15/02/2017
In the vast and intricate realm of chemistry, certain molecules stand out due to their unique properties and the profound insights they offer into fundamental principles. Among these, the dicarbon molecule, C2, holds a special place. Often found as a component of carbon vapours, its electronic structure provides a compelling illustration of molecular orbital theory and the nuanced behaviour of electrons within a bonded system. Understanding C2 isn't just an academic exercise; it sheds light on the very nature of chemical bonding and the forces that govern molecular stability. This article will delve into the heart of the C2 molecule, exploring its molecular orbitals, electron count, and the intriguing implications of its quantum states.
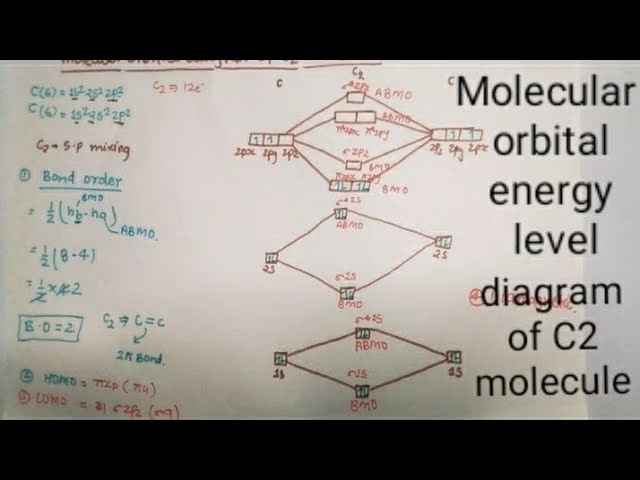
The Architecture of C2: Molecular Orbitals Explained
At the core of any molecule's identity lies its molecular orbital structure. These orbitals, formed from the overlap of atomic orbitals, dictate how electrons are distributed and, consequently, the molecule's stability and reactivity. For the dicarbon molecule, C2, the arrangement is particularly insightful. According to detailed molecular orbital diagrams, the C2 molecule boasts a total of 10 molecular orbitals. These orbitals are filled systematically, adhering to the fundamental principle that lower energy levels are occupied first, ensuring the most stable electronic configuration.
The formation of these molecular orbitals is a fascinating dance between the atomic orbitals of the two carbon atoms. The C2 molecule features two primary types of bonding interactions: sigma (σ) and pi (π) bonds. Sigma bonding orbitals are typically formed by the direct, head-on overlap of atomic orbitals, such as the 2s and 2p orbitals along the internuclear axis. This direct overlap results in a strong bond that is symmetrical around the bond axis.
Conversely, pi bonding orbitals arise from the sideways overlap of atomic p-orbitals. Imagine two p-orbitals on adjacent carbon atoms aligning parallel to each other and overlapping above and below the internuclear axis. This sideways interaction creates regions of electron density that are distinct from the sigma bond. In C2, the interplay between these sigma and pi bonding orbitals is crucial. This intricate bonding arrangement, with electrons occupying specific molecular orbitals, is precisely what allows for the formation of a stable C2 molecule, despite its highly reactive nature in many environments.
Counting the Electrons: The Electronic Nature of C2
To truly grasp the essence of the C2 molecule, one must consider its electron count and how these electrons are arranged. We know that C2 is a significant component of carbon vapours, particularly at high temperatures. Research indicates that carbon vapours can contain around 28% C2, though this proportion is highly dependent on environmental factors such as temperature and pressure. The distribution of electrons within the C2 molecule follows well-established principles of quantum mechanics, most notably Aufbau’s principle, which dictates the filling order of orbitals from lowest to highest energy.
Each electron within a molecule occupies a unique quantum state, each corresponding to a specific energy level. The most stable configuration, where all electrons occupy the lowest possible energy levels, is known as the ground state. For the neutral C2 molecule, this ground state is a singlet state, meaning all electron spins are paired. While the ground state is the most common, C2 also exhibits several excited singlet and triplet states. These excited states possess relatively similar energies to the ground state, making the dicarbon molecule quite dynamic and capable of existing in various energetic forms under ambient conditions.
Let's consider the electron count. A neutral carbon atom, as you may recall from basic chemistry, possesses a total of six electrons. Therefore, a neutral C2 molecule, being composed of two carbon atoms, has a total of 2 × 6 electrons, equating to 12 electrons. Molecular orbital theory elegantly explains how these 12 electrons are distributed. It reveals that C2 has two sets of paired electrons residing in a degenerate bonding set of orbitals. This specific electron configuration leads to a bond order of two, which fundamentally implies the existence of a double bond between the two carbon atoms in a C2 molecule. This is a crucial distinction, as many might initially assume a triple bond based on simple Lewis structures, but MO theory provides a more accurate picture.
C2 and C2-: A Tale of Two Magnetic Personalities
The addition or removal of a single electron can dramatically alter a molecule's properties, and the C2 molecule provides a perfect illustration of this phenomenon. Consider the C2- species, which is the C2 molecule with an additional electron. This anionic species will have 12 electrons from the neutral C2 plus one extra electron, totalling 13 electrons.
The question then arises: where does this extra electron go? In molecular orbital theory, electrons are added to the lowest energy unoccupied molecular orbital (LUMO). For the neutral C2 molecule, the highest energy occupied molecular orbitals (HOMO) are the two 2pπ orbitals (specifically, the 2pπx and 2pπy orbitals). The lowest energy unoccupied molecular orbital (LUMO) for C2 is the 2pσ orbital. Therefore, when an extra electron is introduced to form C2-, it occupies this 2pσ LUMO.
This addition has a profound impact on the molecule's magnetic properties. An unpaired electron, as is the case in C2- with its 13 electrons (an odd number, guaranteeing at least one unpaired electron), makes the C2- ion paramagnetic. This means it is attracted by an externally applied magnetic field. Conversely, the neutral C2 molecule, with its 12 electrons, has no unpaired electrons in its ground state configuration, as all electrons are paired in its degenerate bonding orbitals. Consequently, the neutral C2 molecule is diamagnetic, meaning it is not attracted by an externally applied magnetic field; in fact, it is weakly repelled.
This difference in magnetic behaviour serves as a powerful experimental confirmation of the molecular orbital theory predictions for C2 and C2-. The ability to predict and observe such distinct magnetic properties based on electron configuration highlights the robustness of the theory.

C2 in its Environment: Carbon Vapours and Quantum States
The C2 molecule is not merely a theoretical construct; it plays a real role in various chemical environments, most notably in the composition of carbon vapours. The presence and proportion of C2 in these vapours are highly sensitive to external conditions, particularly temperature and pressure. At extreme temperatures, such as those found in electric arcs or combustion processes, the stable C2 molecule becomes a significant species.
Moreover, the various quantum states of dicarbon, encompassing both its ground state and its numerous excited singlet and triplet states, are not just fleeting theoretical possibilities. These different energetic forms can exist in significant proportions under ambient conditions, contributing to the overall behaviour and spectroscopic signatures of carbon vapours. This complex interplay of states makes C2 an intriguing subject for spectroscopic studies, allowing scientists to probe its structure and dynamics with remarkable precision.
Understanding the C2 molecule, from its fundamental electron count to its intricate molecular orbital structure and magnetic properties, provides a deeper appreciation for the principles that govern chemical bonding. It demonstrates how seemingly abstract concepts like molecular orbitals and quantum states have very real, observable consequences on a molecule's behaviour and its interaction with its environment.
Comparative Analysis: C2 vs. C2-
To summarise the key differences between the neutral dicarbon molecule and its anionic counterpart, consider the following comparison:
| Property | Neutral C2 Molecule | C2- Ion |
|---|---|---|
| Total Electrons | 12 electrons | 13 electrons |
| Ground State Bond Order | Two (double bond) | Likely slightly reduced or altered due to electron in LUMO |
| Highest Occupied Molecular Orbital (HOMO) | Two 2pπ orbitals (2pπx, 2pπy) | 2pσ orbital (after adding electron) |
| Lowest Unoccupied Molecular Orbital (LUMO) | 2pσ orbital | Not applicable (electron added to this orbital) |
| Magnetic Property | Diamagnetic (no unpaired electrons) | Paramagnetic (one unpaired electron) |
| Attraction to Magnetic Field | Not attracted | Attracted |
Frequently Asked Questions About C2
How many molecular orbitals does C2 have?
The C2 molecule has a total of 10 molecular orbitals. These orbitals are formed from the overlap of the atomic orbitals of the two carbon atoms and are filled according to their energy levels, with the lowest energy orbitals being occupied first to achieve the most stable configuration.
How many electrons are in a C2 molecule?
A neutral C2 molecule has a total of 12 electrons. Each carbon atom contributes six electrons, resulting in 2 × 6 = 12 electrons for the dicarbon molecule. These electrons are distributed among the molecular orbitals, leading to specific bonding characteristics and magnetic properties.
Is C2 paramagnetic or diamagnetic?
The neutral C2 molecule is diamagnetic. This is because, in its ground state, all of its 12 electrons are paired within its molecular orbitals, meaning there are no unpaired electrons to interact with an external magnetic field. Conversely, the C2- ion, with its 13 electrons, has an unpaired electron and is therefore paramagnetic.
What is the bond order of C2?
According to molecular orbital theory, the C2 molecule has a bond order of two. This indicates the presence of a double bond between the two carbon atoms. This bond order arises from the specific filling of its molecular orbitals, which includes two sets of paired electrons in degenerate bonding orbitals.
Where does an extra electron go if C2 becomes C2-?
When an extra electron is added to a neutral C2 molecule to form the C2- ion, it occupies the lowest energy unoccupied molecular orbital (LUMO) of the neutral C2 molecule. For C2, the LUMO is the 2pσ orbital. This addition of an electron to the 2pσ orbital leads to the C2- ion having an unpaired electron, making it paramagnetic.
The dicarbon molecule, C2, serves as a compelling case study in molecular orbital theory, illustrating how the fundamental principles of quantum mechanics govern the very structure and behaviour of molecules. From its precise number of molecular orbitals to the exact count and arrangement of its electrons, C2 exemplifies the intricate dance of particles that underpins all chemical interactions. Understanding C2 not only deepens our appreciation for molecular chemistry but also highlights the profound impact that subtle changes, like the addition of a single electron, can have on a molecule's most fundamental properties.
If you want to read more articles similar to Unravelling the Dicarbon Molecule (C2), you can visit the Automotive category.
