29/03/2013
Every time you pull up to the pump, you’re engaging with the final product of an incredibly complex and ingenious industrial process. The clear, free-flowing petrol that powers your vehicle started its life far from this refined state. It began as crude oil, a thick, dark, almost molasses-like substance extracted from deep within the Earth. The transformation from this raw, unappealing material into the highly specific fuels and products we rely on daily is a testament to engineering and chemical science. This article will take you on a journey through the fascinating world of oil refining, explaining how this 'black gold' is meticulously converted into the petrol that gets you from A to B.
- Understanding Crude Oil: Nature's Black Gold
- The Heart of Refining: Fractional Distillation
- Transforming Molecules: Chemical Refining
- The Vital Step: Purification and Treatment
- From Refinery to Road: Blending the Final Product
- The Global Landscape and Future Outlook
- Frequently Asked Questions About Crude Oil Refining
Understanding Crude Oil: Nature's Black Gold
Crude oil, often referred to as 'black gold', is far from a uniform substance. It is, in fact, a complex mixture of hydrocarbons – organic compounds made solely of hydrogen and carbon atoms. These hydrocarbons were formed over millions of years from the remains of ancient plants and animals, subjected to immense heat and pressure beneath the Earth's surface. What makes crude oil so versatile is the variety of these hydrocarbon molecules. They exist in different sizes and structures, ranging from short, light chains to long, heavy ones. Each of these distinct hydrocarbon chains has a unique boiling point and, crucially, a unique application. For instance, light gases like propane might have just three carbon atoms, while the hydrocarbons that make up petrol typically have around eight, and heavier fuels like diesel contain even more. The primary goal of a refinery is to separate these various hydrocarbons and then, if necessary, to chemically alter them to maximise the yield of the most demanded products, with petrol often being at the top of that list.
The Heart of Refining: Fractional Distillation
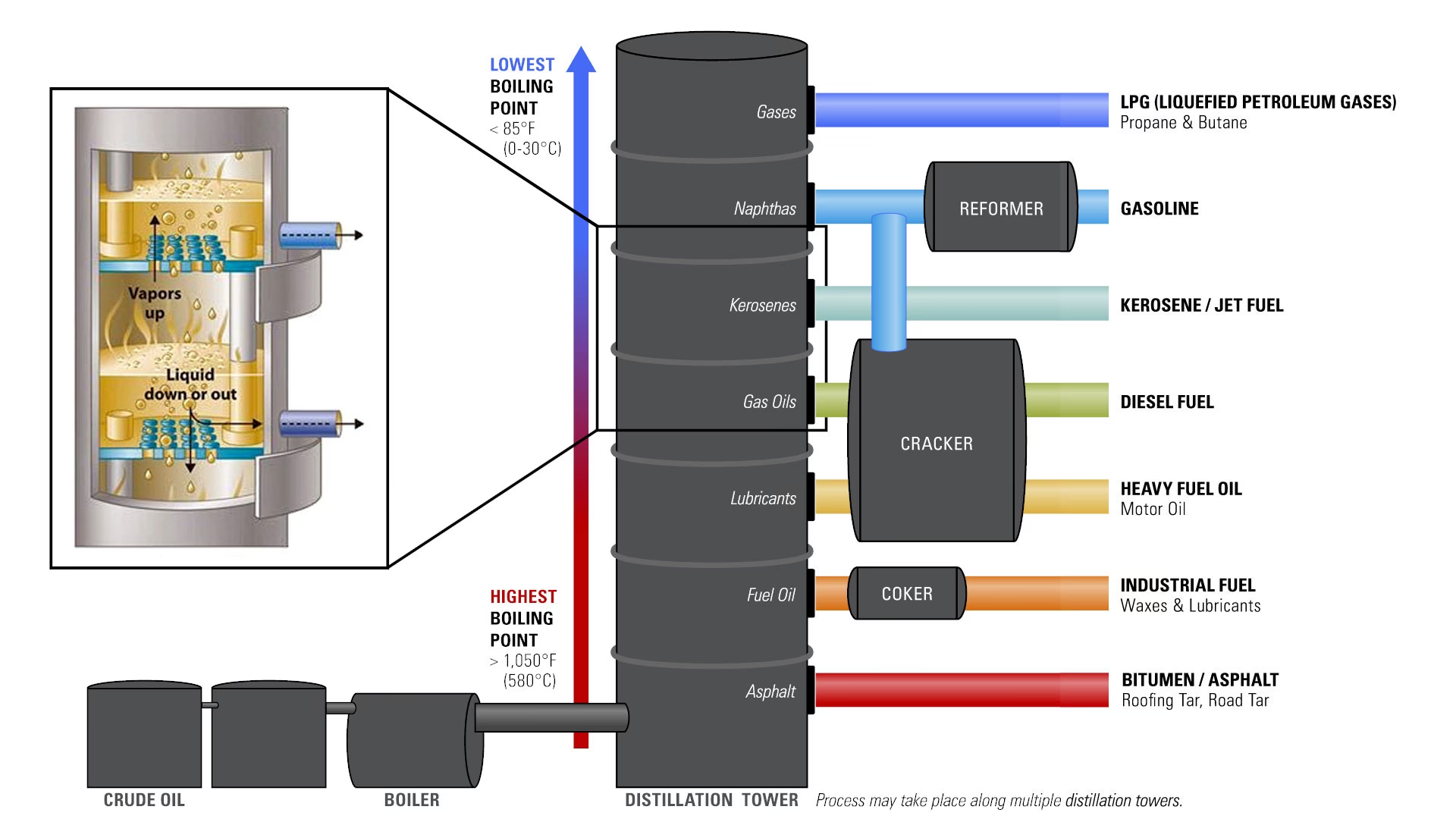
The initial and most fundamental step in transforming crude oil is a process known as fractional distillation. This method exploits the fact that different hydrocarbons have different boiling points. Imagine a vast industrial kitchen where a giant pot of crude oil is heated to incredibly high temperatures, often exceeding 600°C (1112°F). At these extreme temperatures, nearly all the hydrocarbons in the crude oil vaporise, turning into a hot gas.
This hot vapour is then channelled into a towering structure called a distillation column, sometimes referred to as a fractionating column. This column is cooler at the top and progressively hotter towards the bottom. As the hot hydrocarbon vapour rises through the column, it gradually cools. Different hydrocarbons condense back into liquid form at different levels, based on their specific boiling points. The heaviest hydrocarbons, with the highest boiling points, condense first at the bottom of the column. These might include residual fuels, asphalt, or 'coke', which can be further processed or even burned for energy within the refinery itself.
As the vapour continues its ascent, lighter hydrocarbons condense at higher levels where the temperature is lower. Screens and trays strategically placed throughout the column collect these separated liquid fractions. From the bottom upwards, you would typically find distinct layers emerging:
- Heavy Fuel Oils/Asphalt: Highest boiling points, collected at the very bottom.
- Lubricating Oils: Used in engines and machinery.
- Diesel Fuel: A common fuel for lorries and buses.
- Kerosene: Used for jet fuel and heating oil.
- Naphtha: A key feedstock for petrochemicals and a precursor to petrol.
- Petrol: The target product, condensing in the mid-to-upper sections.
- Light Gases (e.g., Propane, Butane): Lowest boiling points, collected at the very top.
This elegant physical separation process is remarkably efficient, allowing refineries to isolate a wide range of products from a single input stream of crude oil. However, distillation alone doesn't always yield the exact proportions of products required by the market, particularly when demand for petrol is high.
Typical Products from Fractional Distillation and Their Characteristics
To better illustrate the separation, here's a simplified overview:
| Product | Approx. Carbon Atoms | Approx. Boiling Point Range (°C) | Common Uses |
|---|---|---|---|
| Refinery Gases | C1-C4 | <30 | LPG, chemical feedstock |
| Petrol (Gasoline) | C5-C12 | 30-200 | Motor fuel |
| Naphtha | C5-C12 | 30-200 | Petrochemicals, solvent |
| Kerosene | C12-C16 | 175-275 | Jet fuel, heating oil |
| Diesel (Gas Oil) | C15-C18 | 250-350 | Diesel engines, heating oil |
| Lubricating Oils | C18-C25 | 350-400 | Engine oils, greases |
| Fuel Oils | C20-C70 | >350 | Ship fuel, power stations |
| Bitumen (Asphalt) | C70+ | Residue | Road construction |
Transforming Molecules: Chemical Refining
While fractional distillation is excellent for separating hydrocarbons, it often doesn't produce enough of the most valuable products, such as petrol, from a given barrel of crude oil. This is where chemical refining comes into play. These processes allow refineries to 'tweak' the output, either breaking down larger, less valuable hydrocarbon chains into smaller, more desirable ones, or combining smaller chains to form larger ones. This flexibility is crucial for meeting fluctuating market demands.
Cracking: Breaking Down for More Petrol
One of the most vital chemical refining processes is cracking. As the name suggests, cracking involves breaking down large, heavy hydrocarbon molecules into smaller, lighter ones. This is particularly important because heavy fuel oil components, which have limited direct use, can be 'cracked' into more valuable products like petrol and diesel.
There are several methods of cracking:
- Thermal Cracking: This involves simply applying intense heat (up to 750°C) and pressure to the heavy hydrocarbons. The high temperatures cause the large molecules to break apart into smaller fragments. An older method, it's still used, but modern refineries often favour catalytic cracking.
- Catalytic Cracking (Cat Cracking): This is the most common form of cracking today. Instead of relying solely on heat, a catalyst (a substance that speeds up a chemical reaction without being consumed itself) is used. Common catalysts include zeolites, which are porous mineral compounds. The heavy hydrocarbon vapours are passed over a hot catalyst, which facilitates the breaking of the molecular bonds at much lower temperatures than thermal cracking. This method is more efficient and produces higher yields of desired products, particularly high-octane petrol components. Fluid Catalytic Cracking (FCC) is a widely used variant.
- Hydrocracking: This process combines cracking with hydrogenation (the addition of hydrogen gas). It's used to break down very heavy, often contaminated, feedstocks into lighter, cleaner products like jet fuel, diesel, and high-quality petrol components. The presence of hydrogen helps to remove impurities like sulphur and nitrogen during the cracking process.
Unification and Alkylation: Building Up for Purpose
Conversely, sometimes it's beneficial to combine smaller hydrocarbon molecules to create larger, more valuable ones. This process is generally known as unification or polymerisation, though alkylation is a specific, important type of unification.

- Polymerisation: This involves combining small, unsaturated hydrocarbon molecules (often gases like propylene and butylene, byproducts of cracking) into longer-chain molecules suitable for petrol. This typically uses an acid catalyst.
- Alkylation: This is a highly significant process that combines low molecular weight iso-paraffins (e.g., isobutane) with olefins (e.g., propylene, butylene) in the presence of a strong acid catalyst (such as sulphuric acid or hydrofluoric acid). The resulting product, called alkylate, is a high-octane, clean-burning petrol component that enhances the overall quality and knock resistance of the final petrol blend. A valuable byproduct of unification processes can often be hydrogen gas, which is then re-used in other refinery processes like hydrocracking or sold.
These chemical conversion processes are what allow refineries to produce almost half a barrel of petrol from a barrel of crude oil, even though the naturally occurring petrol-range hydrocarbons might only account for a smaller fraction. They are the true game-changers in modern refining.
The Vital Step: Purification and Treatment
Once the hydrocarbons have been separated and chemically altered, they are still not ready for sale. Crude oil, and the products derived from it, contain various impurities that must be removed. These impurities can corrode equipment, reduce fuel quality, and, most importantly, cause significant environmental pollution when burned. Key impurities include sulphur, nitrogen, oxygen compounds, and unsaturated hydrocarbons.
The purification process involves several stages:
- Sulphur Removal (Hydrotreating): Sulphur is one of the most problematic impurities, contributing to acid rain and particulate matter when fuels are combusted. Hydrotreating processes, such as hydrodesulphurisation (HDS), involve reacting the hydrocarbon streams with hydrogen gas over a catalyst (like cobalt-molybdenum) at high temperatures and pressures. This converts sulphur compounds into hydrogen sulphide gas, which can then be captured and converted into elemental sulphur, a valuable byproduct used in fertilisers and other industries.
- Nitrogen and Oxygen Compound Removal: Similar to sulphur, nitrogen and oxygen compounds are removed through catalytic processes, often involving hydrogen. Nitrogen compounds can be converted into ammonia through a process called water washing, and this ammonia can then be used as farm fertiliser.
- Water Removal: After various chemical treatments, liquid products are passed through absorption columns or other drying processes to remove any residual water, which can cause corrosion and problems in engines.
- Unsaturated Hydrocarbon Treatment: Unsaturated hydrocarbons can be unstable and form gums or deposits in fuel systems. They are often converted into more stable saturated hydrocarbons through hydrogenation.
This comprehensive treatment ensures that the refined products meet stringent quality and environmental standards before they are distributed.
From Refinery to Road: Blending the Final Product
The various treated hydrocarbon streams are now ready to be blended into the final products we recognise. For petrol, this involves combining different components (such as straight-run petrol from distillation, cracked petrol, alkylate, and reformate – a high-octane component produced from naphtha) in precise proportions. This blending process is crucial for achieving the desired octane rating (a measure of a fuel's resistance to knocking or pre-ignition in an engine) and other performance characteristics. Different grades of petrol, such as unleaded 95 or super unleaded 98, are created by varying these blends. Additives are also introduced at this stage to improve fuel performance, protect engines, and prevent corrosion.
Once blended and quality-checked, the finished petrol is ready for distribution. It is transported via pipelines, tanker lorries, rail cars, or even ships to petrol stations, where it finally makes its way into your vehicle's fuel tank.
The Global Landscape and Future Outlook
The process of converting crude oil into petrol and other products is carried out at an astonishingly high volume across the globe. Modern refineries are incredibly complex and efficient operations, designed to extract maximum value from every barrel of crude. They continuously adjust their output mixes based on global market demands, which are frequently heaviest for petrol. The sheer scale of this industry highlights our global dependence on refined petroleum products.
However, the future of refining faces significant questions. The finite nature of crude oil reserves, coupled with growing environmental concerns about carbon emissions, is driving a global shift towards alternative energy sources and electric vehicles. Furthermore, the geopolitical implications of relying heavily on oil from specific, often unstable, regions of the world add another layer of complexity. While the refining industry is constantly evolving to become more efficient and reduce its environmental footprint, the long-term viability of crude oil as our primary energy source remains a subject of ongoing debate and innovation.

Frequently Asked Questions About Crude Oil Refining
What exactly is crude oil?
Crude oil is a naturally occurring, unrefined petroleum product composed of hydrocarbon deposits and other organic materials. It's a fossil fuel, formed from the remains of ancient marine organisms and plants over millions of years under intense heat and pressure. It's found in geological formations beneath the Earth's surface and is the raw material for a vast array of products, not just fuels.
Why is "cracking" so important in refining?
Cracking is crucial because the natural yield of petrol from direct distillation of crude oil is often insufficient to meet market demand. Cracking allows refiners to break down heavier, less valuable hydrocarbon molecules (like those found in heavy fuel oil) into lighter, more valuable ones (like petrol and diesel). This significantly increases the overall yield of high-demand products from each barrel of crude oil, making the refining process more economically viable and responsive to market needs.
Are there other products made from crude oil besides petrol?
Absolutely! While petrol is a major output, crude oil is the raw material for an astonishing variety of products. These include diesel, jet fuel, heating oil, lubricating oils, asphalt for roads, and liquefied petroleum gas (LPG). Beyond fuels, petroleum is also the primary feedstock for the petrochemical industry, which produces plastics, synthetic fibres, fertilisers, solvents, pharmaceuticals, cosmetics, and countless other everyday items, from crayons to spectacle frames.
How much petrol can be produced from one barrel of crude oil?
On average, a 42-gallon (approximately 159 litres) barrel of crude oil can yield about 19-20 gallons (around 72-76 litres) of petrol. This figure is significantly higher than the amount of naturally occurring petrol-range hydrocarbons in crude oil. The increased yield is largely due to the chemical refining processes like cracking and alkylation, which convert heavier or lighter hydrocarbons into petrol components, maximising the output of this highly demanded fuel.
Is the refining process environmentally friendly?
While modern refineries are designed for high efficiency and have implemented significant measures to reduce their environmental impact compared to historical operations, the refining process is still energy-intensive and has environmental considerations. Emissions of greenhouse gases, sulphur dioxide, and nitrogen oxides are carefully monitored and regulated. Additionally, the handling and transportation of crude oil and refined products carry risks of spills. The industry is continuously investing in technologies to improve efficiency, reduce emissions, and enhance safety, but the overall environmental footprint of fossil fuel refining remains a key area of focus for global sustainability efforts.
From the moment crude oil is extracted from the ground to the second it powers your engine, it undergoes a remarkable transformation. The intricate dance of heating, cooling, separating, breaking, and building molecules within a refinery is a testament to human ingenuity. It’s a process that underpins much of our modern world, providing the energy that fuels our transport and powers countless industries. Understanding this complex journey offers a deeper appreciation for the energy that drives our daily lives and the sophisticated engineering behind every drop of petrol.
If you want to read more articles similar to From Goo to Go: The Crude Oil to Petrol Journey, you can visit the Fuel category.
