27/03/2001
CAR-T therapy represents a significant leap forward in the fight against certain types of cancer. This innovative treatment harnesses the power of a patient's own immune system to target and destroy cancerous cells. For individuals residing in Cheshire and Merseyside, understanding where to access this life-changing therapy is crucial. This article aims to provide comprehensive information on the availability and specifics of CAR-T treatment within this region, focusing on the key provider and what patients can expect.

- What is CAR-T Therapy?
- The Clatterbridge Cancer Centre: A Beacon of Hope in Cheshire & Merseyside
- Location and Patient Admission
- The CAR-T Treatment Process: A Personalised Journey
- Eligibility and Conditions Treated
- What to Expect as a Patient
- Key Benefits of CAR-T Therapy
- Challenges and Side Effects
- Frequently Asked Questions
- Conclusion
What is CAR-T Therapy?
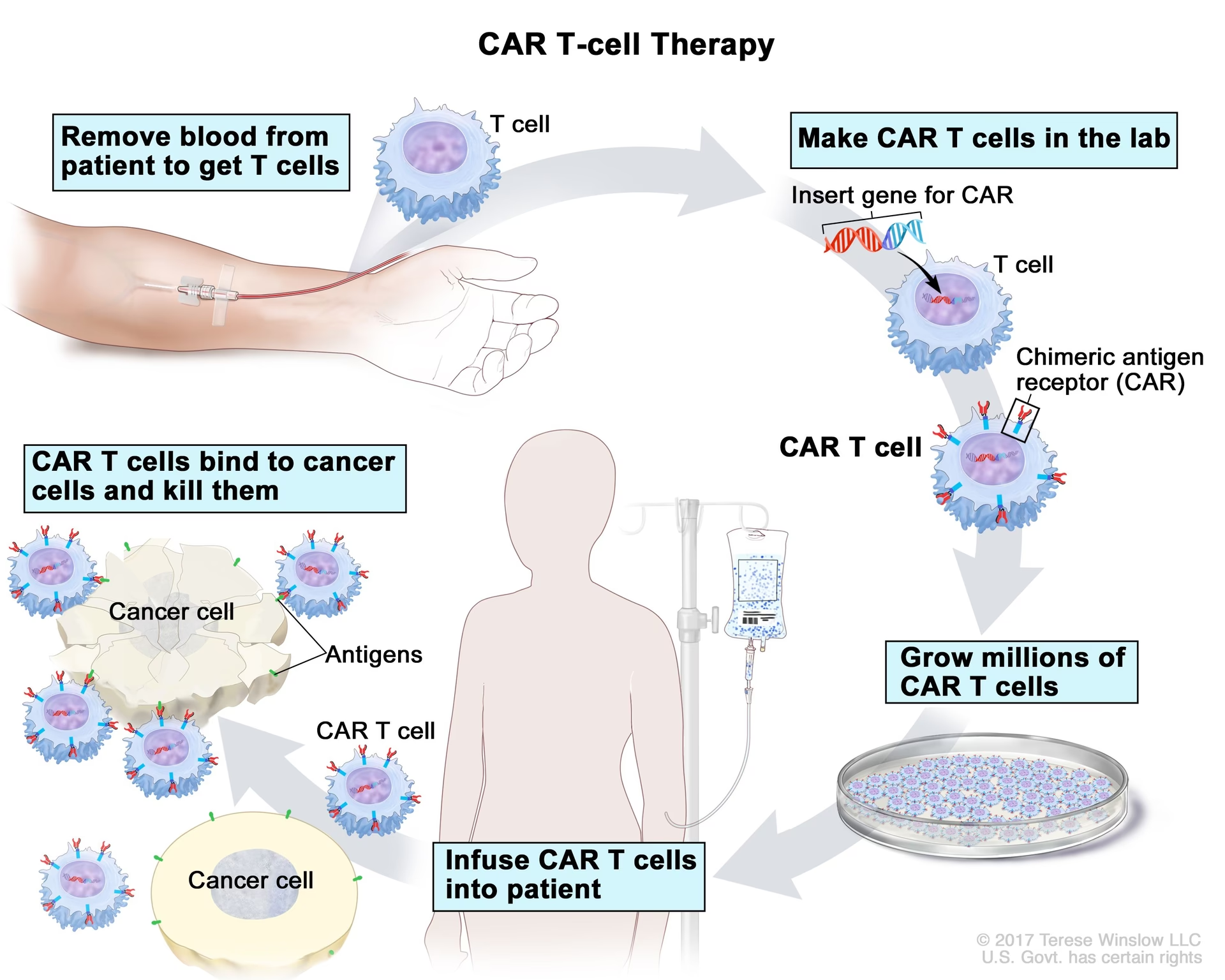
CAR-T stands for Chimeric Antigen Receptor T-cell therapy. It's a form of immunotherapy, a treatment that uses the body's immune system to fight diseases. In essence, CAR-T therapy involves genetically modifying a patient's own T-cells (a type of white blood cell) to recognise and attack specific cancer cells. These modified T-cells, now equipped with the CAR, are then reinfused into the patient's body. Once back in circulation, they act as a targeted army, seeking out and destroying cancer cells that express the specific antigen the CAR is designed to recognise. This personalised approach offers a new avenue of hope for patients with certain blood cancers that have been resistant to traditional treatments.
The Clatterbridge Cancer Centre: A Beacon of Hope in Cheshire & Merseyside
For residents of Cheshire and Merseyside, and indeed the surrounding areas, The Clatterbridge Cancer Centre stands as the primary provider of CAR-T therapy. This esteemed institution is at the forefront of cancer care, offering advanced treatments and dedicated expertise. The centre has established a specialised CAR-T therapy unit, ensuring that patients receive the highest standard of care in a state-of-the-art facility. This commitment to innovation means that cutting-edge treatments are accessible locally, reducing the need for patients to travel extensively for specialised care.
Location and Patient Admission
The CAR-T therapy unit is conveniently located on Level 5 of The Clatterbridge Cancer Centre – Liverpool. This modern facility is specifically designed to accommodate the complex needs of patients undergoing CAR-T treatment. Patients receiving this therapy are admitted to Ward 5 within the Liverpool centre. This dedicated ward ensures that patients are under constant observation by a highly trained medical team, ready to manage any aspect of their treatment and recovery. The focus on a specific unit and ward underscores the specialised nature of CAR-T therapy and the centre's commitment to providing focused, expert care.
The CAR-T Treatment Process: A Personalised Journey
The beauty of CAR-T therapy lies in its highly personalised nature. The process begins with the collection of a patient's own T-cells. This is typically done through a process called apheresis, where blood is drawn from the patient, and the T-cells are separated. These T-cells are then sent to a specialised laboratory where they are genetically engineered to produce chimeric antigen receptors (CARs) on their surface. These CARs are designed to recognise a specific protein (antigen) found on the surface of the patient's cancer cells.
Once the T-cells have been successfully modified and expanded in the lab, they are frozen and transported back to The Clatterbridge Cancer Centre. Before the CAR-T cells are infused back into the patient, the patient often undergoes a conditioning chemotherapy regimen. This helps to prepare the body by reducing the number of existing immune cells, making space for the new CAR-T cells to effectively proliferate and fight the cancer. Finally, the CAR-T cells are infused into the patient's bloodstream, much like a blood transfusion.
Following the infusion, patients remain in the hospital for a period of close monitoring. This is a critical phase as the CAR-T cells begin to work. Doctors will monitor for efficacy (how well the treatment is working) and potential side effects. Common side effects can include cytokine release syndrome (CRS), which is a systemic inflammatory response, and neurological toxicities. The medical team at Ward 5 is expertly trained to manage these potential complications promptly and effectively, ensuring patient safety and well-being throughout the process.
Eligibility and Conditions Treated
CAR-T therapy is not suitable for all cancer types or all patients. It is typically used for certain types of blood cancers, such as some forms of leukaemia and lymphoma, particularly in cases where other treatments have not been successful. The decision to proceed with CAR-T therapy is made by a multidisciplinary team of specialists, including oncologists, haematologists, and other healthcare professionals, who will carefully assess each patient's individual case, including their medical history, the specific type and stage of cancer, and their overall health status. Patients and their families are encouraged to have open discussions with their medical team about the potential benefits, risks, and alternatives to CAR-T therapy.
What to Expect as a Patient
Undergoing CAR-T therapy is a significant undertaking. Patients can expect a comprehensive process that involves:
- Initial Consultation and Assessment: A thorough evaluation to determine eligibility.
- T-cell Collection: A procedure to harvest the patient's T-cells.
- Manufacturing: The process of genetically modifying and expanding the T-cells in the laboratory, which can take several weeks.
- Conditioning Chemotherapy: Preparation of the body prior to CAR-T cell infusion.
- CAR-T Cell Infusion: The reintroduction of the modified T-cells into the patient.
- Hospital Stay and Monitoring: A period of intensive observation for treatment effectiveness and side effects.
- Follow-up Care: Ongoing monitoring and support after discharge to track long-term outcomes.
The Clatterbridge Cancer Centre provides a supportive environment throughout this journey, with dedicated nursing staff and clinical nurse specialists available to guide patients and their families.
Key Benefits of CAR-T Therapy
The advantages of CAR-T therapy are numerous, offering new hope where previously there was little:
- High Efficacy: For some patients with relapsed or refractory cancers, CAR-T therapy has shown remarkable success rates, leading to long-term remission.
- Personalised Treatment: Utilising the patient's own cells minimises the risk of graft-versus-host disease, a common complication in stem cell transplants.
- Targeted Approach: The therapy specifically targets cancer cells, potentially reducing damage to healthy tissues compared to some conventional treatments.
- Potential for Cure: In certain patient populations, CAR-T therapy has demonstrated the potential for a cure.
Challenges and Side Effects
While CAR-T therapy is revolutionary, it is not without its challenges and potential side effects. The most significant concerns include:
- Cytokine Release Syndrome (CRS): This is a common and potentially serious side effect caused by the rapid activation and proliferation of CAR-T cells, leading to a systemic inflammatory response. Symptoms can range from flu-like symptoms to severe organ dysfunction.
- Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS): Neurological side effects can also occur, including confusion, seizures, and speech difficulties.
- Manufacturing Complexity: The process of producing CAR-T cells is complex, time-consuming, and expensive.
- Limited Availability: As a highly specialised treatment, access can be limited by the availability of treatment centres and trained personnel.
The medical team at The Clatterbridge Cancer Centre is vigilant in monitoring for and managing these side effects, with established protocols in place to mitigate risks.
Frequently Asked Questions
Q1: Who is eligible for CAR-T therapy at The Clatterbridge Cancer Centre?
Eligibility is determined on a case-by-case basis by the specialist medical team. It is typically considered for patients with specific types of relapsed or refractory blood cancers.
Q2: How long does the CAR-T therapy process take?
The entire process, from T-cell collection to infusion and initial recovery, can span several weeks to months, including the time required for laboratory manufacturing.
Q3: What are the main side effects of CAR-T therapy?
The most common and significant side effects are Cytokine Release Syndrome (CRS) and neurotoxicity (ICANS).
Q4: Is CAR-T therapy available anywhere else in the North West?
The Clatterbridge Cancer Centre is the designated provider for CAR-T therapy for Cheshire and Merseyside and surrounding areas, offering specialised services within the region.
Q5: What support is available for patients and their families?
The Clatterbridge Cancer Centre provides comprehensive support, including specialist nurses, psychological support services, and information resources for patients and their families throughout the treatment journey.
Conclusion
For individuals in Cheshire and Merseyside facing certain blood cancers, The Clatterbridge Cancer Centre offers a vital and advanced treatment option in the form of CAR-T therapy. Located at their Liverpool facility, the centre provides a specialised unit and dedicated ward to ensure the highest quality of care. While the treatment journey is complex and requires careful management of potential side effects, the personalised nature and remarkable efficacy of CAR-T therapy offer a profound sense of hope and the potential for significant clinical benefit. Patients considering this groundbreaking treatment are encouraged to engage in detailed discussions with their oncology team to understand the process, benefits, and risks involved.
If you want to read more articles similar to CAR-T Therapy in Cheshire & Merseyside, you can visit the Automotive category.