30/08/2009
The air we breathe, an invisible yet vital cocktail, is predominantly composed of nitrogen gas (N2). Despite its abundance, nitrogen is remarkably unreactive, a characteristic crucial for life on Earth. But what makes this seemingly simple diatomic molecule so stable, so inert? The answer lies deep within its atomic structure, specifically in the intricate dance of its electrons, beautifully visualised through the N2 orbital diagram. Understanding this diagram isn't just an academic exercise; it's a window into the fundamental forces that govern chemical bonding and the properties of matter around us.
An orbital diagram, at its core, is a pictorial representation of the electron configuration within an atom or a molecule. It illustrates how electrons are distributed among different energy levels and orbitals, providing critical insights into the molecule's stability, reactivity, and magnetic properties. For the nitrogen molecule, the N2 orbital diagram helps us comprehend the robust bonding that makes it such a resilient chemical species. Let's embark on a journey to unravel the mysteries of N2's electronic architecture.
- The Building Blocks: Atomic Nitrogen
- From Atoms to Molecules: The LCAO Approach
- Constructing the N2 Molecular Orbital Diagram
- Populating the Orbitals: Electron Configuration of N2
- The Strength of the Bond: Bond Order Calculation
- Why N2 is So Stable and Inert
- N2 vs. O2: A Comparative Look
- Frequently Asked Questions
- What is the primary purpose of an N2 orbital diagram?
- Why is the sigma bonding molecular orbital lower in energy than the sigma antibonding molecular orbital?
- How many sigma and pi bonds are in an N2 molecule?
- Are there any antibonding electrons in N2?
- How does the N2 orbital diagram explain its physical properties, like its gaseous state?
The Building Blocks: Atomic Nitrogen
Before we delve into the complexities of the nitrogen molecule, it's essential to understand the individual nitrogen atom. Nitrogen (N) is element number seven on the periodic table, meaning a neutral nitrogen atom has seven electrons. Its electron configuration, following the Aufbau principle, Hund's rule, and the Pauli exclusion principle, is 1s² 2s² 2p³. This configuration tells us that nitrogen has two core electrons in the 1s orbital and five valence electrons in its outermost shell (2s² 2p³). These five valence electrons are the key players in chemical bonding, as they are the ones involved in forming connections with other atoms.
The atomic orbitals involved in bonding for nitrogen are the 2s and 2p orbitals. The 2s orbital is spherical, while the three 2p orbitals (2px, 2py, 2pz) are dumbbell-shaped and oriented along the x, y, and z axes, respectively. When two nitrogen atoms approach each other to form a molecule, their atomic orbitals interact and combine, giving rise to an entirely new set of orbitals known as molecular orbitals.
From Atoms to Molecules: The LCAO Approach
The formation of molecular orbitals from atomic orbitals is explained by the Linear Combination of Atomic Orbitals (LCAO) theory. According to LCAO, when two atomic orbitals overlap, they combine to form two new molecular orbitals: a bonding molecular orbital and an antibonding molecular orbital. The bonding molecular orbital is lower in energy than the original atomic orbitals, leading to increased stability when electrons occupy it. Conversely, the antibonding molecular orbital is higher in energy, and electrons occupying it destabilise the molecule.
The combination of atomic orbitals can occur in two primary ways:
- Sigma (σ) Bonds: Formed by the direct, head-on overlap of atomic orbitals (e.g., s-s overlap, s-p overlap, or p-p head-on overlap). Electron density is concentrated along the internuclear axis.
- Pi (π) Bonds: Formed by the sideways overlap of p orbitals. Electron density is concentrated above and below the internuclear axis.
The number of molecular orbitals formed is always equal to the number of atomic orbitals that combine. Since each nitrogen atom contributes 1s, 2s, and three 2p orbitals, a total of ten atomic orbitals (five from each N atom) will combine to form ten molecular orbitals in the N2 molecule. However, for bonding, we primarily focus on the valence orbitals.
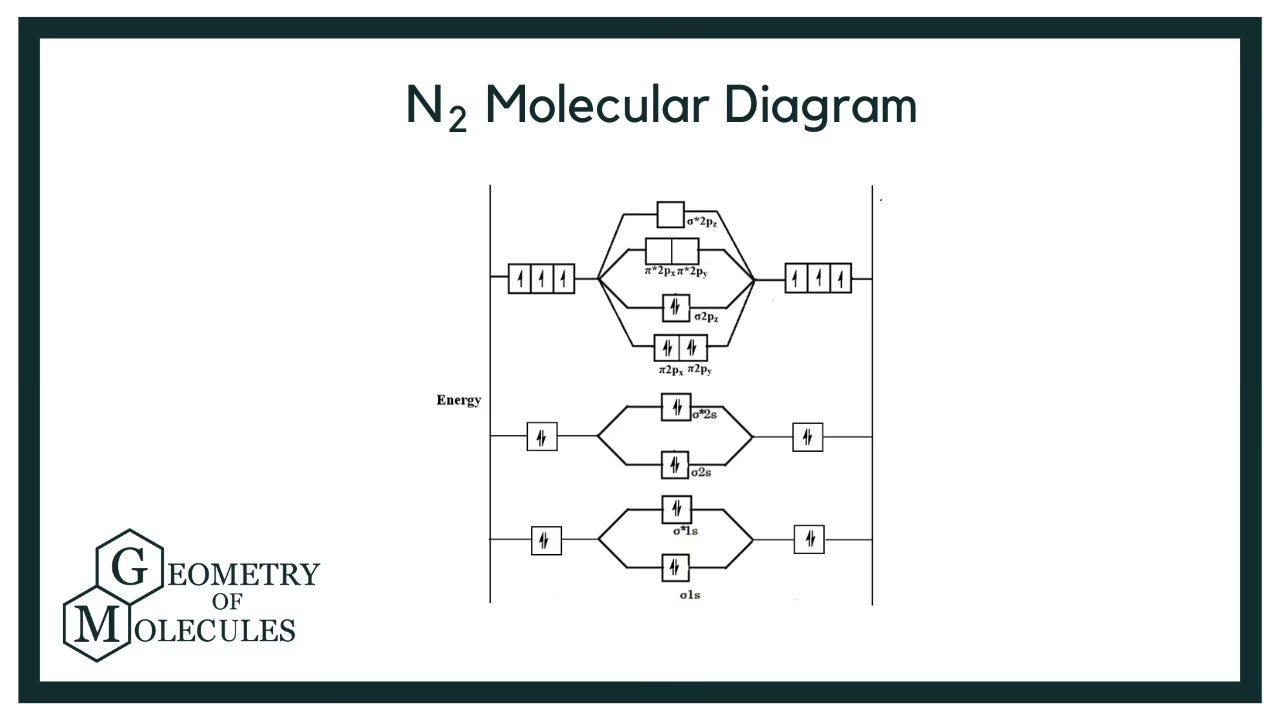
Constructing the N2 Molecular Orbital Diagram
Let's construct the molecular orbital diagram for N2 by considering the interaction of the valence 2s and 2p atomic orbitals from each nitrogen atom. Each nitrogen atom brings five valence electrons to the table, making a total of ten valence electrons for the N2 molecule.
Interaction of 2s Atomic Orbitals
When the 2s atomic orbitals from the two nitrogen atoms overlap, they form two molecular orbitals:
- Sigma (σ2s) Bonding Molecular Orbital: This orbital is formed by the constructive interference of the 2s orbitals. It is lower in energy than the original 2s atomic orbitals and contributes to the stability of the molecule.
- Sigma Anti-bonding (σ*2s) Molecular Orbital: This orbital is formed by the destructive interference of the 2s orbitals. It is higher in energy than the original 2s atomic orbitals and, if occupied by electrons, would destabilise the molecule.
Interaction of 2p Atomic Orbitals
The three 2p atomic orbitals from each nitrogen atom interact in a more complex manner. We can designate one set of 2p orbitals (e.g., 2pz) to undergo head-on overlap, forming sigma bonds, while the other two sets (2px and 2py) undergo sideways overlap, forming pi bonds.
- Sigma (σ2p) Bonding Molecular Orbital: This is formed by the head-on overlap of the 2pz orbitals. As stated in the initial information, this sigma bonding molecular orbital is indeed lower in energy compared to its corresponding sigma antibonding molecular orbital, indicating the formation of a stable sigma bond between the two nitrogen atoms.
- Sigma Anti-bonding (σ*2p) Molecular Orbital: Formed by the destructive head-on overlap of the 2pz orbitals. It is higher in energy than the σ2p orbital.
- Pi (π2p) Bonding Molecular Orbitals: These are formed by the sideways overlap of the 2px and 2py orbitals. There are two such orbitals (π2px and π2py), and they are degenerate, meaning they have the same energy level. They are lower in energy than the original 2p atomic orbitals.
- Pi Anti-bonding (π*2p) Molecular Orbitals: These are formed by the destructive sideways overlap of the 2px and 2py orbitals. There are two such orbitals (π*2px and π*2py), and they are degenerate. They are higher in energy than the π2p orbitals.
Energy Level Ordering in N2
The specific order of filling molecular orbitals is crucial. For N2, due to significant s-p mixing, the energy order of the molecular orbitals derived from the 2p atomic orbitals is slightly different from heavier diatomic molecules like O2 or F2. The correct ascending energy order for N2's valence molecular orbitals is:
σ2s < σ*2s < π2p (degenerate) < σ2p < π*2p (degenerate) < σ*2p
This means the pi bonding orbitals (π2p) are slightly lower in energy than the sigma bonding orbital (σ2p) derived from the 2p atomic orbitals. This is a characteristic feature for N2 and other light diatomic molecules (Li2 to N2).
Populating the Orbitals: Electron Configuration of N2
Now that we have our molecular orbitals arranged by energy, we can fill them with the ten total valence electrons from the two nitrogen atoms. We follow Hund's rule (fill degenerate orbitals singly first before pairing) and the Pauli exclusion principle (maximum two electrons per orbital with opposite spins):
- The σ2s orbital receives two electrons.
- The σ*2s orbital receives two electrons.
- The two degenerate π2p orbitals receive four electrons (two in each, paired up).
- The σ2p orbital receives two electrons.
This gives us the molecular orbital electron configuration for N2:
(σ2s)² (σ*2s)² (π2p)⁴ (σ2p)²
All ten valence electrons are accounted for, and all are paired. This pairing of electrons means that N2 is diamagnetic (it is not attracted to a magnetic field).
The Strength of the Bond: Bond Order Calculation
One of the most powerful insights derived from the molecular orbital diagram is the bond order. Bond order is a measure of the number of chemical bonds between two atoms and is calculated using the following formula:
Bond Order = (Number of electrons in bonding molecular orbitals - Number of electrons in antibonding molecular orbitals) / 2
Let's calculate the bond order for N2:
- Bonding electrons: 2 (from σ2s) + 4 (from π2p) + 2 (from σ2p) = 8 electrons
- Antibonding electrons: 2 (from σ*2s) = 2 electrons
Bond Order = (8 - 2) / 2 = 6 / 2 = 3
A bond order of 3 indicates that there is a triple bond between the two nitrogen atoms in the N2 molecule. This triple bond consists of one sigma bond and two pi bonds, a structure that aligns perfectly with the Lewis structure of N≡N. This high bond order is a direct explanation for the exceptional stability of the nitrogen molecule.

Why N2 is So Stable and Inert
The N2 orbital diagram provides a clear explanation for the well-known inertness and high stability of nitrogen gas:
- High Bond Order: A bond order of 3 signifies a very strong triple bond, requiring a significant amount of energy to break. This makes N2 chemically unreactive under normal conditions.
- All Bonding Orbitals Filled: All the lower-energy bonding molecular orbitals (σ2s, π2p, σ2p) are fully occupied by electrons, contributing maximum stability.
- Large HOMO-LUMO Gap: The Highest Occupied Molecular Orbital (HOMO) is the σ2p orbital, and the Lowest Unoccupied Molecular Orbital (LUMO) is the π*2p orbital. There is a substantial energy gap between the HOMO and LUMO. This large energy difference means it requires considerable energy to excite electrons into higher energy levels or to accept electrons into the LUMO, further contributing to N2's low reactivity.
- Diamagnetic Nature: All electrons are paired, meaning N2 has no unpaired electrons to readily participate in reactions, unlike paramagnetic molecules such as O2.
These factors collectively explain why N2 constitutes roughly 78% of Earth's atmosphere yet participates in relatively few chemical reactions unless subjected to extreme conditions, such as high temperatures or pressures, or the presence of specific catalysts.
N2 vs. O2: A Comparative Look
To further appreciate the unique features of the N2 molecular orbital diagram, a brief comparison with oxygen (O2) can be insightful. While both are diatomic molecules, their MO diagrams and properties differ significantly, primarily due to the different number of valence electrons and the extent of s-p mixing.
| Feature | Nitrogen (N2) | Oxygen (O2) |
|---|---|---|
| Valence Electrons | 10 | 12 |
| MO Energy Order (2p derived) | π2p < σ2p | σ2p < π2p |
| MO Configuration | (σ2s)² (σ*2s)² (π2p)⁴ (σ2p)² | (σ2s)² (σ*2s)² (σ2p)² (π2p)⁴ (π*2p)² |
| Bonding Electrons | 8 | 8 |
| Antibonding Electrons | 2 | 4 |
| Bond Order | 3 (Triple Bond) | 2 (Double Bond) |
| Magnetic Property | Diamagnetic | Paramagnetic |
| Stability/Reactivity | Very stable, inert | Reactive, supports combustion |
This table clearly highlights how the subtle differences in orbital interactions and electron count lead to vastly different chemical properties. The higher bond order and diamagnetic nature of N2 stand in stark contrast to O2's double bond and paramagnetism, illustrating the predictive power of molecular orbital theory.
Frequently Asked Questions
What is the primary purpose of an N2 orbital diagram?
The primary purpose of an N2 orbital diagram is to illustrate the distribution of electrons in the nitrogen molecule's molecular orbitals, providing a visual representation of its electron configuration, the nature of its chemical bonds, and ultimately explaining its stability and chemical properties.
Why is the sigma bonding molecular orbital lower in energy than the sigma antibonding molecular orbital?
Bonding molecular orbitals, including the sigma bonding orbital, are formed by the constructive overlap of atomic orbitals. This constructive interference leads to an increased electron density between the nuclei, which is a lower energy, more stable arrangement. Conversely, antibonding orbitals result from destructive overlap, leading to a node (zero electron density) between nuclei, which is a higher energy, less stable arrangement.
How many sigma and pi bonds are in an N2 molecule?
Based on its molecular orbital diagram and bond order of 3, the N2 molecule contains one sigma (σ) bond and two pi (π) bonds. This combination forms the characteristic triple bond between the two nitrogen atoms.
Are there any antibonding electrons in N2?
Yes, there are antibonding electrons in N2. Specifically, the σ*2s molecular orbital is occupied by two electrons. While these electrons do reduce the overall bond order, the number of bonding electrons significantly outweighs the antibonding electrons, leading to a net strong bond.
How does the N2 orbital diagram explain its physical properties, like its gaseous state?
The strong triple bond within the N2 molecule means that individual N2 molecules are very stable. However, the forces *between* N2 molecules (intermolecular forces) are very weak London dispersion forces. These weak intermolecular forces mean that relatively little energy is required to overcome them, allowing N2 molecules to move freely and independently at typical atmospheric temperatures, hence its gaseous state.
In conclusion, the N2 orbital diagram is more than just a theoretical concept; it's a powerful tool that demystifies the incredible stability and inertness of one of Earth's most abundant gases. By meticulously mapping out the energy levels and electron distributions, we gain a profound appreciation for the elegant principles of quantum mechanics that govern the very fabric of our world.
If you want to read more articles similar to Unravelling N2's Electron Dance: A Deep Dive, you can visit the Automotive category.
