24/02/2005
The question of whether C2 is a neutral molecule is a fundamental one in molecular chemistry. The answer is a resounding yes. Diatomic carbon, or C2, is indeed a neutral molecule. This might seem straightforward, but understanding *why* it's neutral and exploring its electronic structure reveals some intriguing aspects of chemical bonding. Unlike ionic compounds where electrons are transferred, neutral molecules are formed when atoms share electrons, or in the case of C2, when atoms of the same element bond together.
Understanding Molecular Neutrality
A molecule is considered neutral if its net electrical charge is zero. This means the total number of protons (positive charges) in its constituent atoms equals the total number of electrons (negative charges) in the molecule. Carbon, in its elemental form and in most of its compounds, is electrically neutral. When two carbon atoms bond to form C2, they share their valence electrons, maintaining an overall neutral charge for the resulting molecule.
The Homonuclear Nature of C2
C2 is classified as a homonuclear diatomic molecule. This means it is composed of two atoms of the same element. This is a crucial distinction because it simplifies the analysis of its bonding compared to heteronuclear diatomic molecules (like CO or NO), where atoms of different elements are involved. In homonuclear molecules, the electron distribution is generally more symmetrical, and the molecular orbitals formed from the atomic orbitals of the two identical atoms have predictable energy levels.
Electron Configuration and Bonding in C2
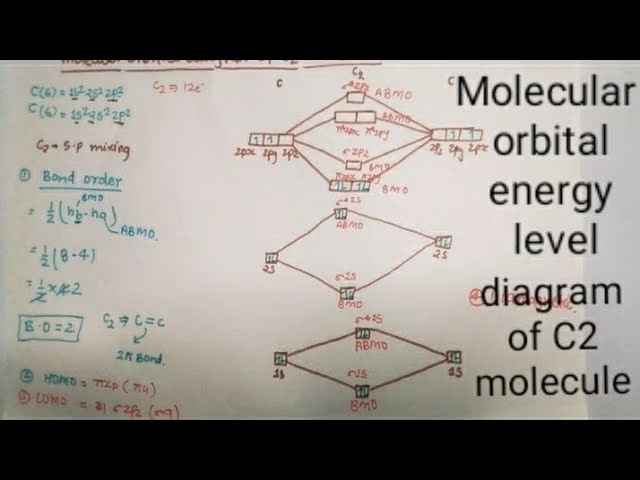
To understand the bonding in C2, we can utilise the Linear Combination of Atomic Orbitals (LCAO) method. This approach explains how atomic orbitals overlap to form molecular orbitals. Each carbon atom has an atomic number of 6, meaning it has 6 protons and typically 6 electrons. The electron configuration of a free carbon atom is 1s²2s²2p². When two carbon atoms come together to form C2, we consider their valence electrons, which are in the 2s and 2p orbitals.
Molecular Orbital Diagram for C2
Applying the LCAO theory to C2, we consider the overlap of atomic orbitals:
- 1s Atomic Orbitals: The two 1s atomic orbitals from each carbon atom overlap. This overlap results in the formation of two molecular orbitals: a lower-energy bonding molecular orbital (σ1s) and a higher-energy antibonding molecular orbital (σ*1s).
- 2s Atomic Orbitals: Similarly, the two 2s atomic orbitals overlap to form a bonding σ2s molecular orbital and an antibonding σ*2s molecular orbital.
- 2p Atomic Orbitals: The four 2p atomic orbitals (two from each carbon atom) combine to form molecular orbitals. For diatomic molecules of second-period elements, the order of energy levels for these molecular orbitals can vary. However, for C2, the typical order involves two pi (π2p) bonding orbitals, followed by a sigma (σ2p) bonding orbital, and then antibonding orbitals (π*2p and σ*2p).
The electron configuration of C2 can be written as (σ1s)²(σ*1s)²(σ2s)²(σ2p)² or (σ1s)²(σ*1s)²(σ2s)²(π2p)². The latter is more commonly accepted based on experimental evidence and more sophisticated calculations.
Bond Order and Stability
The bond order is a crucial concept in determining the stability and nature of a chemical bond. It is calculated as half the difference between the number of electrons in bonding molecular orbitals and the number of electrons in antibonding molecular orbitals:
Bond Order = 0.5 * (Number of bonding electrons - Number of antibonding electrons)
Let's consider the valence electrons for C2. Each carbon atom contributes 4 valence electrons (2s²2p²), so C2 has a total of 8 valence electrons. Using the more accepted molecular orbital configuration (σ2s)²(π2p)²:
- Bonding electrons = 2 (from σ2s) + 2 (from π2p) = 4
- Antibonding electrons = 0 (in the valence shell considered here)
Therefore, the bond order for C2 is 0.5 * (4 - 0) = 2. This indicates a double bond between the two carbon atoms. This double bond contributes to the molecule's stability, although C2 is still quite reactive and typically exists under specific conditions, such as in interstellar space or high-temperature environments.
Experimental Evidence and C2's Existence
The existence of the C2 molecule has been confirmed through various spectroscopic methods. It is observed in flames, comets, and the spectra of stars. The characteristic Swan bands in molecular spectroscopy are attributed to electronic transitions within the C2 molecule. These observations provide strong evidence for its existence and the nature of its bonding.
Why C2 is Not an Ion
It's important to distinguish C2 from ionic species. An ion is formed when an atom or molecule gains or loses electrons, resulting in a net positive or negative charge. For example, a carbon anion like C⁻ or C²⁻ would involve a different number of electrons compared to protons. C2, however, is formed by the sharing of electrons between two neutral carbon atoms, thus maintaining an overall charge of zero. It does not possess a positive or negative charge.
Comparison with Other Diatomic Molecules
To further illustrate the concept of neutral diatomic molecules, let's compare C2 with others:
| Molecule | Composition | Bond Order | Nature | Neutral? |
|---|---|---|---|---|
| H₂ | Two Hydrogen atoms | 1 | Single bond (σ) | Yes |
| O₂ | Two Oxygen atoms | 2 | Double bond (one σ, one π) | Yes |
| N₂ | Two Nitrogen atoms | 3 | Triple bond (one σ, two π) | Yes |
| CO | One Carbon, One Oxygen atom | 3 | Triple bond (polar) | Yes |
| NO | One Nitrogen, One Oxygen atom | 2.5 | Paramagnetic (unpaired electrons) | Yes |
| C₂ | Two Carbon atoms | 2 | Double bond (two π) | Yes |
As seen in the table, molecules like H₂, O₂, N₂, CO, NO, and C₂ are all neutral. The difference lies in their composition (homonuclear vs. heteronuclear) and the number of electrons involved in bonding, leading to different bond orders and properties.
Frequently Asked Questions
Is C2 a stable molecule?
C2 is considered to be relatively unstable under standard laboratory conditions. It readily reacts with other substances. However, it is stable enough to exist in high-temperature environments like flames and in interstellar space where collisions are infrequent.
What is the bond order of C2?
The bond order of C2 is 2, indicating a double bond between the two carbon atoms, typically described as consisting of two pi (π) bonds.
Why is C2 sometimes described as having two pi bonds and no sigma bond?
This is a simplified but often used description based on the molecular orbital filling order for C2. In more detailed molecular orbital theory, the σ2s and σ*2s orbitals are filled, and then the 2p atomic orbitals combine. For C2, the order of filling is often considered to be (σ2s)²(σ*2s)²(π2p)². This configuration places electrons in two π bonding orbitals, leading to a bond order of 2. While there are σ molecular orbitals involved (σ2s and σ*2s), the valence bonding is often discussed in terms of the π orbitals contributing to the bond.
Does C2 have unpaired electrons?
No, according to the standard molecular orbital filling, all electrons in C2 are paired. This means C2 is diamagnetic.
Where is C2 found?
C2 is commonly found in high-temperature environments such as flames (e.g., acetylene flames), comets, and in the atmospheres of stars. It is also a key species in carbon vapour.
Conclusion
In summary, C2 is definitively a neutral molecule. Its neutral status arises from the equal sharing of electrons between two carbon atoms, maintaining a zero net charge. While its bonding structure, characterised by a double bond formed primarily through pi interactions in its valence shell, makes it reactive, its existence is well-established through spectroscopic and astronomical observations. Understanding C2 provides valuable insight into the complexities of chemical bonding and the behaviour of molecules in extreme environments.
If you want to read more articles similar to Is C2 a Neutral Molecule?, you can visit the Automotive category.
