20/12/2006
Xenon Difluoride, commonly known as XeF2, stands as a captivating example of noble gas chemistry, challenging earlier notions that these elements were entirely unreactive. Far from being inert, XeF2 is a remarkable compound, celebrated for its powerful fluorinating and oxidising capabilities. It's a white crystalline solid with a distinctive, somewhat nauseating odour, often found in laboratories and electrochemical procedures. Understanding its fundamental properties, from its intricate bonding mechanisms to its precise molecular geometry, is crucial for appreciating its chemical behaviour and applications. This article will delve deep into the various interpretations of XeF2, exploring its unique bonding, electronic configuration, and structural characteristics.
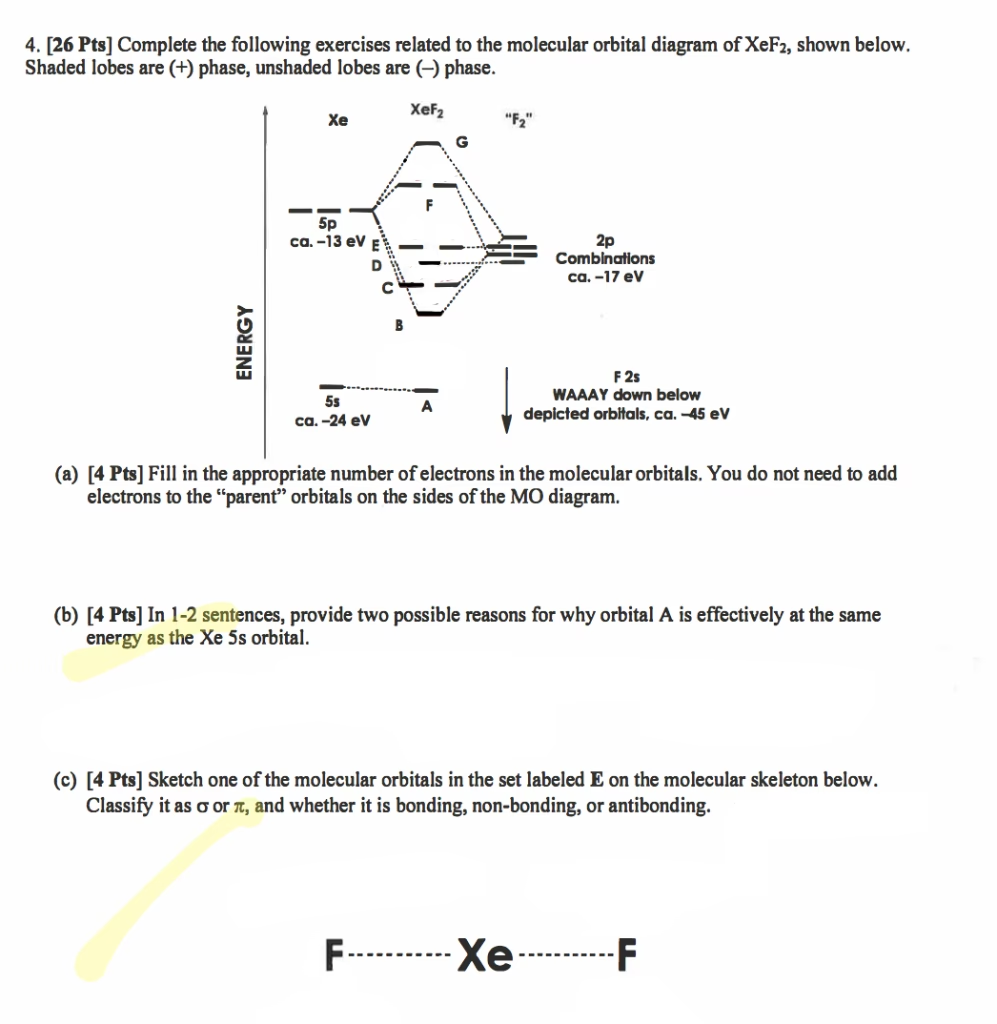
The Enigma of XeF2 Bonding: Three-Center Four-Electron Model
The bonding in linear XeF2 is elegantly interpreted through the lens of the three-center four-electron bond (3c-4e bond), a concept vital for understanding hypervalent molecules. This model posits that three atoms share four electrons in a delocalised molecular orbital system, which is particularly useful when traditional Lewis structures or simple valence bond theory fall short. In the case of XeF2, the central xenon atom and the two fluorine atoms participate in this unique bonding arrangement, contributing to the molecule's stability and linear form.
Molecular Orbital Perspective
From a molecular orbital (MO) perspective, the 3c-4e bond in XeF2 can be visualised as a linear combination of the fluorine 2p orbitals and the central xenon 5p orbital. This combination gives rise to three distinct molecular orbitals: one bonding, one non-bonding, and one anti-bonding. To determine the energies and electron distribution within these orbitals, a semi-empirical approach, such as the Hückel method, can be employed.
The Hückel matrix, a simplified representation of the Hamiltonian, helps us calculate the energies (eigenvalues) and the composition (eigenvectors) of these molecular orbitals. For XeF2, the matrix incorporates parameters like Coulomb integrals (α), which represent the energy of an electron in an isolated atomic orbital, and resonance integrals (β), which describe the interaction between adjacent orbitals. These parameters are often approximated using experimental data, such as the negative of valence orbital ionisation energies. For instance, the Xe 5p orbital's α is set to -12.13 eV, and the F 2p orbital's α is -17.42 eV, with a non-zero resonance integral (β) between Xe and F orbitals at -2.0 eV.
Solving this matrix yields three eigenvalues, corresponding to the energies of the molecular orbitals: E0 = -18.647 eV (bonding), E1 = -17.42 eV (non-bonding), and E2 = -10.903 eV (anti-bonding). The corresponding eigenvectors describe the contribution of each atomic orbital to the molecular orbital. The bonding MO shows significant contributions from all three atoms, the non-bonding MO primarily from the fluorine atoms with zero contribution from xenon, and the anti-bonding MO from the xenon atom with out-of-phase contributions from fluorine.
Crucially, the stability of the XeF2 molecule can be assessed by comparing its total electronic energy with that of its isolated atomic components. With two electrons occupying the bonding MO and two in the non-bonding MO, the total energy for XeF2 is calculated to be -72.14 eV. In contrast, the energy of the isolated atoms (two fluorine and one xenon) is 2(-17.42 eV) + 1(-12.13 eV) = -46.97 eV, or if considering the electrons specifically involved in the 3c-4e bond, it's 2(-12.13 eV) + 2(-17.42 eV) = -59.1 eV. Regardless of the exact baseline, the molecular energy of -72.14 eV is significantly lower, indicating that XeF2 is indeed more stable than its isolated atomic constituents, according to this semi-empirical model.
Furthermore, this model allows for the calculation of partial charges on each atom. By crediting each atom with its non-bonding electrons and its share of bonding and non-bonding MO electrons, we can deduce the charge distribution. The fluorine atoms, for example, end up with a partial charge of approximately -0.842, while the xenon atom carries a partial charge of about +1.684. This large positive charge on xenon is entirely reasonable, given that it is bonded to fluorine, the most electronegative element in the periodic table, which pulls electron density strongly towards itself.
Valence Bond Representation: A Resonance Hybrid
While the molecular orbital theory provides a detailed electronic picture, the valence bond theory offers an alternative perspective, particularly useful for understanding the distribution of electrons in terms of resonance structures. For XeF2, its electronic properties can be represented as a resonance hybrid of two equivalent structures:
F- - Xe+ - F- ↔ F- - Xe+ - F-
These structures depict a formal positive charge on the central xenon atom and negative charges on the fluorine atoms, reflecting the highly electronegative nature of fluorine. This resonance contributes to the overall stability of the molecule. The ground electronic state configuration of XeF2 is quite complex, represented by a series of molecular orbitals: (8σg)2 (5σu)2 (9σg)2 (6σu)2 (4πu)4 (3πg)4 (10σg)2 (5πu)4 (7σu)0, illustrating the intricate arrangement of its 22 valence electrons across various bonding and non-bonding orbitals.
Unpacking the Molecular Geometry of XeF2
Xenon Difluoride, or XeF2, is not just a theoretical curiosity; it's a tangible compound with specific physical and chemical properties. It’s a powerful fluorinating and oxidising agent, more stable than other xenon fluorides like XeF4 and XeF6. As a white crystalline solid, it finds use in various electrochemical procedures and laboratory settings. Its distinctive nauseating odour is a clear warning sign, and it's known to decompose upon contact with light or water vapour. To truly grasp its chemical behaviour, a detailed understanding of its Lewis structure, hybridisation, and molecular geometry is paramount.
Decoding the Lewis Structure of XeF2
The Lewis structure of a chemical compound is a foundational tool for visualising the arrangement of valence electrons and how they participate in bonding. It helps in understanding formal charges and the distribution of electrons, distinguishing between bonding pairs (shared electrons, represented as lines) and lone pairs (unshared electrons, represented as dots). The octet rule, which states that atoms tend to achieve eight electrons in their outermost shell for stability, guides the construction of these structures.
For XeF2, we consider one xenon atom and two fluorine atoms. Xenon, being a noble gas, has eight valence electrons. Each fluorine atom has seven valence electrons. Therefore, the total number of valence electrons for XeF2 is calculated as:
Total valence electrons = (Valence electrons for Xenon) + (Valence electrons for Fluorine × 2)
= 8 + (7 × 2)
= 8 + 14
= 22 valence electrons
In the Lewis structure, Xenon will be the central atom, bonded to two fluorine atoms. After forming two single bonds (4 electrons), the remaining 18 electrons are distributed as lone pairs. Each fluorine atom receives three lone pairs (6 electrons each, total 12), fulfilling its octet. The remaining 6 electrons are placed on the central xenon atom as three lone pairs. This results in xenon being surrounded by two bonding pairs and three lone pairs.
XeF2 Hybridisation: The sp3d Story
Hybridisation is a crucial concept for understanding the geometry of molecules. It describes the mixing of atomic orbitals to form new hybrid orbitals with different shapes and energies, which are then used for bonding. For XeF2, the central xenon atom's hybridisation plays a key role.
In its ground state, Xenon has an electron configuration of [Kr] 4d10 5s2 5p6, with all valence electrons paired. However, for Xe to form bonds, especially with highly electronegative elements like fluorine, it must undergo excitation. In the case of XeF2, one of the 5p electrons is promoted to an empty 5d orbital, leading to an excited state configuration of 5s2 5p5 5d1, with two unpaired electrons. These two unpaired electrons can then form bonds with the two fluorine atoms.
To accommodate the two bonding pairs and the three lone pairs, the central xenon atom undergoes sp3d hybridisation. This involves the mixing of one 5s orbital, three 5p orbitals, and one 5d orbital, resulting in five equivalent sp3d hybrid orbitals. These hybrid orbitals are then oriented in a trigonal bipyramidal electron geometry around the central xenon atom.
VSEPR Theory and the Linear Shape
While hybridisation suggests a trigonal bipyramidal electron geometry, the actual molecular geometry of XeF2, which refers to the arrangement of only the atoms, is different. This is where the VSEPR theory (Valence Shell Electron Pair Repulsion theory) becomes indispensable. VSEPR theory states that electron pairs, both bonding and lone pairs, around a central atom will arrange themselves as far apart as possible to minimise repulsion.
For XeF2, the central xenon atom has a steric number of 5 (two bonding pairs with fluorine and three lone pairs). According to VSEPR theory, five electron domains arrange themselves in a trigonal bipyramidal electron geometry. In this geometry, there are two types of positions: axial and equatorial. Lone pairs typically occupy equatorial positions to minimise repulsion with other electron pairs, as these positions offer more space.
In XeF2, the three lone pairs occupy the three equatorial positions of the trigonal bipyramid. This leaves the two bonding pairs (Xe-F bonds) to occupy the two axial positions. The result of this specific arrangement, with the three lone pairs effectively pushing the two fluorine atoms into a straight line, is a linear geometry for the XeF2 molecule. Despite the underlying trigonal bipyramidal electron geometry, the molecular shape, defined by the positions of the atoms, is distinctly linear.
Bond Angle of XeF2
Given the linear geometry of Xenon Difluoride, the bond angle between the two fluorine atoms and the central xenon atom (F-Xe-F) is straightforward. With the two fluorine atoms positioned directly opposite each other along an axis, the bond angle is precisely 180 degrees. This perfect linearity is a direct consequence of the symmetrical placement of the lone pairs in the equatorial plane, forcing the bonded atoms into an axial arrangement.
Polarity of XeF2: A Nonpolar Molecule
The polarity of a molecule is determined by its molecular geometry and the electronegativity differences between its constituent atoms. While fluorine is significantly more electronegative than xenon, creating polar Xe-F bonds, the overall molecule of XeF2 is nonpolar. This apparent contradiction is resolved by considering the molecule's symmetrical structure.
In XeF2, the two fluorine atoms are arranged symmetrically on opposite sides of the central xenon atom. Each Xe-F bond has a dipole moment pointing towards the more electronegative fluorine atom. However, because these two bond dipoles are equal in magnitude and point in exactly opposite directions along the linear axis, they effectively cancel each other out. As a result, there is no net dipole moment across the entire molecule, rendering XeF2 a nonpolar compound despite its polar bonds.
Key Properties of XeF2 at a Glance
| Property | Description |
|---|---|
| Total Valence Electrons | 22 |
| Bonding Pairs | 2 |
| Lone Pairs on Central Xe | 3 |
| Electron Geometry (VSEPR) | Trigonal Bipyramidal |
| Hybridisation of Xe | sp3d |
| Molecular Geometry | Linear |
| Bond Angle (F-Xe-F) | 180 degrees |
| Polarity | Nonpolar |
| Bonding Model | Three-Center Four-Electron (3c-4e) |
Frequently Asked Questions About XeF2
- Is XeF2 a stable molecule?
Yes, XeF2 is surprisingly stable for a noble gas compound. Theoretical calculations and experimental observations confirm its stability, making it the most stable of the known xenon fluorides. - Why is XeF2 linear and not trigonal bipyramidal?
While the electron geometry around the central xenon atom is trigonal bipyramidal (due to five electron domains: two bonding pairs and three lone pairs), the molecular geometry considers only the arrangement of atoms. The three lone pairs occupy the equatorial positions, pushing the two fluorine atoms into the axial positions, resulting in a linear molecular shape. - How many lone pairs are on the central xenon atom in XeF2?
There are three lone pairs of electrons on the central xenon atom in XeF2. - Why is XeF2 considered a hypervalent molecule?
XeF2 is considered hypervalent because the central xenon atom appears to have more than eight electrons in its valence shell (two bonding pairs + three lone pairs = 10 electrons), exceeding the traditional octet rule. This is explained by the involvement of d-orbitals in its hybridisation and the 3c-4e bonding model. - What are the main uses of XeF2?
XeF2 is a powerful fluorinating and oxidising agent, primarily used in chemical synthesis for adding fluorine atoms to organic and inorganic compounds. It's also employed in etching processes in microelectronics.
Concluding Thoughts
In summary, Xenon Difluoride is a truly remarkable compound that defies the once-held belief in the complete inertness of noble gases. Its intricate bonding can be elucidated through the three-center four-electron bond model, further detailed by molecular orbital theory calculations that confirm its stability and reveal partial charges. The molecule's valence bond representation as a resonance hybrid provides an additional layer of understanding its electronic structure. From a structural perspective, XeF2 possesses 22 valence electrons, undergoes sp3d hybridisation, and, most notably, adopts a linear geometry with a 180-degree bond angle, as explained by the VSEPR theory. This highly symmetrical arrangement of atoms and electron pairs ultimately results in XeF2 being a nonpolar molecule, despite the inherent polarity of its Xe-F bonds. These combined insights paint a comprehensive picture of Xenon Difluoride, highlighting its unique place in chemistry and its continued relevance in various scientific applications.
If you want to read more articles similar to Unveiling Xenon Difluoride: Structure and Stability, you can visit the Automotive category.
