: one bonding orbital and one anti-bonding orbital. A bonding molecular orbital is formed when atomic orbitals combine constructively, leading to an increased electron density between the nuclei. This enhanced electron density acts as a 'glue', pulling the nuclei together and stabilising the molecule. Conversely, an anti-bonding molecular orbital is formed when atomic orbitals combine destructively, resulting in a region of zero electron density (a node) between the nuclei. Electrons in anti-bonding orbitals actually push the nuclei apart, destabilising the molecule. Just like atomic orbitals, each molecular orbital can hold a maximum of two electrons with opposite spins, following the Pauli Exclusion Principle. Electrons fill these molecular orbitals starting from the lowest energy levels, adhering to the Aufbau principle and Hund's rule.
Understanding Bond Order: A Measure of Stability
One of the crucial quantitative outputs of MO Theory is the bond order. This concept provides a direct measure of the number of chemical bonds between a pair of atoms and is a strong indicator of molecular stability. A higher bond order generally correlates with a stronger and more stable bond. The beauty of MO Theory lies in its ability to calculate this value directly from the distribution of electrons in bonding and anti-bonding molecular orbitals.
The formula for calculating bond order is straightforward:
Bond order = 1/2 (Number of electrons in bonding MOs - Number of electrons in anti-bonding MOs)
It's important to note that only valence electrons typically contribute significantly to the bond order. Inner-shell electrons, while present, often occupy molecular orbitals derived from 1s atomic orbitals where the number of bonding and anti-bonding electrons cancels out, resulting in no net effect on the bond order.
Let's consider the hypothetical He₂ molecule, a classic example that beautifully illustrates the power of bond order. Each helium atom has two electrons in its 1s atomic orbital. When two helium atoms approach, their 1s orbitals combine to form one bonding (σ1s) and one anti-bonding (σ*1s) molecular orbital. The total of four valence electrons from the two helium atoms fill these orbitals: two electrons go into the bonding σ1s orbital, and the remaining two go into the anti-bonding σ*1s orbital. Applying the bond order formula:
Bond order (He₂) = 1/2 (2 - 2) = 0
A bond order of zero signifies that no stable chemical bond will form between the two helium atoms, which is consistent with experimental observations. Helium exists as a monatomic gas. While a transient helium dimer can be detected under extreme conditions, it's a weak van der Waals molecule, not a true covalent bond, reinforcing the MO Theory prediction.
Consider another comparison to see how bond order relates to bond strength:
| Molecule | Bonding Electrons | Anti-bonding Electrons | Bond Order | Bond Energy (kJ/mol) |
|---|---|---|---|---|
| H₂ | 2 | 0 | 1/2 (2 - 0) = 1 | 436 |
| H₂⁺ | 1 | 0 | 1/2 (1 - 0) = 1/2 | 171 |
As the table clearly shows, H₂ has a bond order of 1, indicating a single covalent bond, and a high bond energy. In contrast, H₂⁺, with only one electron in the bonding orbital, has a bond order of 0.5. This lower bond order directly correlates with its significantly lower bond energy, making it less stable than H₂, as observed experimentally. This quantitative prediction of stability based on electron distribution is a key strength of MO Theory.
Magnetism Explained: The Oxygen Anomaly Revisited
Perhaps one of the most compelling triumphs of Molecular Orbital Theory is its ability to explain the magnetic behaviour of the oxygen molecule, O₂. As mentioned earlier, Lewis structures depict O₂ with a double bond and all electrons paired, implying it should be diamagnetic (weakly repelled by a magnetic field). However, liquid oxygen is famously attracted to magnetic fields, a phenomenon known as paramagnetism, which arises from the presence of unpaired electrons.
MO Theory resolves this apparent contradiction beautifully. Let's look at the molecular orbital diagram for O₂. Oxygen has an atomic number of 8, with an electronic configuration of 1s²2s²2p⁴. When two oxygen atoms combine, their valence atomic orbitals (2s and 2p) form molecular orbitals. The filling order of these MOs for O₂ is crucial:
σ1s² < σ*1s² < σ2s² < σ*2s² < σ2pz² < [π2px² = π2py²] < [π*2px¹ = π*2py¹] < σ*2pz
Counting the electrons in bonding and anti-bonding orbitals for O₂ (considering only valence electrons for bond order and magnetic properties):
- Bonding electrons: (σ2s²) + (σ2pz²) + (π2px²) + (π2py²) = 2 + 2 + 2 + 2 = 8
- Anti-bonding electrons: (σ*2s²) + (π*2px¹) + (π*2py¹) = 2 + 1 + 1 = 4
Wait, the provided text shows 10 bonding and 6 anti-bonding electrons for O2. Let's re-evaluate based on the provided configuration: Electronic configuration of oxygen molecule; ó1s² < *ó1s² < ó2s² < *ó2s², [ π2px² = π2py²] < ó 2pz² < [*π2px¹ =*π2py¹] < *ó2pz This implies: Bonding: 2 (σ1s) + 2 (σ2s) + 2 (π2px) + 2 (π2py) + 2 (σ2pz) = 10 Anti-bonding: 2 (*σ1s) + 2 (*σ2s) + 1 (*π2px) + 1 (*π2py) = 6 Bond order = (10 - 6) / 2 = 2. This is correct for O₂.
Now, let's look at the highest occupied molecular orbitals (HOMOs). According to the MO diagram, the two highest energy orbitals are the degenerate π*2px and π*2py anti-bonding orbitals. Each of these orbitals receives one electron, according to Hund's Rule, before pairing occurs. This means O₂ has two unpaired electrons in its anti-bonding π* orbitals. The presence of these two unpaired electrons directly accounts for the observed paramagnetism of oxygen, a phenomenon that Lewis structures simply cannot predict. This remarkable agreement between theoretical prediction and experimental observation highlights the superior explanatory power of quantum mechanics in MO Theory.
Beyond Simple Bonds: Delocalisation and Extended Systems
MO Theory truly shines when describing molecules with extensive electron delocalisation, where electrons are not confined to a single bond or atom but are spread over multiple atoms. This is particularly evident in aromatic compounds and conjugated systems.
Consider benzene (C₆H₆), a classic example of an aromatic molecule. In Valence Bond Theory, benzene is often described as a resonance hybrid of two Kekulé structures, with alternating single and double bonds. MO Theory offers a more elegant and direct explanation. It describes the 24 sigma (σ) bonding electrons as being localised between C-C and C-H atoms, similar to Valence Bond Theory. However, the remaining six pi (π) bonding electrons are described as occupying three delocalised π molecular orbitals that extend over the entire hexagonal carbon ring, both above and below the plane of the ring. Two of these electrons occupy an MO with equal contributions from all six carbon atoms, while the other four occupy orbitals with vertical nodes. This delocalisation explains why all carbon-carbon bonds in benzene are chemically equivalent and have a bond order of approximately 1.5, intermediate between a single and a double bond. This unified description naturally accounts for benzene's extraordinary stability and unique chemical properties.
The concept of delocalisation also extends to much larger systems, such as beta-carotene, chlorophyll, or heme. In these molecules, electrons in π orbitals are spread out over long distances, forming extensive conjugated systems. This delocalisation leads to light absorption at lower energies, often in the visible spectrum, which is why these substances exhibit characteristic colours. MO Theory provides a robust framework for interpreting ultraviolet-visible (UV-Vis) spectroscopy data, where changes in electronic structure are observed through the absorption of light at specific wavelengths. The transitions of electrons from lower-energy MOs to higher-energy MOs are directly related to the absorbed light, making MO Theory indispensable for understanding molecular electronic states.
Furthermore, MO Theory offers insights into electrical phenomena, such as the high electrical conductivity of graphite. Graphite consists of layers of hexagonal carbon sheets. MO Theory explains that some electrons in these sheets are completely delocalised over arbitrary distances, residing in very large molecular orbitals that span an entire graphite sheet. This continuous band overlap of half-filled p orbitals allows these electrons to move freely, akin to electrons in a metal, thereby explaining graphite's excellent electrical conductivity along its planar direction. This fundamental understanding of electron mobility is crucial for materials science, differentiating conductors, semiconductors, and insulators.
MO Theory vs. Valence Bond Theory: A Brief Comparison
While both Molecular Orbital Theory and Valence Bond Theory aim to describe chemical bonding, they approach it from different perspectives and excel in different areas.
| Feature | Molecular Orbital Theory | Valence Bond Theory |
|---|---|---|
| Electron Localisation | Electrons are delocalised over the entire molecule. | Electrons are localised between specific atoms or as lone pairs. |
| Orbital Formation | Atomic orbitals combine to form molecular orbitals before electrons are added. | Atomic orbitals hybridise on individual atoms, then overlap to form bonds. |
| Magnetic Properties | Successfully explains paramagnetism (e.g., O₂) due to unpaired electrons in MOs. | Often struggles with paramagnetism if Lewis structures show all paired electrons. |
| Resonance | Naturally accounts for delocalisation in resonant structures (e.g., benzene) through delocalised MOs. | Requires the concept of resonance structures to describe delocalisation. |
| Spectroscopy | Provides a direct framework for understanding electronic transitions and absorption spectra. | Less direct in explaining spectroscopic data related to electron energies. |
| Complex Molecules | More suitable for describing extended systems, conjugated molecules, and electron-deficient compounds. | More intuitive for simple, localised bonds and molecular geometry. |
In essence, MO Theory provides a more comprehensive and quantum mechanically rigorous description of electron behaviour in molecules, especially when dealing with phenomena like magnetism, extensive delocalisation, and spectroscopic properties. It treats the molecule as a single, unified entity where electrons are not bound to specific atoms but rather to the molecule as a whole.
Frequently Asked Questions About MO Theory
- What is the main difference between bonding and anti-bonding orbitals?
- Bonding orbitals have increased electron density between nuclei, stabilising the molecule. Anti-bonding orbitals have a node (zero electron density) between nuclei, destabilising the molecule.
- Why is MO Theory considered more accurate than Lewis structures?
- MO Theory is based on quantum mechanics and allows for electron delocalisation over the entire molecule, accurately predicting properties like magnetism (e.g., oxygen's paramagnetism) and spectroscopic behaviour that Lewis structures cannot explain.
- Can MO Theory predict molecular geometry?
- While MO Theory primarily focuses on electron distribution and energy levels, the shapes and symmetries of molecular orbitals can indirectly influence molecular geometry, often in agreement with VSEPR theory, but it's not its primary strength.
- Does MO Theory replace Valence Bond Theory?
- No, they are complementary. Valence Bond Theory is often simpler and more intuitive for understanding basic bonding and molecular shapes, while MO Theory provides a deeper, more accurate picture, especially for complex systems and specific phenomena like magnetism or spectroscopy.
- What is a degenerate molecular orbital?
- Degenerate molecular orbitals are orbitals that have the same energy level. For example, the π2px and π2py orbitals in diatomic molecules are often degenerate.
In conclusion, Molecular Orbital Theory is a cornerstone of modern chemistry, providing an indispensable framework for understanding the fundamental nature of chemical bonds. By treating electrons as delocalised waves occupying orbitals that span the entire molecule, it successfully explains phenomena that simpler models cannot, such as the paramagnetism of oxygen, the stability of benzene, and the electrical conductivity of graphite. Its ability to quantify bond strength, predict molecular stability, and interpret spectroscopic data makes it an invaluable tool for chemists, allowing for a far more complete and accurate picture of the molecular world.
If you want to read more articles similar to Unravelling Molecular Orbital Theory, you can visit the Automotive category.
