23/04/2026
In the ever-evolving landscape of cancer treatment, a groundbreaking therapy has emerged, offering a new beacon of hope for patients facing challenging diagnoses. This innovative approach, known as CAR T-cell therapy, represents a significant leap forward in immunotherapy, harnessing the body's own immune system to wage a targeted war against cancer cells. It's a highly specialised and complex treatment, but one that has demonstrated remarkable potential in specific types of blood cancers, transforming the outlook for many. If you or a loved one are exploring advanced cancer care options, understanding CAR T-cell therapy is crucial, as it may open doors to previously unavailable avenues of treatment.

- Understanding Your Immune System's Defenders: T-cells
- What Exactly is CAR T-cell Therapy?
- How CAR T-cells Target Cancer
- Who Can Benefit from CAR T-cell Therapy?
- Life After CAR T-cell Infusion: Monitoring and Care
- Potential Side Effects of CAR T-cell Therapy
- Frequently Asked Questions About CAR T-cell Therapy
- What are T-cells and why are they important?
- How are my T-cells collected?
- What happens to my T-cells in the laboratory?
- Why do I need chemotherapy before receiving the CAR T-cells?
- What are the most serious side effects I should be aware of?
- How long will I need to stay in hospital after CAR T-cell therapy?
- Is CAR T-cell therapy available for all types of cancer?
- Conclusion
Understanding Your Immune System's Defenders: T-cells
To truly grasp the ingenuity behind CAR T-cell therapy, it's helpful to first understand the role of T-cells within our immune system. Lymphocytes, a type of white blood cell, are the frontline soldiers in our body's defence against infections and diseases, including cancer. Among these, T-cells are particularly vital. Their primary function is to patrol the body, identifying and destroying abnormal or infected cells. When our body encounters a new threat, such as a virus or a developing cancer, specific T-cells are mobilised and multiplied to combat that particular invader. Crucially, the immune system retains a 'memory' of these encounters, allowing for a swift and robust response if the same threat reappears. However, cancer cells are often adept at evading this natural surveillance, sometimes appearing indistinguishable from healthy cells, thus escaping the T-cells' detection.
What Exactly is CAR T-cell Therapy?
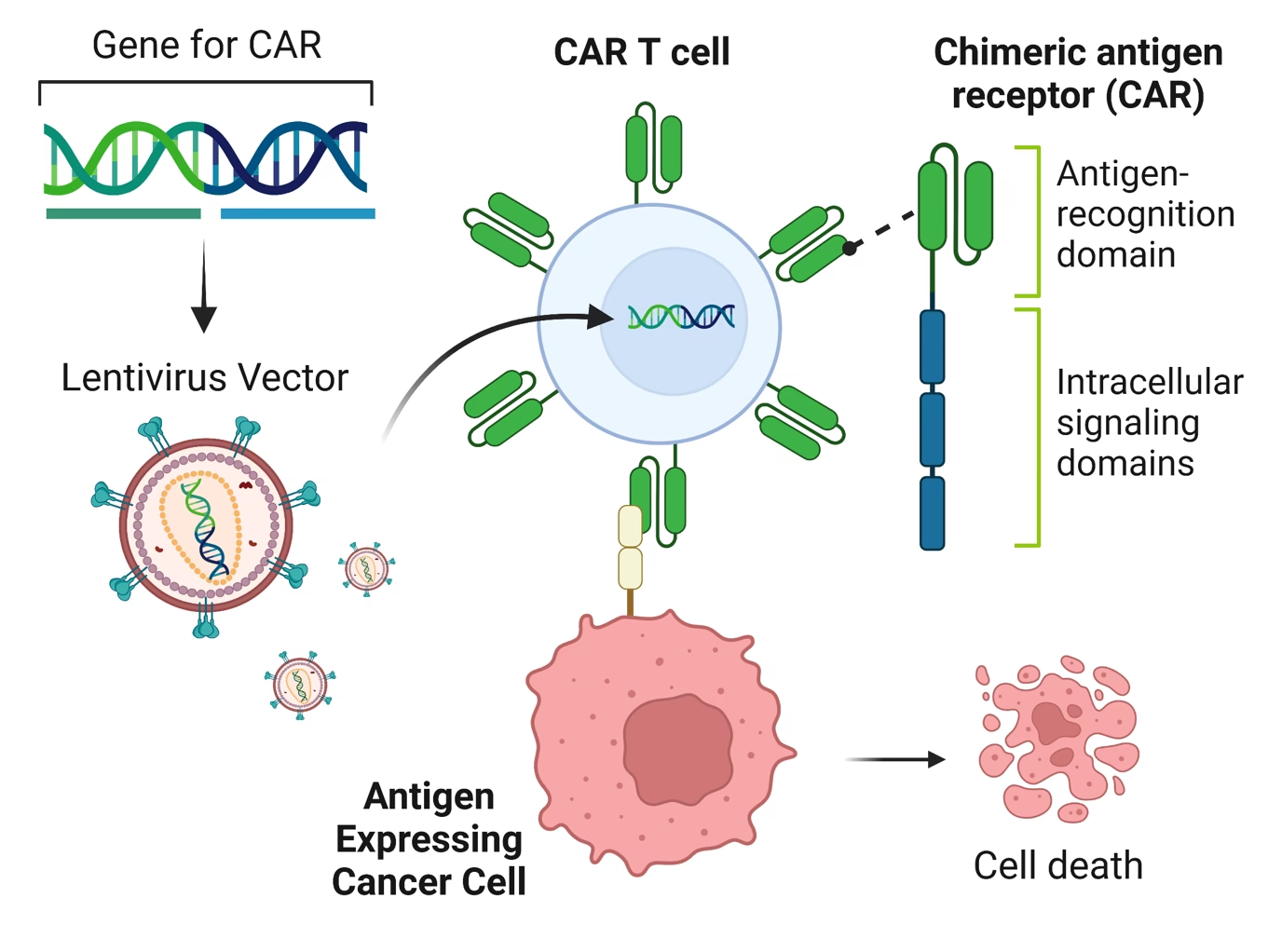
CAR T-cell therapy, where CAR stands for chimeric antigen receptor, is a sophisticated form of immunotherapy, sometimes referred to as adoptive cell transfer. It's a highly complex and specialist treatment available only in registered CAR T-cell therapy hospitals, staffed by healthcare professionals with the specific expertise required for its administration and patient care. The fundamental principle involves genetically modifying a patient's own T-cells to enhance their ability to recognise and destroy cancer cells.
The Process: From Collection to Infusion
The journey of CAR T-cell therapy involves several distinct stages:
- T-cell Collection (Apheresis): The process begins with the collection of your T-cells. Before this, your doctor might advise you to pause other treatments, such as steroids or chemotherapy, to help boost the number and quality of T-cells circulating in your blood. You'll also undergo various tests to ensure you are fit enough for the collection. During the procedure, a specialist nurse will insert a tube (cannula) into a vein in each arm. If your arm veins aren't suitable, a special tube like a vascath or a central line might be used. One tube draws blood into an aphresis machine, which then separates the different components of your blood, specifically extracting your T-cells. The remaining blood cells and fluid are returned to your body through the cannula in your other arm. This collection process typically takes between 4 to 6 hours.
- Laboratory Engineering: Once collected, your T-cells are sent to a specialised laboratory. Here, scientists genetically engineer them to become CAR T-cells. This involves adding a gene that instructs the T-cells to produce unique proteins on their surface called chimeric antigen receptors (CARs). These CARs are designed to specifically recognise and bind to a particular protein found on the surface of the cancer cells, acting like a highly targeted homing missile.
- Multiplication and Freezing: The newly engineered CAR T-cells are then grown and multiplied in the laboratory until there are enough for treatment. This stage can take several weeks. Once the required number of CAR T-cells is achieved, they are frozen until you are ready for infusion.
- Bridging Treatment (Optional): While your CAR T-cells are being prepared in the lab, some patients may require 'bridging treatment,' such as chemotherapy or radiotherapy. This is administered to help keep the cancer under control and prevent its progression until the CAR T-cells are ready for infusion.
- Lymphodepletion: Immediately before you receive the CAR T-cells, you will undergo a short course of chemotherapy over a few days, typically starting about a week beforehand. This crucial step, known as lymphodepletion or conditioning treatment, aims to lower and kill existing T-cells in your blood. This creates 'space' and a more favourable environment within your body, allowing the newly infused CAR T-cells to expand and function effectively without competition.
- CAR T-cell Infusion: On the day of infusion, a specially trained nurse will defrost the CAR T-cells on the ward. You'll be given medicines intravenously to help prevent any allergic reactions. The CAR T-cells are then administered as a drip directly into your bloodstream, usually taking less than 30 minutes. You will be closely monitored throughout and after this procedure.
How CAR T-cells Target Cancer
The beauty of CAR T-cell therapy lies in its targeted mechanism. Once infused, the genetically modified CAR T-cells circulate in your bloodstream. Their newly acquired chimeric antigen receptors enable them to specifically 'see' and attach to the unique protein on the surface of the cancer cells, which your natural T-cells might have missed. Upon binding, the CAR T-cells become activated, unleashing a powerful immune response that directly attacks and destroys the cancer cells. A remarkable feature of these engineered cells is their ability to persist in your body for extended periods, continuously recognising and eliminating the specific cancer cells, potentially offering long-term disease control.
Who Can Benefit from CAR T-cell Therapy?
CAR T-cell therapy is a highly specific treatment, currently available for certain children and adults with particular types of leukaemia and lymphoma. Eligibility is determined by a national panel of specialists and patient advocates (such as the National CAR T Clinical Panel - NCCP) who consider many factors for each individual's situation. It's crucial to discuss with your healthcare team if you believe this treatment might be suitable for you.
Here's a summary of the conditions for which CAR T-cell therapies are currently available:
| CAR T-cell Therapy Type | Manufacturer | Eligible Conditions | Patient Age Group |
|---|---|---|---|
| Tisagenlecleucel (Kymriah) | (Information not provided) | B cell acute lymphoblastic leukaemia (B cell ALL) | Children and young people up to 25 years old |
| Brexucabtagene autoleucel (Tecartus) | (Information not provided) | B cell acute lymphoblastic leukaemia (B cell ALL) Mantle cell lymphoma (MCL) (for some in England & Wales) | People 26 years of age and over (for ALL) Adults (for MCL) |
| Axicabtagene ciloleucel (Yescarta) | (Information not provided) | Diffuse large B cell lymphoma (DLBCL) Primary mediastinal B cell lymphoma Transformed follicular lymphoma | Adults |
People with other types of cancer might also have the opportunity to receive CAR T-cell therapy as part of a clinical trial, contributing to the ongoing research and expansion of this innovative field.
Life After CAR T-cell Infusion: Monitoring and Care
The period immediately following CAR T-cell infusion is critical and requires very close monitoring due to the potential for significant side effects. Patients typically remain in hospital for at least the first two weeks after treatment.
Post-Discharge Requirements
If you are well enough after two weeks, you may be able to be discharged, provided you meet certain criteria. One key requirement is living within one hour of the hospital. Alternatively, the hospital may arrange for you to stay in a nearby hotel or residence. Crucially, you must have someone with you 24 hours a day for continuous support and observation. If this 24-hour care cannot be arranged, you will need to remain in hospital for the entire first month post-treatment.
Ongoing Outpatient Care
Even after discharge, close monitoring continues. This involves frequent visits to a day care unit, also known as an ambulatory clinic or 'Ambi-care', several times a week. On days you don't attend the clinic, you'll receive phone calls to check on your well-being. You will also be provided with contact numbers for your healthcare team should any problems arise. Your healthcare team generally advises that you stay within one hour of the hospital for up to 30 days following treatment, though specific advice may vary slightly between different CAR T-cell therapy hospitals.
Potential Side Effects of CAR T-cell Therapy
As with all powerful medical treatments, CAR T-cell therapy can cause side effects. Given that it's a relatively new treatment, doctors are continually learning about all possible effects. The specific side effects can also depend on the type of CAR T-cell therapy received. Your healthcare team will discuss all potential side effects with you in detail.

Common Side Effects Include:
- Allergic Reaction: Though nurses administer preventative medicines beforehand, some patients may experience an allergic reaction to the CAR T-cells. Symptoms can include a high temperature (fever), chills, feeling or being sick, and difficulty breathing. It's vital to inform your nurse immediately if you experience any of these symptoms, as they will monitor you closely.
- Cytokine Release Syndrome (CRS): This is a common and potentially serious side effect. Cytokines are proteins that play a vital role in boosting the immune system. CAR T-cell therapy can stimulate the immune system to release a large amount of these cytokines, leading to a systemic inflammatory response. Symptoms of cytokine release syndrome can range from mild to severe and include fever, chills, dizziness due to low blood pressure, difficulty breathing, headaches, and a general feeling of being unwell, often accompanied by tiredness and weakness (fatigue). Your medical team will monitor you closely. Depending on the severity, treatment with a monoclonal antibody called tocilizumab, and sometimes steroids, may be necessary. In severe cases, patients might require care in an Intensive Care Unit (ICU). CRS typically manifests within the first couple of weeks after treatment.
- Neurological Side Effects (Neurotoxicity): CAR T-cells can sometimes affect the brain, leading to what doctors term neurotoxicity, also known as immune effector cell associated neurotoxicity syndrome (ICANS). These symptoms can be mild or severe and may include shaking (tremor), sudden confusion and agitation, disorientation, hallucinations (seeing things that aren't there), headaches, changes in awareness or consciousness, changes in speech (such as difficulty speaking or understanding), and even seizures (fits). Your healthcare team will continuously monitor you for any such changes. While some symptoms may resolve on their own, others might require treatment, often with steroids.
- Increased Risk of Infection: Certain types of CAR T-cell therapy increase the risk of infection. This is because many CAR T-cell therapies are designed to target a protein called CD19, which is present on the surface of most B cells. B cells are another type of white blood cell crucial for fighting infections. By targeting CD19, the therapy destroys both cancerous and normal B cells, significantly reducing their numbers or eliminating them entirely. This leaves the body vulnerable to infections. To counteract this, some patients may receive immunoglobulin therapy, which provides antibodies to help fight off infections.
- Tumour Lysis Syndrome (TLS): This can occur when a large number of cancer cells are rapidly destroyed by the CAR T-cells. As these cells break down, they release their contents, including various chemicals, into the bloodstream. This rapid influx can disrupt the body's normal chemical balance, affecting levels of potassium, sodium, phosphates, and uric acid. Abnormally high levels of these chemicals can negatively impact heart rhythm and kidney function. Regular blood tests are performed to detect TLS. If it develops, treatment typically involves intravenous fluids and medication to help lower uric acid levels in the blood.
Frequently Asked Questions About CAR T-cell Therapy
What are T-cells and why are they important?
T-cells are a type of white blood cell, specifically lymphocytes, which are a critical part of your immune system. They are responsible for identifying and destroying abnormal cells, including those infected by viruses or cancer cells. They are vital for fighting off infections and diseases.
How are my T-cells collected?
Your T-cells are collected through a process called apheresis. This involves drawing blood from one arm, passing it through a machine that separates and collects the T-cells, and then returning the rest of your blood components to your body through a vein in your other arm. This procedure typically takes 4 to 6 hours.
What happens to my T-cells in the laboratory?
In the laboratory, your T-cells undergo genetic modification. Scientists introduce a new gene that enables them to produce special proteins called chimeric antigen receptors (CARs) on their surface. These CARs are designed to specifically recognise and attach to proteins found on the surface of your cancer cells, essentially transforming them into 'super T-cells' capable of targeting the cancer.
Why do I need chemotherapy before receiving the CAR T-cells?
Before the CAR T-cells are infused, you'll undergo a short course of chemotherapy known as lymphodepletion. This is done to reduce the number of your existing T-cells, creating a more suitable environment for the newly introduced CAR T-cells to expand and function effectively without competition, thereby enhancing the treatment's success.
What are the most serious side effects I should be aware of?
The most significant side effects are Cytokine Release Syndrome (CRS) and neurological side effects (neurotoxicity or ICANS). CRS involves a systemic inflammatory response with symptoms like fever, chills, and low blood pressure. Neurotoxicity can cause confusion, headaches, speech difficulties, and even seizures. Your healthcare team will monitor you very closely for these and other side effects.
How long will I need to stay in hospital after CAR T-cell therapy?
You will be closely monitored in hospital for at least the first two weeks after the infusion. If you are well and meet specific criteria, including living within an hour of the hospital and having 24-hour care at home, you may be discharged. Otherwise, you might need to stay longer, potentially for up to a month.
Is CAR T-cell therapy available for all types of cancer?
Currently, CAR T-cell therapy is a possible treatment option for specific types of relapsed or refractory B cell acute lymphoblastic leukaemia (ALL) in children and adults, and for certain adults with relapsed or refractory diffuse large B cell lymphoma (DLBCL), primary mediastinal large B cell lymphoma (PMBCL), and mantle cell lymphoma (MCL). It is not yet widely available for all cancer types, though research continues.
Conclusion
CAR T-cell therapy represents a remarkable achievement in medical science, offering a powerful, personalised approach to fighting cancer by harnessing the body's own immune system. While it is a complex treatment with potential side effects, for eligible patients with specific blood cancers, it has provided a profound new avenue for treatment where other options may have been exhausted. Ongoing research and clinical trials continue to expand our understanding and the potential applications of this innovative therapy, solidifying its place as a significant advancement in the battle against cancer.
If you want to read more articles similar to CAR T-cell Therapy: A New Horizon in Cancer Treatment, you can visit the Automotive category.