21/10/2019
Exploring the Reactivity of Dimanganese Decacarbonyl: Protonation and Methylation
Dimanganese decacarbonyl, with the chemical formula Mn₂(CO)₁₀, stands as a cornerstone in the organometallic chemistry of manganese. This fascinating metal carbonyl, often represented as (CO)₅Mn-Mn(CO)₅, is notable for its structure featuring two manganese atoms directly bonded, each adorned with five carbonyl ligands. While it exists as a stable crystalline solid, its solutions necessitate careful handling under inert atmospheres, employing techniques such as Schlenk lines. Its utility in organic synthesis, though limited, is underpinned by its characteristic reactivity, particularly its response to reduction, halogenation, and crucially, protonation and methylation. This article delves into the specific differences between these two important reactions, illuminating the distinct pathways and resulting species.
The Genesis of Dimanganese Decacarbonyl
The initial preparation of Mn₂(CO)₁₀, reported in low yield, involved the reduction of manganese iodide using magnesium under a carbon monoxide atmosphere. Over time, more efficient synthetic routes have been developed. A significant advancement came with the reduction of anhydrous MnCl₂ using sodium benzophenone ketyl under high pressures of CO (200 atmospheres). More recently, the availability of inexpensive Methylcyclopentadienyl manganese tricarbonyl (MMT) has paved the way for a lower-pressure synthetic route, making this vital reagent more accessible.
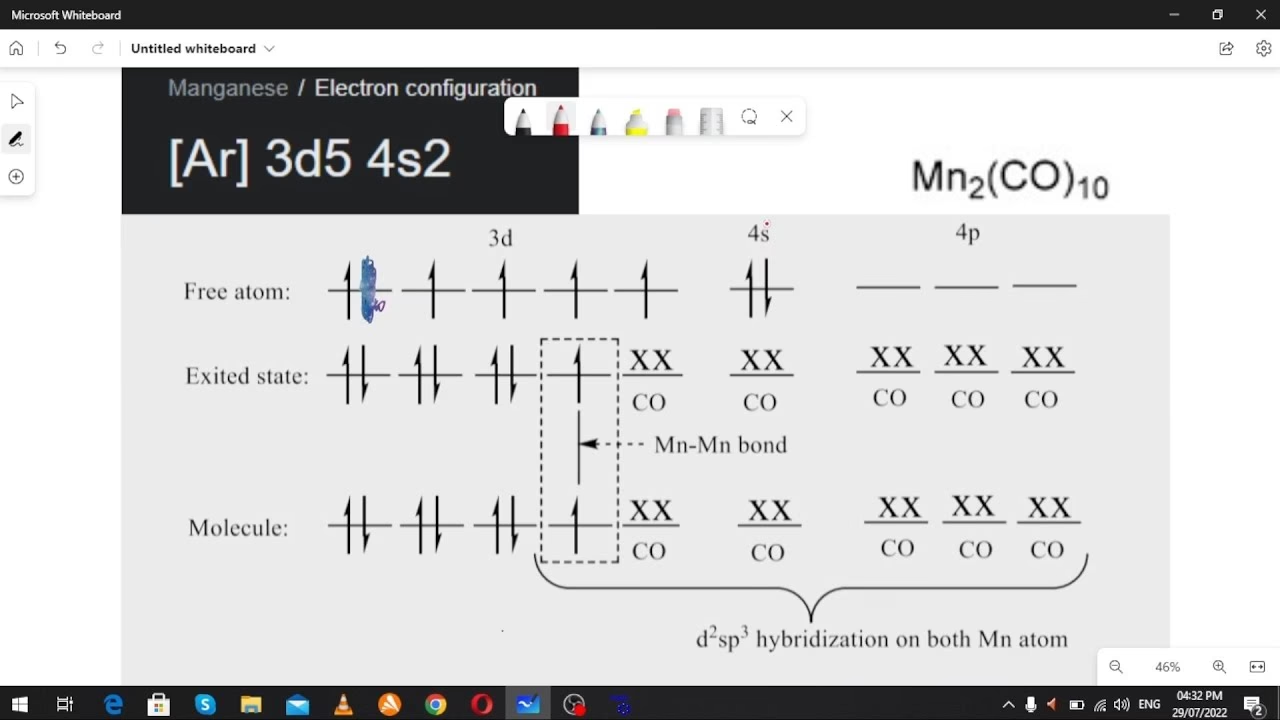
Structural Insights: A Unique D₄d Symmetry
The structure of Mn₂(CO)₁₀ is characterised by the absence of bridging carbonyl ligands, adopting a linear Mn-Mn bond. Each manganese atom is bonded to five carbonyl groups. Within this arrangement, there are two distinct types of CO ligands. One CO ligand on each manganese atom is positioned coaxially with the Mn-Mn bond, while the remaining four are situated perpendicularly, often referred to as equatorial. The most stable conformation, or rotamer, sees the two Mn(CO)₅ subunits staggered, resulting in an overall molecular symmetry belonging to the D₄d point group. This specific symmetry is relatively uncommon among metal carbonyls, adding to the compound's unique character.
Reactions: Unpacking the Transformations
The reactivity of Mn₂(CO)₁₀ is central to its role in organometallic chemistry. Its stability as a solid contrasts with the air sensitivity of its solutions. Key reactions include:
- Reduction: Treatment with reducing agents, such as alkali metals, cleaves the Mn-Mn bond, yielding the manganese pentacarbonyl anion, [Mn(CO)₅]⁻. This anion is a highly versatile species, often isolated as a salt with a suitable counterion, for example, sodium ([Na][Mn(CO)₅]).
- Halogenation: Halogenation reactions, such as with bromine (Br₂), also lead to the cleavage of the Mn-Mn bond, producing halopentacarbonyl manganese species, like [Mn(CO)₅Br].
The Crucial Distinction: Protonation vs. Methylation
The focus of this discussion lies in the contrasting outcomes of protonation and methylation when applied to the manganese pentacarbonyl anion, [Mn(CO)₅]⁻, which is readily generated from Mn₂(CO)₁₀. Both reactions involve the addition of an electrophile to the nucleophilic manganese centre of the anion, but the nature of the electrophile dictates the final product.
Protonation: The Formation of a Hydride
Protonation involves the reaction of the manganese pentacarbonyl anion with a protic acid or a source of protons. The highly nucleophilic manganese atom readily attacks the proton. This process results in the formation of a manganese-hydrogen bond, yielding the neutral manganese pentacarbonyl hydride, [HMn(CO)₅].
The reaction can be represented as:
[Mn(CO)₅]⁻ + H⁺ → HMn(CO)₅
The resulting hydride, HMn(CO)₅, is a significant organometallic compound in its own right. It serves as a precursor for other manganese-containing species and is involved in various catalytic cycles. The addition of a proton effectively neutralises the negative charge on the manganese centre.
Methylation: Introducing a Methyl Group
Methylation, on the other hand, involves the reaction of the manganese pentacarbonyl anion with a methylating agent, typically an alkyl halide or a similar electrophilic source of a methyl group. Again, the nucleophilic manganese atom attacks the electrophilic carbon atom of the methyl group.
A common methylating agent is methyl iodide (CH₃I). The reaction proceeds as follows:
[Mn(CO)₅]⁻ + CH₃X → CH₃Mn(CO)₅ + X⁻
(Where X is a leaving group, e.g., I)
This reaction results in the formation of methylmanganese pentacarbonyl, CH₃Mn(CO)₅. In this product, a direct bond exists between the manganese atom and the carbon atom of the methyl group. This represents an organometallic compound with a metal-carbon sigma bond, a common feature in many organometallic complexes.
Key Differences Summarised
The fundamental difference between protonation and methylation of the manganese pentacarbonyl anion lies in the nature of the atom that becomes directly bonded to the manganese centre:
| Reaction | Electrophile | Bond Formed to Mn | Product | Charge of Product |
|---|---|---|---|---|
| Protonation | Proton (H⁺) | Hydrogen (H) | HMn(CO)₅ | Neutral |
| Methylation | Methyl group (CH₃⁺) | Carbon (C) | CH₃Mn(CO)₅ | Neutral |
While both reactions start from the same anionic precursor, [Mn(CO)₅]⁻, and result in neutral products, the chemical identity of these products is distinct. HMn(CO)₅ is a metal hydride, whereas CH₃Mn(CO)₅ is an alkylmetal complex. These differences in bonding and structure lead to variations in their physical properties and subsequent chemical reactivity.
Safety Considerations
It is imperative to acknowledge the safety aspects associated with dimanganese decacarbonyl and its derivatives. Mn₂(CO)₁₀ is a volatile compound and a source of carbon monoxide (CO), a highly toxic gas. Therefore, all manipulations should be carried out in well-ventilated areas, preferably within a fume hood, and appropriate personal protective equipment should be worn. The handling of solutions requires the aforementioned Schlenk techniques to prevent decomposition and exposure to air.
Frequently Asked Questions
What is the primary difference in the resulting bond to manganese after protonation versus methylation?
After protonation, a manganese-hydrogen (Mn-H) bond is formed. Following methylation, a manganese-carbon (Mn-C) bond is formed.
Are the products of protonation and methylation of [Mn(CO)₅]⁻ charged or neutral?
Both products, HMn(CO)₅ and CH₃Mn(CO)₅, are neutral species. The negative charge on the [Mn(CO)₅]⁻ anion is neutralised by the addition of the proton (H⁺) or the methyl group (CH₃⁺), respectively.
Can protonation and methylation occur directly on Mn₂(CO)₁₀?
While Mn₂(CO)₁₀ can undergo reactions that lead to the formation of species that are subsequently protonated or methylated, the direct protonation or methylation of the intact Mn-Mn bond in Mn₂(CO)₁₀ is not the typical pathway described. These reactions are generally discussed in the context of the manganese pentacarbonyl anion, [Mn(CO)₅]⁻, which is formed by the reductive cleavage of the Mn-Mn bond.
What is the significance of the manganese pentacarbonyl anion?
The manganese pentacarbonyl anion is a potent nucleophile and a versatile building block in organometallic synthesis. Its ability to react with various electrophiles, such as protons and alkyl halides, allows for the creation of a diverse range of manganese-containing compounds with different functionalities.
Conclusion
In summary, the reactions of dimanganese decacarbonyl, particularly through its derived anion [Mn(CO)₅]⁻, with protons and methylating agents highlight fundamental principles of organometallic reactivity. Protonation leads to the formation of a metal hydride, HMn(CO)₅, characterised by an Mn-H bond. Methylation, conversely, yields an alkylmetal complex, CH₃Mn(CO)₅, featuring an Mn-C bond. Understanding these distinctions is crucial for anyone working with or studying the chemistry of manganese carbonyls, opening doors to further synthetic possibilities and applications in catalysis and materials science.
If you want to read more articles similar to Protonation vs Methylation of Dimanganese Decacarbonyl, you can visit the Automotive category.
