16/05/2019
Oxygen, the very breath of life, holds fascinating secrets within its molecular structure. While simple Lewis structures or valence bond theory can describe many molecules, they fall short when it comes to explaining some of oxygen's most peculiar and vital characteristics. This is where Molecular Orbital (MO) Theory steps in, offering a sophisticated and remarkably accurate picture of chemical bonding, particularly for diatomic molecules like O2. Understanding its molecular orbital energy-level diagram is key to unlocking the enigma of its behaviour, including its unusual magnetic properties.
MO theory postulates that when atomic orbitals combine, they form new molecular orbitals that span the entire molecule. These molecular orbitals can be either bonding (lower energy, stabilising) or antibonding (higher energy, destabilising). Unlike localised bonding models, MO theory provides a delocalised view of electrons, which is crucial for explaining phenomena such as paramagnetism in O2.
- The Fundamental Principles of Molecular Orbital Theory
- Constructing the Molecular Orbital Diagram for O2
- The Paramagnetism Puzzle Solved
- Energy Level Reversal: A Nuance for Lighter Diatomics
- Beyond Homonuclear Diatomics: Heteronuclear Molecules
- Frequently Asked Questions About O2 Molecular Orbital Diagrams
The Fundamental Principles of Molecular Orbital Theory
Before we delve into the specifics of oxygen, it's essential to grasp the core tenets that guide the construction of any molecular orbital diagram. These four key points are paramount:
- Conservation of Orbitals: The number of molecular orbitals produced is always equal to the number of atomic orbitals used to create them. If two atomic orbitals combine, they will yield two molecular orbitals (one bonding, one antibonding). This is often referred to as the "law of conservation of orbitals."
- Overlap and Energy Difference: As the overlap between two atomic orbitals increases, the difference in energy between the resulting bonding and antibonding molecular orbitals also increases. Greater overlap leads to stronger bonds and more significant energy splitting.
- Stabilisation and Destabilisation: When two atomic orbitals combine, the bonding molecular orbital is stabilised by approximately the same amount as the corresponding antibonding molecular orbital is destabilised. This ensures that the overall energy balance is maintained.
- Energy Matching: The interaction between atomic orbitals is greatest, and thus the most effective bonding occurs, when they have similar energies. Orbitals with vastly different energies interact minimally.
These principles form the bedrock for constructing accurate energy-level diagrams and predicting molecular properties.
Constructing the Molecular Orbital Diagram for O2
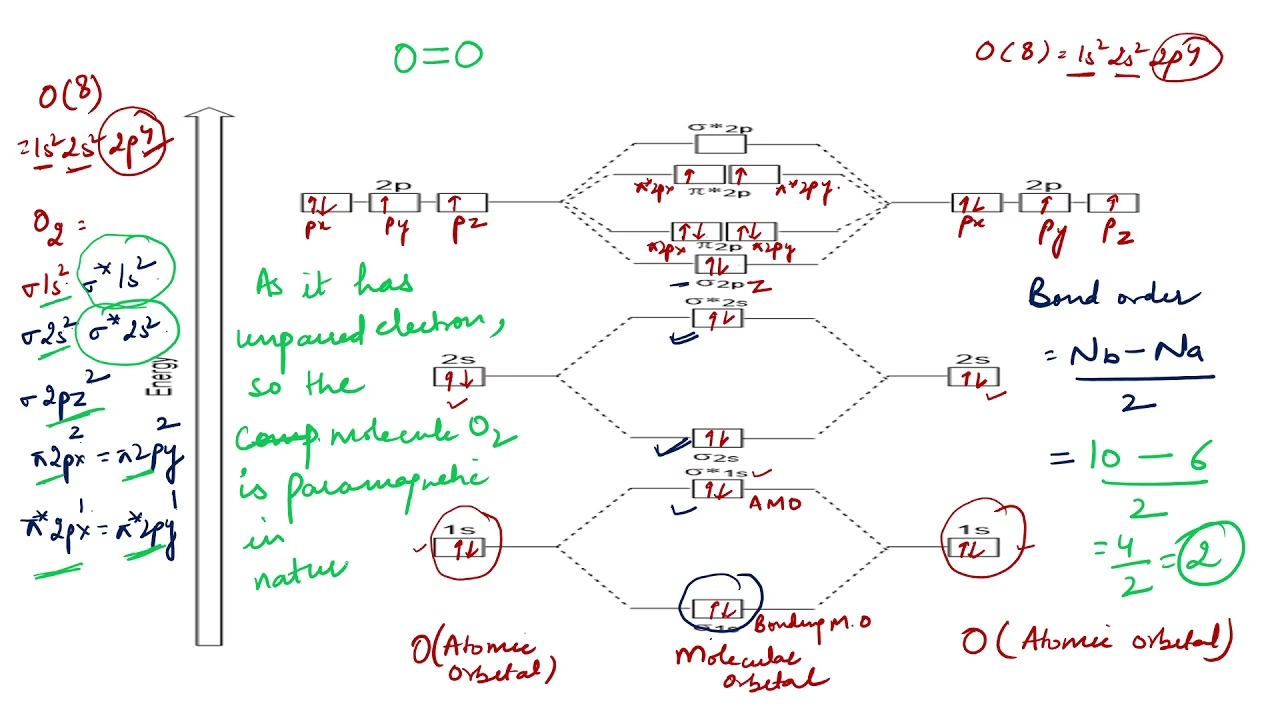
To construct the molecular orbital energy-level diagram for O2, we must first consider the atomic orbitals of the individual oxygen atoms. Each oxygen atom has an electron configuration of 1s22s22p4. For bonding purposes, we primarily focus on the valence electrons in the outermost shell, which are the 2s and 2p electrons. Each oxygen atom contributes 6 valence electrons, meaning the O2 molecule has a total of 12 valence electrons.
The molecular orbitals are formed by combining these atomic orbitals. The 1s orbitals are typically much lower in energy and form non-bonding core orbitals, so they are often excluded from simplified valence MO diagrams. We focus on the 2s and 2p orbitals:
Formation of Sigma (σ) Orbitals
The two 2s atomic orbitals, one from each oxygen atom, combine to form two molecular orbitals: a bonding sigma (σ2s) orbital and an antibonding sigma-star (σ2s) orbital. The σ2s orbital is lower in energy than the original 2s atomic orbitals, while the σ2s orbital is higher.
Next, we consider the three 2p atomic orbitals from each oxygen atom (2px, 2py, 2pz). One pair of 2p orbitals, typically designated as 2pz, aligns head-on along the internuclear axis to form a sigma (σ) bond. This interaction produces a bonding σ2pz molecular orbital and an antibonding σ2pz molecular orbital.
Formation of Pi (π) Orbitals
The remaining two pairs of 2p orbitals (2px and 2py) are perpendicular to the internuclear axis. They overlap side-on to form pi (π) bonds. This results in two degenerate (equal energy) bonding π2px and π2py molecular orbitals, and two degenerate antibonding π2px and π2py molecular orbitals. The π orbitals are generally higher in energy than the σ2pz orbital for elements like oxygen and fluorine, due to less effective overlap compared to the head-on sigma interaction.
Filling the Molecular Orbitals for O2
Once the energy levels of the molecular orbitals are established, we fill them with the 12 valence electrons according to the Pauli exclusion principle (maximum two electrons per orbital with opposite spins) and Hund's rule (electrons fill degenerate orbitals singly before pairing up).
The filling order for O2 is as follows, from lowest to highest energy:
- σ2s (2 electrons)
- σ2s (2 electrons)
- σ2pz (2 electrons)
- π2px and π2py (4 electrons, 2 in each degenerate orbital)
- π2px and π2py (2 electrons)
It is at the π* antibonding orbitals where Hund's rule becomes critical for O2. With two degenerate π* orbitals and only two electrons remaining, these electrons will occupy each orbital separately with parallel spins, rather than pairing up in one. This leads to the presence of two unpaired electrons in the O2 molecule.
Calculating the Bond Order
The bond order in MO theory is calculated as: (Number of bonding electrons - Number of antibonding electrons) / 2.
For O2:
- Bonding electrons: 2 (σ2s) + 2 (σ2pz) + 4 (π2px, π2py) = 8 bonding electrons.
- Antibonding electrons: 2 (σ2s) + 2 (π2px, π2py) = 4 antibonding electrons.
Bond Order = (8 - 4) / 2 = 2.
This predicted bond order of 2 aligns perfectly with experimental data, indicating a double bond between the two oxygen atoms. The O-O bond length (120.7 pm) and bond energy (498.4 kJ/mol) are consistent with a strong double bond.
The Paramagnetism Puzzle Solved
Perhaps the most significant triumph of MO theory in explaining O2 is its ability to account for oxygen's unique magnetic properties. Unlike most substances, liquid O2 is attracted to a magnetic field and can even be suspended between the poles of a powerful magnet. This behaviour, known as paramagnetism, is a direct consequence of the presence of unpaired electrons.
Classical bonding models, such as Lewis structures, depict O2 with all electrons paired, implying it should be diamagnetic (repelled by a magnetic field). However, MO theory's prediction of two unpaired electrons in the π orbitals perfectly explains its observed paramagnetism. This was one of the earliest and most compelling pieces of evidence favouring MO theory over older bonding approaches.
The Crucial Role of Paramagnetism in Life
The magnetic properties of O2 are not merely a scientific curiosity; they are absolutely fundamental to life on Earth. Earth's atmosphere contains approximately 20% oxygen, and organic compounds, including those that make up our bodies, are inherently reactive with oxygen. Without a significant energy barrier, these compounds would rapidly oxidise to form water, carbon dioxide, and nitrogen in highly exothermic reactions.
Fortunately, this reaction is incredibly slow. The reason for this unexpected stability of organic compounds in an oxygen atmosphere lies in the spin of electrons. Most organic compounds have only paired electrons, whereas oxygen possesses two unpaired electrons with parallel spins. For a reaction between O2 and an organic compound to occur, at least one of these unpaired electrons in O2 would need to change its spin. This requires a substantial input of energy, a hurdle chemists refer to as a spin barrier. Consequently, such reactions are usually exceedingly slow, allowing life to flourish.
Energy Level Reversal: A Nuance for Lighter Diatomics
While the molecular orbital diagram for O2 and F2 follows a specific energy ordering (σ2pz lower than π2p), it's important to note that for lighter period 2 diatomic molecules (Li2, Be2, B2, C2, and N2), there's a slight reversal in the energy levels. For these molecules, the σ2pz molecular orbital is slightly higher in energy than the degenerate π2px and π2py orbitals.
This phenomenon is attributed to the increasing difference in energy between the 2s and 2p atomic orbitals as the nuclear charge increases across the period. In lighter elements, the 2s and 2p atomic orbitals are closer in energy, leading to significant mixing between the σ2s and σ2pz molecular orbitals. This s-p mixing pushes the σ2pz orbital to a higher energy level, above the π2p orbitals. As we move towards oxygen and fluorine, the 2s and 2p energy gap widens, reducing this mixing and causing the σ2pz to drop below the π2p levels again.
For example, in N2, with 10 valence electrons, the filling order places electrons in σ2s, σ2s, π2px, π2py, and then σ2pz. This results in 8 bonding and 2 antibonding electrons, yielding a bond order of (8-2)/2 = 3, consistent with the observed N≡N triple bond, which is exceptionally strong and short.
| Molecule | Valence Electrons | Bonding Electrons | Antibonding Electrons | Bond Order | Magnetic Property | Key MO Energy Order |
|---|---|---|---|---|---|---|
| N2 | 10 | 8 | 2 | 3 | Diamagnetic | π2p < σ2pz |
| O2 | 12 | 8 | 4 | 2 | Paramagnetic | σ2pz < π2p |
| F2 | 14 | 8 | 6 | 1 | Diamagnetic | σ2pz < π2p |
Beyond Homonuclear Diatomics: Heteronuclear Molecules
Molecular orbital theory is not limited to molecules composed of identical atoms (homonuclear diatomics). It can also elegantly describe heteronuclear diatomic molecules, which consist of two different atoms. When non-identical atoms combine, their atomic orbitals do not have the same energy. The atomic orbitals of the more electronegative atom will be uniformly lower in energy than those of the less electronegative atom.
This energy difference results in a "skewed" molecular orbital energy-level diagram. The bonding molecular orbitals will be closer in energy to the atomic orbitals of the more electronegative atom, meaning the electron density of bonding electrons will be concentrated closer to that atom. This beautifully explains the concept of a polar covalent bond, where electrons are not shared equally.
Consider nitric oxide (NO), a molecule with an odd number of valence electrons (5 from N, 6 from O, total 11). Neither Lewis structures nor valence bond theory can fully explain its bonding. The MO diagram for NO, similar to O2 but skewed towards the more electronegative oxygen, shows the 11th electron occupying one of the π orbitals, making NO paramagnetic. Its predicted bond order is (8-3)/2 = 2.5, which is consistent with experimental data falling between a double and triple bond. This highlights MO theory's capability to describe molecules with odd electron counts.
Another example is HCl. The H 1s orbital interacts primarily with the Cl 3pz orbital. The Cl 3s, 3px, and 3py orbitals, having no effective overlap with H 1s, remain largely unchanged in energy and are termed nonbonding molecular orbitals. Electrons in these orbitals act as lone pairs and do not contribute to the bond order. The bond order for HCl is (2-0)/2 = 1, representing a single bond. The MO diagram also shows the bonding sigma orbital is closer in energy to chlorine's atomic orbitals, indicating the bond's polarity with electron density concentrated on the more electronegative chlorine.
Frequently Asked Questions About O2 Molecular Orbital Diagrams
How many valence electrons are there in O2?
Each oxygen atom has 6 valence electrons (2s22p4). Since O2 consists of two oxygen atoms, there are a total of 12 valence electrons in the O2 molecule.
What are the three most important molecular orbitals for O2 bonding?
While many orbitals are involved, the most crucial for describing O2's unique properties are the σ2pz, π2px, and π2py bonding orbitals, and especially the π2px and π2py antibonding orbitals. The presence of unpaired electrons in these degenerate π* orbitals is what gives O2 its distinctive paramagnetic character.
Why is O2 paramagnetic according to MO theory?
O2 is paramagnetic because its molecular orbital energy-level diagram shows two unpaired electrons in the degenerate π2px and π2py antibonding orbitals. According to Hund's rule, electrons fill degenerate orbitals singly before pairing up, leading to these unpaired spins. This directly explains why O2 is attracted to a magnetic field.
How does the MO diagram for O2 differ from that of N2?
The primary difference lies in the relative energy ordering of the σ2pz and π2p molecular orbitals. For N2 (and other lighter period 2 diatomics), the σ2pz orbital is higher in energy than the π2p orbitals due to s-p mixing. For O2 and F2, the σ2pz orbital is lower in energy than the π2p orbitals. This difference impacts electron filling and, consequently, properties like bond order and magnetism.
What is the bond order of the peroxide ion (O22-) based on MO theory?
The peroxide ion (O22-) has 14 valence electrons (12 from two oxygen atoms + 2 for the 2- charge). Following the O2 energy level diagram, all bonding and antibonding orbitals up to the π2px and π2py orbitals are filled. This results in 8 bonding electrons and 6 antibonding electrons. Therefore, the bond order is (8 - 6) / 2 = 1. This indicates a single bond and no unpaired electrons, making it diamagnetic.
If you want to read more articles similar to Decoding O2: The Molecular Orbital Diagram Unveiled, you can visit the Automotive category.