31/08/2024
The fluorine molecule, F2, is a fascinating entity in the realm of chemistry, renowned for its incredible reactivity and a bond that, while strong, contributes to its distinctive behaviour. Yet, one of its most intriguing characteristics, often overlooked, is its diamagnetic nature. This property, where a substance is repelled by an external magnetic field, is not merely a curious anomaly but a direct consequence of its electronic structure, meticulously explained through the lens of Molecular Orbital Theory (MOT).
Understanding F2's diamagnetism requires us to delve beyond simple Lewis structures and embrace a more sophisticated model of chemical bonding. MOT provides a comprehensive picture, illustrating how atomic orbitals combine to form new molecular orbitals that span the entire molecule. It’s within this intricate dance of electrons that we uncover the fundamental reason why F2 exhibits no permanent magnetic moment, leading to its characteristic repulsion by magnetic fields.
- Demystifying Molecular Orbital Theory (MOT)
- The Molecular Orbital Diagram of F2 Explained
- The Crucial Detail: Why F2 is Diamagnetic
- Calculating the Bond Order of F2
- The Strong Bond and Reactivity of F2
- F2 Molecular Geometry
- Frequently Asked Questions About the F2 Molecule
- 1. What is the MOT diagram of F2 Class 12?
- 2. How does the MOT diagram of F2 Class 12 explain the bonding in fluorine?
- 3. What are the energy levels represented in the MOT diagram of F2 Class 12?
- 4. How does the MOT diagram of F2 Class 12 explain the stability of the molecule?
- 5. How many antibonding electrons are in F2?
- 6. What is the difference between diamagnetic and paramagnetic substances?
- 7. Is F2 a strong molecule?
- Conclusion
Demystifying Molecular Orbital Theory (MOT)
Molecular Orbital Theory (MOT) is a powerful quantum mechanical model that describes the electronic structure of molecules. Unlike Valence Bond Theory, which focuses on localised bonds between atoms, MOT proposes that atomic orbitals from individual atoms combine to form new, delocalised molecular orbitals that encompass the entire molecule. These molecular orbitals have specific energy levels and shapes, just like atomic orbitals.
When atomic orbitals combine, they can do so in two primary ways: constructively or destructively. Constructive interference leads to the formation of bonding molecular orbitals (MOs), which are lower in energy than the original atomic orbitals and concentrate electron density between the nuclei, thus stabilising the molecule. Destructive interference, conversely, leads to the formation of antibonding molecular orbitals (MOs), which are higher in energy and have a nodal plane between the nuclei, destabilising the molecule. Electrons preferentially fill the lower-energy bonding orbitals first, contributing to the molecule's stability.
The Molecular Orbital Diagram of F2 Explained
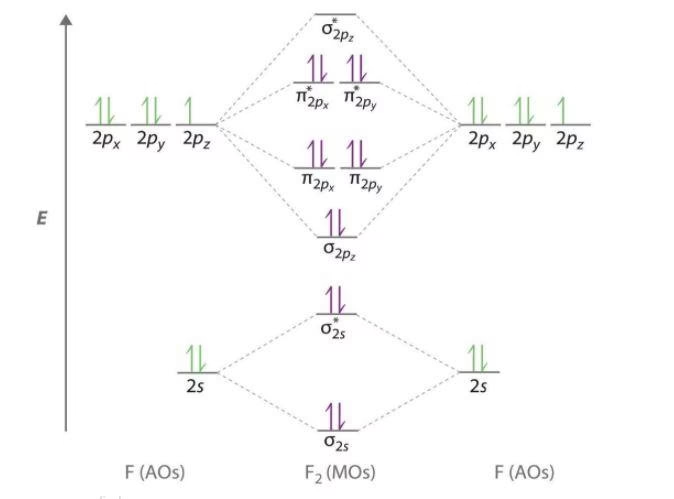
To fully grasp why F2 is diamagnetic, we must construct its molecular orbital (MO) diagram. Each fluorine atom (F) has an atomic number of 9, meaning it possesses 9 electrons. Its electron configuration is 1s² 2s² 2p⁵. When two fluorine atoms come together to form F2, they bring a total of 18 electrons.
We primarily focus on the valence electrons (those in the 2s and 2p orbitals) as these are the ones involved in bonding. The inner 1s orbitals also combine to form σ1s and σ*1s, but their electrons are tightly held and don't significantly influence the molecule's properties in the same way the valence electrons do.

Here's how the valence atomic orbitals of two fluorine atoms combine to form molecular orbitals:
- 2s Atomic Orbitals: The two 2s atomic orbitals combine to form a lower-energy bonding sigma (σ2s) molecular orbital and a higher-energy antibonding sigma (σ*2s) molecular orbital. Each F atom contributes two 2s electrons, so four electrons will fill these two MOs.
- 2p Atomic Orbitals: Each fluorine atom has three 2p atomic orbitals (2px, 2py, 2pz). These combine to form a more complex set of molecular orbitals:
- Sigma (σ2p) Molecular Orbital: Formed by the head-on overlap of the 2pz orbitals. This is a bonding orbital and is relatively low in energy.
- Pi (π2p) Molecular Orbitals: Formed by the side-on overlap of the 2px and 2py orbitals. There are two degenerate (equal energy) bonding pi (π2p) molecular orbitals.
- Pi-star (π*2p) Molecular Orbitals: Formed from the destructive overlap of the 2px and 2py orbitals. There are two degenerate (equal energy) antibonding pi (π*2p) molecular orbitals.
- Sigma-star (σ*2p) Molecular Orbital: Formed from the destructive head-on overlap of the 2pz orbitals. This is the highest energy antibonding orbital.
Now, let's fill these molecular orbitals with the 14 valence electrons (7 from each F atom):
- The four electrons from the 2s orbitals fill the σ2s and σ*2s orbitals (2 electrons in each).
- The remaining ten electrons (from the 2p orbitals) are filled according to the Aufbau principle, Hund's rule, and the Pauli exclusion principle:
- Two electrons go into the σ2p bonding orbital.
- Four electrons go into the two degenerate π2p bonding orbitals (two in each).
- The last four electrons go into the two degenerate π*2p antibonding orbitals (two in each).
- The highest energy antibonding orbital, σ*2p, remains empty.
The electron configuration for F2 in terms of molecular orbitals is: (σ1s)² (σ*1s)² (σ2s)² (σ*2s)² (σ2p)² (π2p)⁴ (π*2p)⁴.
The Crucial Detail: Why F2 is Diamagnetic
The key to F2's diamagnetic property lies in the complete pairing of all its electrons within its molecular orbitals. As we observed from the filling of the MO diagram, every single electron in F2 occupies an orbital alongside another electron with an opposite spin. There are simply no unpaired electrons anywhere in the molecule, not even in the higher-energy antibonding orbitals.
Substances are classified as diamagnetic when they possess no net magnetic moment. This is because the magnetic moments generated by paired electrons cancel each other out. When an external magnetic field is applied, diamagnetic substances are weakly repelled by it. This is in stark contrast to paramagnetic substances, which have one or more unpaired electrons, leading to a net magnetic moment and a weak attraction to an external magnetic field.
So, the answer is clear: F2 is diamagnetic because its molecular orbital diagram shows that all 18 electrons are paired up in their respective molecular orbitals, including the highest occupied molecular orbitals (HOMOs), which in F2's case are the π*2p antibonding orbitals.
Diamagnetic vs. Paramagnetic: A Quick Comparison
| Property | Diamagnetic Substances | Paramagnetic Substances |
|---|---|---|
| Electron Configuration | All electrons are paired | One or more unpaired electrons |
| Magnetic Moment | No permanent magnetic moment | Possess a permanent magnetic moment |
| Interaction with External Magnetic Field | Weakly repelled | Weakly attracted |
| Examples | F2, N2, H2O, NaCl | O2, NO, Transition metal ions (e.g., Fe³⁺) |
Calculating the Bond Order of F2
The bond order is a crucial concept derived from the molecular orbital diagram that indicates the number of chemical bonds between a pair of atoms. It's a measure of the stability and strength of the bond. The formula for bond order is:
Bond Order = ½ (Number of electrons in bonding MOs - Number of electrons in antibonding MOs)
Let's calculate the bond order for F2:
- Electrons in Bonding MOs:
- σ1s: 2
- σ2s: 2
- σ2p: 2
- π2p: 4
- Total Bonding Electrons = 2 + 2 + 2 + 4 = 10
- Electrons in Antibonding MOs:
- σ*1s: 2
- σ*2s: 2
- π*2p: 4
- σ*2p: 0 (empty)
- Total Antibonding Electrons = 2 + 2 + 4 = 8
Now, apply the formula:
Bond Order = ½ (10 - 8) = ½ (2) = 1
A bond order of 1 indicates a single covalent bond between the two fluorine atoms, which aligns with its Lewis structure. This positive bond order confirms that the F2 molecule is stable and can exist.
The Strong Bond and Reactivity of F2
While a bond order of 1 might seem to suggest a relatively weak bond compared to double or triple bonds, the F-F bond in F2 is actually quite strong in terms of its bond energy (around 159 kJ/mol). This strength is predicted by the presence of filled bonding orbitals, particularly the σ2p and π2p orbitals, which contribute significantly to the stability of the molecule. The electrons in these bonding orbitals are lower in energy than they would be in the isolated atoms, leading to a stable molecular structure.

However, despite its strong bond, F2 is famously known for being one of the most reactive elements. This high reactivity isn't contradictory to its bond strength but rather stems from other factors, primarily fluorine's extremely high electronegativity. Each fluorine atom has a strong desire to gain an electron to achieve a noble gas configuration. This drives F2 to readily break its relatively strong F-F bond to form even stronger bonds with other elements, making it an exceptionally potent oxidising agent.
F2 Molecular Geometry
The molecular geometry of F2 is quite straightforward. With only two atoms, it is inherently a linear molecule. It features a single F-F bond, as confirmed by its bond order of 1. While MOT primarily focuses on the electronic structure and energy levels, it indirectly supports this linear arrangement by describing the overlap of atomic orbitals along the internuclear axis to form sigma bonds.
Frequently Asked Questions About the F2 Molecule
1. What is the MOT diagram of F2 Class 12?
The MOT (Molecular Orbital Theory) diagram of F2, often studied in Class 12 Chemistry, graphically represents the energy levels and electron distribution within the F2 molecule. It illustrates how the atomic orbitals of two fluorine atoms combine to form bonding (lower energy) and antibonding (higher energy) molecular orbitals. For F2, this includes the formation and filling of σ2s, σ*2s, σ2p, π2p, π*2p, and σ*2p orbitals.
2. How does the MOT diagram of F2 Class 12 explain the bonding in fluorine?
In the MOT diagram of F2, the 2p atomic orbitals of each fluorine atom combine to form various molecular orbitals. The lower-energy bonding molecular orbitals (σ2p and π2p) are formed by constructive interference, leading to a stable bond between the fluorine atoms by concentrating electron density between the nuclei. The electrons preferentially occupy these bonding orbitals, thus stabilising the molecule. The higher-energy antibonding molecular orbitals (σ*2p and π*2p) are formed by destructive interference, and while some electrons fill the π*2p orbitals in F2, the overall energy reduction from the filled bonding orbitals ensures stability.
3. What are the energy levels represented in the MOT diagram of F2 Class 12?
The MOT diagram of F2 primarily represents several distinct energy levels for its molecular orbitals. From lowest to highest energy, these include: σ2s (bonding), σ*2s (antibonding), σ2p (bonding), π2p (bonding, degenerate), π*2p (antibonding, degenerate), and σ*2p (antibonding). The electrons fill these levels sequentially, starting from the lowest energy.

4. How does the MOT diagram of F2 Class 12 explain the stability of the molecule?
The MOT diagram of F2 explains its stability by demonstrating that the number of electrons occupying bonding molecular orbitals is greater than the number occupying antibonding molecular orbitals. This results in a positive bond order (1 in the case of F2). The filling of electrons into the lower-energy bonding orbitals signifies a net decrease in the molecule's overall energy compared to the isolated atoms, making the F2 molecule stable and able to exist.
5. How many antibonding electrons are in F2?
In the MOT diagram of the F2 molecule, there are 8 electrons occupying antibonding orbitals. These are distributed as follows: 2 electrons in σ*1s, 2 electrons in σ*2s, and 4 electrons in the degenerate π*2p orbitals.
6. What is the difference between diamagnetic and paramagnetic substances?
Diamagnetic substances have all their electrons paired within their orbitals, resulting in no net magnetic moment. They are weakly repelled by an external magnetic field. Paramagnetic substances, conversely, possess one or more unpaired electrons, which gives them a net magnetic moment, causing them to be weakly attracted to an external magnetic field.
7. Is F2 a strong molecule?
Yes, the F2 molecule has a relatively strong covalent bond, as indicated by its bond order of 1 and its bond energy. This strength comes from the effective overlap of atomic orbitals to form stable bonding molecular orbitals. However, its high reactivity is due to fluorine's electronegativity, making it eager to form even stronger bonds with other elements.
Conclusion
The F2 molecule, while deceptively simple in its diatomic form, holds a wealth of fascinating chemical properties explained through the sophisticated framework of Molecular Orbital Theory. Its diamagnetic nature, a direct consequence of having all its valence electrons perfectly paired within its molecular orbitals, serves as a prime example of how electronic configuration dictates macroscopic behaviour in the presence of magnetic fields. By understanding the intricate details of its MO diagram, from the combination of atomic orbitals to the precise filling of electrons, we gain profound insights into F2's stability, bond strength, and its remarkable place in the world of chemical reactivity.
If you want to read more articles similar to F2 Molecule: Unveiling Its Diamagnetic Secrets, you can visit the Automotive category.
