02/10/2018
The humble oxygen molecule, O2, is fundamental to life as we know it. We breathe it in, it fuels combustion, and it plays a crucial role in countless chemical reactions. Yet, beneath its ubiquitous presence lies a fascinating complexity, particularly when it comes to its electronic structure and magnetic behaviour. While a simple Lewis dot structure might suggest a straightforward arrangement of electrons, the reality, as explained by Molecular Orbital Theory (MOT), is far more intriguing and accurately predicts oxygen's observed magnetic properties.
- How Many Electrons Does an O2 Molecule Have?
- Understanding Molecular Orbital Theory (MOT)
- The Molecular Orbital Configuration of O2
- Bond Order of O2
- Magnetic Properties of O2: Paramagnetism vs. Diamagnetism
- The Misleading Nature of the Lewis Dot Structure
- Comparison: MOT vs. Lewis Structure for O2
- Frequently Asked Questions (FAQs)
- Conclusion
How Many Electrons Does an O2 Molecule Have?
To determine the total number of electrons in an O2 molecule, we first need to consider the atomic number of oxygen. Oxygen (O) has an atomic number of 8, meaning each oxygen atom possesses 8 protons and, in its neutral state, 8 electrons. Since an oxygen molecule, O2, is formed from two oxygen atoms bonded together, the total number of electrons in an O2 molecule is simply the sum of the electrons from each atom:
Total electrons in O2 = Electrons in atom 1 + Electrons in atom 2
Total electrons in O2 = 8 + 8 = 16 electrons
Therefore, an oxygen molecule contains a total of 16 electrons. These electrons are distributed among various atomic and molecular orbitals.
Understanding Molecular Orbital Theory (MOT)
Molecular Orbital Theory provides a more sophisticated understanding of chemical bonding than simpler models like Valence Bond Theory or Lewis structures. MOT postulates that when atoms combine to form a molecule, their atomic orbitals (AOs) merge to form new molecular orbitals (MOs) that are delocalised over the entire molecule. These molecular orbitals can be either bonding (lower in energy, stabilising the molecule) or antibonding (higher in energy, destabilising the molecule).
The formation of molecular orbitals follows specific rules, including the principle of linear combination of atomic orbitals (LCAO). For diatomic molecules like O2, the AOs of the constituent atoms combine to form sigma (σ) and pi (π) molecular orbitals. The relative energy levels of these MOs depend on factors such as the overlap between the AOs and the energy of the original AOs.
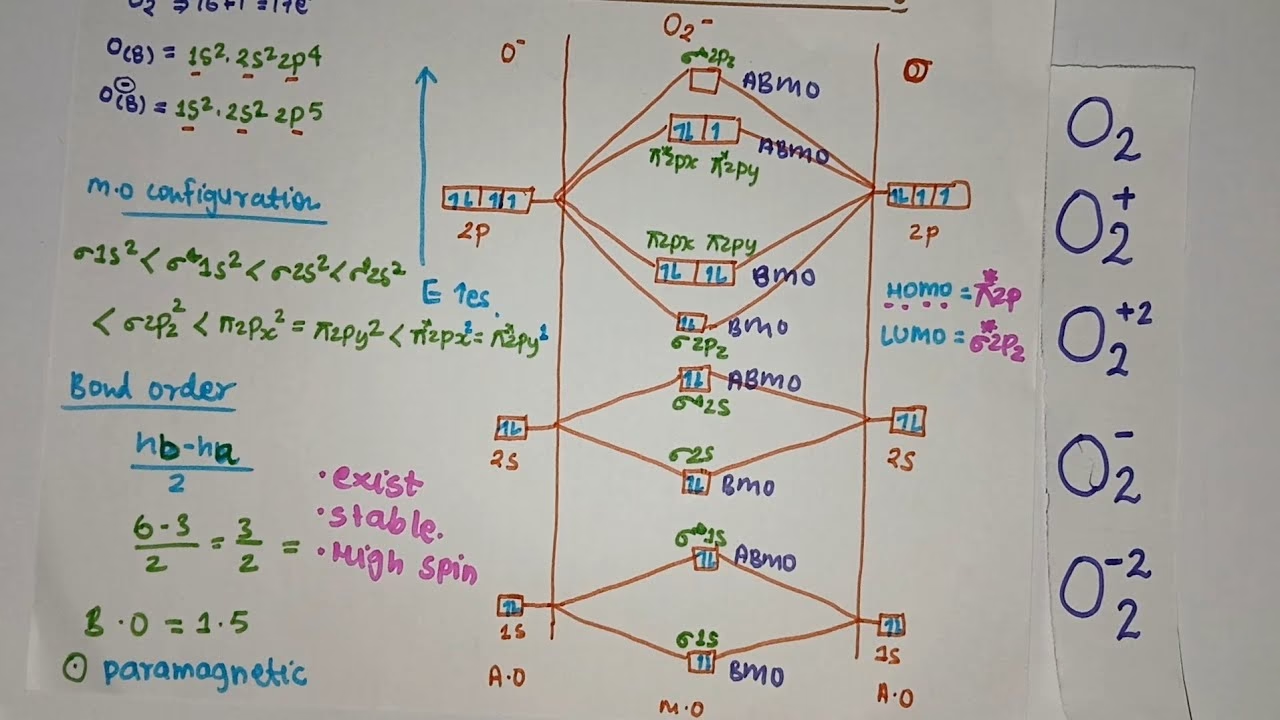
The Molecular Orbital Configuration of O2
The electronic configuration of a single oxygen atom is 1s2 2s2 2p4. When two oxygen atoms come together to form O2, their atomic orbitals combine to form molecular orbitals. The filling of these molecular orbitals follows the Aufbau principle, Hund's rule, and the Pauli exclusion principle. The molecular orbital configuration of O2, considering the 16 electrons, is as follows:
σ1s2, σ*1s2, σ2s2, σ*2s2, σ2pz2, π2px2, π2py2, π*2px1, π*2py1
Let's break down this configuration:
- σ1s2, σ*1s2: The 1s atomic orbitals from each oxygen atom combine to form a bonding σ1s MO and an antibonding σ*1s MO. Both are filled with 2 electrons each.
- σ2s2, σ*2s2: Similarly, the 2s atomic orbitals combine to form σ2s and σ*2s MOs, each filled with 2 electrons.
- σ2pz2: The 2pz atomic orbitals, which are directed along the internuclear axis, overlap head-on to form a sigma bonding MO, σ2pz, which is filled with 2 electrons.
- π2px2, π2py2: The 2px and 2py atomic orbitals, oriented perpendicular to the internuclear axis, overlap side-by-side to form two degenerate pi bonding MOs, π2px and π2py. Each of these is filled with 2 electrons, for a total of 4 electrons in these bonding orbitals.
- π*2px1, π*2py1: The remaining 4 electrons occupy the antibonding pi orbitals, π*2px and π*2py. According to Hund's rule, these electrons will occupy the degenerate orbitals singly before pairing up. Thus, one electron goes into π*2px and another into π*2py.
Bond Order of O2
The bond order of a molecule is a measure of the number of chemical bonds between two atoms. It can be calculated using the number of electrons in bonding and antibonding molecular orbitals:
Bond Order = ½ (Number of bonding electrons - Number of antibonding electrons)
From the molecular orbital configuration of O2:
- Bonding electrons: 2 (in σ1s) + 2 (in σ2s) + 2 (in σ2pz) + 4 (in π2px and π2py) = 10 electrons
- Antibonding electrons: 2 (in σ*1s) + 2 (in σ*2s) + 2 (in π*2px and π*2py) = 6 electrons
Using the formula:
Bond Order = ½ (10 - 6) = ½ (4) = 2
This indicates that there is a double bond between the two oxygen atoms in the O2 molecule, which aligns with experimental observations and the Lewis structure.
Magnetic Properties of O2: Paramagnetism vs. Diamagnetism
The magnetic property of a molecule is determined by the presence or absence of unpaired electrons in its molecular orbitals.
- Paramagnetic: Molecules with one or more unpaired electrons are attracted to a magnetic field.
- Diamagnetic: Molecules with all electrons paired are weakly repelled by a magnetic field.
Looking at the MOT diagram for O2, we observe that the π*2px and π*2py antibonding molecular orbitals each contain one unpaired electron. This is a crucial insight provided by MOT.
Crucially, the presence of these two unpaired electrons makes the oxygen molecule paramagnetic. This is why liquid oxygen is attracted to a magnet. This prediction is a significant success of Molecular Orbital Theory, as simpler models often fail to explain this property.

The Misleading Nature of the Lewis Dot Structure
The typical Lewis dot structure for O2 shows a double bond between the two oxygen atoms, with each oxygen atom having two lone pairs of electrons:
:Ö=Ö:
In this representation, all 16 valence electrons are shown as paired. If we were to rely solely on the Lewis structure, we would incorrectly conclude that O2 is diamagnetic. However, experimental evidence clearly demonstrates that O2 is paramagnetic. This discrepancy highlights the limitations of the Lewis structure model for accurately predicting magnetic properties and underscores the importance of Molecular Orbital Theory.
Comparison: MOT vs. Lewis Structure for O2
Here's a table summarising the differences in how MOT and Lewis structures describe the O2 molecule:
| Feature | Lewis Structure Prediction | Molecular Orbital Theory Prediction | Experimental Observation |
|---|---|---|---|
| Total Electrons | 16 (implied) | 16 | 16 |
| Bond Order | 2 (double bond) | 2 | 2 |
| Magnetic Property | Diamagnetic (all electrons paired) | Paramagnetic (two unpaired electrons in π* MOs) | Paramagnetic |
| Electron Pairing | All electrons paired | Unpaired electrons in π* antibonding orbitals | Confirms unpaired electrons |
Frequently Asked Questions (FAQs)
Q1: Why is oxygen paramagnetic?
Oxygen is paramagnetic because its molecular orbital configuration features two unpaired electrons in the degenerate π* antibonding orbitals. These unpaired electrons interact with external magnetic fields, causing the molecule to be attracted to the magnet.
Q2: Can the Lewis structure predict paramagnetism?
No, the standard Lewis structure for O2 does not predict paramagnetism. It incorrectly suggests all electrons are paired, leading to a prediction of diamagnetism. Molecular Orbital Theory is required to accurately predict the magnetic properties of O2.
Q3: How many bonding and antibonding electrons are in O2?
In the O2 molecule, there are 10 bonding electrons and 6 antibonding electrons according to MOT.
Q4: What is the bond order of O2?
The bond order of O2 is 2, indicating a double bond between the two oxygen atoms.
Q5: What is the electronic configuration of the oxygen atom?
The electronic configuration of a neutral oxygen atom is 1s2 2s2 2p4.
Conclusion
The oxygen molecule, O2, contains 16 electrons. While a simple Lewis structure might suggest a diamagnetic nature, Molecular Orbital Theory provides a more accurate and insightful description. The MOT configuration reveals that O2 possesses two unpaired electrons, rendering it paramagnetic – a property that has been experimentally verified. Understanding MOT is essential for truly grasping the electronic behaviour and bonding characteristics of molecules like oxygen, which are fundamental to chemistry and biology.
If you want to read more articles similar to Oxygen Molecule: Electrons and Magnetism, you can visit the Automotive category.
