21/08/2018
CAR T-cell therapy represents a significant advancement in the fight against certain cancers, particularly blood cancers like leukaemia and lymphoma. This innovative treatment harnesses the power of a patient's own immune system, genetically engineering T-cells to specifically target and destroy cancer cells. While the prospect of such a targeted approach is exciting, many patients and their families understandably want to know: how long does CAR T-cell therapy take? Understanding the timeline involved is crucial for planning, managing expectations, and preparing for the journey ahead. This article aims to demystify the process, breaking down the various stages and providing a realistic overview of the duration.
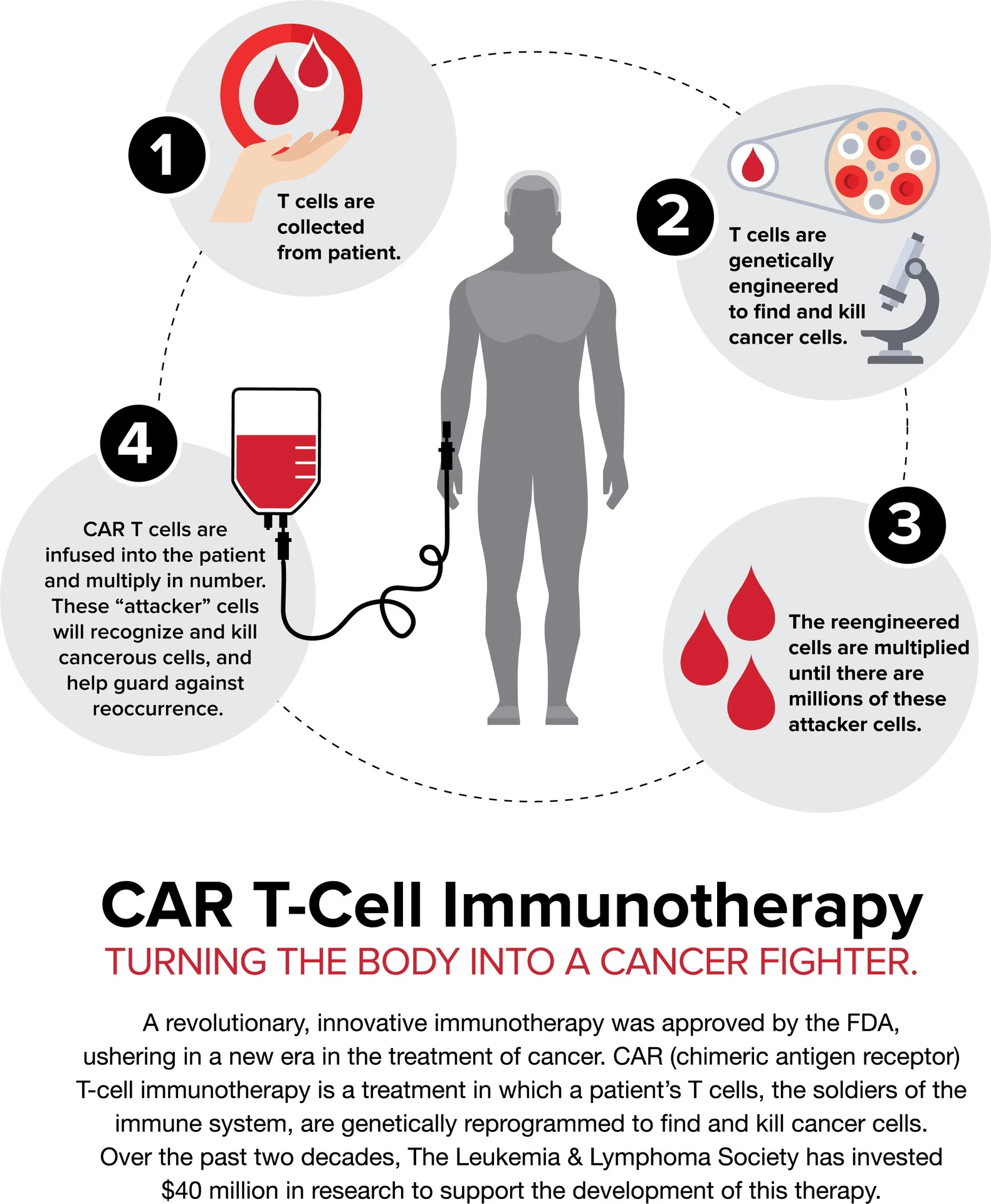
- The CAR T-Cell Therapy Journey: A Phased Approach
- Phase 1: Initial Consultation and Preparation (Typically 1-4 Weeks)
- Phase 2: Apheresis – Collecting Your T-Cells (1-3 Hours, Plus Processing Time)
- Phase 3: Manufacturing of CAR T-Cells (Typically 2-6 Weeks)
- Phase 4: Conditioning Chemotherapy (1-7 Days)
- Phase 5: CAR T-Cell Infusion (1-2 Hours)
- Phase 6: Post-Infusion Monitoring and Management (Weeks to Months)
- Factors Influencing the Total Timeline
- Summary of Typical Timelines
- Frequently Asked Questions about CAR T-Cell Therapy Timelines
- Q1: Can the manufacturing time for CAR T-cells be shortened?
- Q2: What happens if my cancer progresses while waiting for CAR T-cells?
- Q3: How long do I need to stay in the hospital after the CAR T-cell infusion?
- Q4: What is the total time commitment for CAR T-cell therapy?
- Q5: Is CAR T-cell therapy a one-time treatment?
- Conclusion
The CAR T-Cell Therapy Journey: A Phased Approach
The CAR T-cell therapy process isn't a single event; it's a multi-stage journey that requires careful coordination between the patient, their medical team, and the specialised manufacturing facility. The overall timeline can vary depending on individual circumstances, the specific type of CAR T-cell therapy being administered, and the patient's response. However, we can broadly divide the process into several key phases:
Phase 1: Initial Consultation and Preparation (Typically 1-4 Weeks)
Before any treatment begins, a thorough evaluation is essential. This phase involves a series of appointments and tests to determine if CAR T-cell therapy is the right option for the patient. This includes:
- Medical History Review: Doctors will meticulously review your medical history, previous treatments, and overall health status.
- Diagnostic Tests: This may involve blood tests, imaging scans (like CT or PET scans), and bone marrow biopsies to confirm the diagnosis, assess the extent of the disease, and identify specific markers on the cancer cells that the CAR T-cells will target.
- Eligibility Assessment: CAR T-cell therapy has specific eligibility criteria, and doctors will ensure the patient meets these requirements. Factors like organ function and the presence of certain co-existing conditions can influence eligibility.
- Patient Education: A crucial part of this phase is educating the patient and their family about the treatment, its potential benefits, risks, and side effects. This allows for informed decision-making.
- Treatment Planning: Once eligibility is confirmed, a personalised treatment plan is developed, including the logistics of apheresis and the subsequent infusion.
The duration of this initial phase can range from a few days to several weeks, depending on the complexity of the patient's case and the availability of appointments and testing slots.
Phase 2: Apheresis – Collecting Your T-Cells (1-3 Hours, Plus Processing Time)
This is a critical step where the patient's own T-cells are collected. Apheresis is a medical procedure similar to dialysis. Blood is drawn from the patient's body, passed through a machine that separates and collects the white blood cells (including T-cells), and then the remaining blood is returned to the patient. This process typically takes 1 to 3 hours to complete.
Following apheresis, the collected T-cells are sent to a specialised manufacturing facility. Here, they undergo a complex process of genetic modification to become CAR T-cells. This involves introducing a gene that allows the T-cells to recognise and bind to a specific antigen (a protein) on the surface of the cancer cells.
Phase 3: Manufacturing of CAR T-Cells (Typically 2-6 Weeks)
This is the most time-consuming part of the process. Once the T-cells are collected and sent to the lab, they are cultured and genetically engineered. This involves several steps:
- Cell Isolation and Expansion: The collected T-cells are isolated and grown in a laboratory setting, significantly increasing their numbers.
- Genetic Engineering: The T-cells are genetically modified to express the Chimeric Antigen Receptor (CAR) on their surface. This CAR is designed to bind to the specific antigen present on the patient's cancer cells.
- Quality Control: Throughout the manufacturing process, rigorous quality control measures are in place to ensure the safety, potency, and purity of the CAR T-cells. This includes testing for viability, sterility, and correct CAR expression.
The manufacturing process can take anywhere from 2 to 6 weeks, and sometimes longer, depending on the specific CAR T-cell product and the manufacturing facility's capacity. During this time, the patient may undergo further medical assessments or receive bridging therapy if their disease progresses rapidly.
Phase 4: Conditioning Chemotherapy (1-7 Days)
Before the CAR T-cells are infused back into the patient, a course of conditioning chemotherapy is typically administered. This chemotherapy is designed to:
- Deplete Existing Lymphocytes: It helps to clear out the patient's existing T-cells and other lymphocytes, creating space for the newly engineered CAR T-cells to engraft and proliferate effectively.
- Reduce the Risk of Graft-Versus-Host Disease (GvHD): While CAR T-cell therapy uses the patient's own cells, some conditioning regimens can help minimise the potential for immune-related complications.
This conditioning chemotherapy is usually given in the days leading up to the CAR T-cell infusion and typically lasts for 1 to 7 days.
Phase 5: CAR T-Cell Infusion (1-2 Hours)
Once the CAR T-cells have been manufactured and the patient has completed their conditioning chemotherapy, the CAR T-cells are infused back into the patient's bloodstream. This is a relatively quick procedure, similar to a blood transfusion, and usually takes between 1 to 2 hours.
Following the infusion, the patient is closely monitored for immediate reactions and side effects.
Phase 6: Post-Infusion Monitoring and Management (Weeks to Months)
This is a crucial and often lengthy phase. After the CAR T-cell infusion, patients are typically hospitalised for intensive monitoring for at least several weeks. This is because CAR T-cell therapy can cause significant side effects, including:
- Cytokine Release Syndrome (CRS): A potentially serious side effect where the activated CAR T-cells release cytokines, leading to a systemic inflammatory response. Symptoms can range from fever and chills to more severe organ dysfunction.
- Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS): Neurological side effects that can manifest as confusion, seizures, or speech difficulties.
- Haematological Toxicities: Effects on blood cell counts, such as low white blood cells, red blood cells, or platelets.
Medical teams closely monitor patients for these potential side effects, managing them proactively with supportive care and specific medications if necessary. Patients will undergo regular blood tests and clinical evaluations to assess their response to the therapy and monitor for any emerging complications.

The duration of hospitalisation varies but is often a minimum of 2 to 4 weeks. After discharge, patients will continue to have regular follow-up appointments for several months, and potentially longer, to assess the long-term efficacy of the treatment and monitor for any late-onset side effects or relapse.
Factors Influencing the Total Timeline
Several factors can influence the overall duration of the CAR T-cell therapy process:
- Patient's Health Status: The overall health and resilience of the patient can impact how quickly they progress through each stage and their ability to tolerate potential side effects.
- Disease Activity: The aggressiveness of the cancer and how quickly it progresses can necessitate adjustments to the treatment schedule or the use of bridging therapies.
- Manufacturing Complexity: Different CAR T-cell products may have slightly different manufacturing protocols, leading to variations in production time.
- Facility Capacity: The availability of apheresis slots, manufacturing slots, and hospital beds at specialised treatment centres can also influence the timeline.
- Response to Treatment: While not directly impacting the initial timeline, the patient's response to the therapy will dictate the long-term follow-up schedule.
Summary of Typical Timelines
To provide a clearer picture, here's a general breakdown of the typical durations:
| Phase | Typical Duration |
|---|---|
| Initial Consultation & Preparation | 1-4 Weeks |
| Apheresis | 1-3 Hours (plus processing) |
| CAR T-Cell Manufacturing | 2-6 Weeks |
| Conditioning Chemotherapy | 1-7 Days |
| CAR T-Cell Infusion | 1-2 Hours |
| Post-Infusion Hospitalisation & Monitoring | 2-4+ Weeks |
| Total Estimated Time (Active Treatment & Hospitalisation) | Approximately 1-3 Months (excluding long-term follow-up) |
It's important to reiterate that this is an estimated timeline. The entire process, from the initial diagnosis and consultation through to the completion of initial intensive monitoring, can take anywhere from one month to several months. Long-term follow-up will continue for a much longer period.
Frequently Asked Questions about CAR T-Cell Therapy Timelines
Q1: Can the manufacturing time for CAR T-cells be shortened?
While efforts are continuously made to streamline manufacturing processes, the current biological and regulatory requirements mean that the 2-6 week timeframe is generally standard. Advances in technology may lead to faster production in the future.
Q2: What happens if my cancer progresses while waiting for CAR T-cells?
If a patient's disease progresses significantly during the manufacturing period, their medical team may administer 'bridging therapy'. This is a standard chemotherapy or other treatment to help control the cancer until the CAR T-cells are ready for infusion. The decision to use bridging therapy is made on a case-by-case basis.
Q3: How long do I need to stay in the hospital after the CAR T-cell infusion?
Patients are typically hospitalised for at least 2 to 4 weeks after the infusion for close monitoring of potential side effects like CRS and ICANS. This duration can be extended depending on the patient's recovery and the management of any complications.
Q4: What is the total time commitment for CAR T-cell therapy?
The active treatment phase, including manufacturing and initial hospitalisation, typically spans 1 to 3 months. However, it's vital to remember that ongoing medical follow-up appointments are necessary for many months, and sometimes years, after the infusion to monitor for long-term efficacy and safety.
Q5: Is CAR T-cell therapy a one-time treatment?
For many patients, CAR T-cell therapy is a one-time infusion. However, in some cases, a second infusion might be considered if the initial treatment is not fully effective or if there is a relapse of the cancer. This decision is made by the treating physician based on the individual's circumstances.
Conclusion
Understanding the timeline for CAR T-cell therapy is an essential part of the patient experience. While the process involves multiple stages and can take several months from start to finish, it's crucial to remember that this is a highly personalised and potentially life-saving treatment. Open communication with your medical team is key to navigating this complex journey. They will provide the most accurate and up-to-date information regarding your specific situation and the expected timelines. The investment of time and the rigorous nature of the process are ultimately aimed at achieving the best possible outcomes in the fight against cancer, offering a beacon of hope for many.
If you want to read more articles similar to CAR T-Cell Therapy: The Timeline Unveiled, you can visit the Automotive category.

