13/06/2003
- The Magic of Fractional Distillation: Unlocking Crude Oil's Potential
- How Does Fractional Distillation Work?
- The Journey Up the Column: Separating the Fractions
- Why is Crude Distillation So Important?
- Challenges in Crude Distillation
- Beyond Distillation: Cracking and Further Processing
- Frequently Asked Questions
The Magic of Fractional Distillation: Unlocking Crude Oil's Potential
Crude oil, that thick, black, and often pungent liquid extracted from the earth, is a cornerstone of modern civilisation. While it might seem uninspiring in its raw state, crude oil is a complex mixture of countless hydrocarbon compounds. Without a sophisticated refining process, this valuable resource would remain largely unusable. The primary method employed to transform crude oil into the fuels and materials we rely upon daily is called fractional distillation. This ingenious process leverages a fundamental scientific principle: that different substances, when present in a mixture, will vaporise and condense at distinct temperatures.
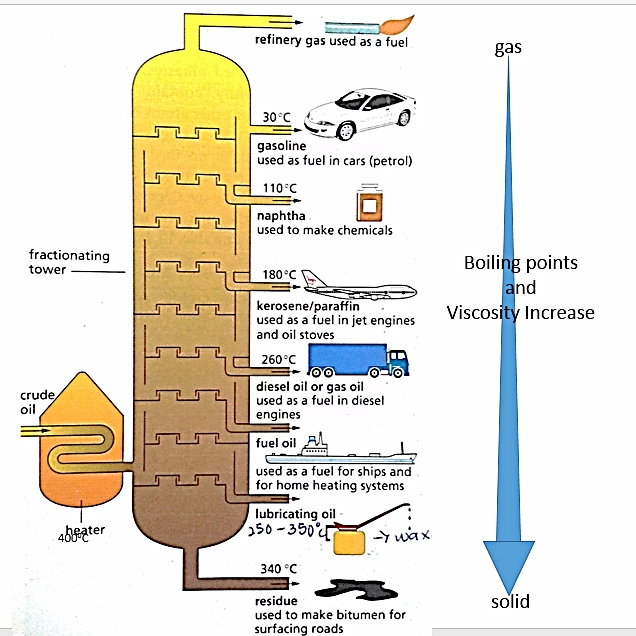
How Does Fractional Distillation Work?
At the heart of crude oil refining lies the fractionating column, a towering structure designed to exploit the differing boiling points of the hydrocarbons within crude oil. The process begins by heating the crude oil, often in the presence of high-pressure steam, to a high temperature. This causes the majority of the hydrocarbons to vaporise, transforming the liquid into a gaseous state.
This hot, gaseous mixture is then introduced into the lower part of the fractionating column. The column itself is ingeniously designed with a temperature gradient. The base of the column is incredibly hot, while the temperature gradually decreases as you ascend towards the top. This creates distinct temperature zones throughout the column, each conducive to the condensation of specific hydrocarbon fractions.
As the hydrocarbon vapours rise through the column, they begin to cool. When a hydrocarbon vapour reaches a temperature equal to its specific boiling point, it will condense back into a liquid. This liquid then collects on trays or plates strategically positioned at different levels within the column. Hydrocarbons with higher boiling points will condense at lower, hotter levels, while those with lower boiling points will continue to rise higher up the column before condensing in cooler zones.
The Journey Up the Column: Separating the Fractions
The beauty of fractional distillation lies in its ability to separate the complex crude oil mixture into more manageable and useful groups, known as fractions. Each fraction is essentially a mixture of hydrocarbons with a similar range of boiling points and, consequently, similar physical properties. Let's explore some of the key fractions obtained:
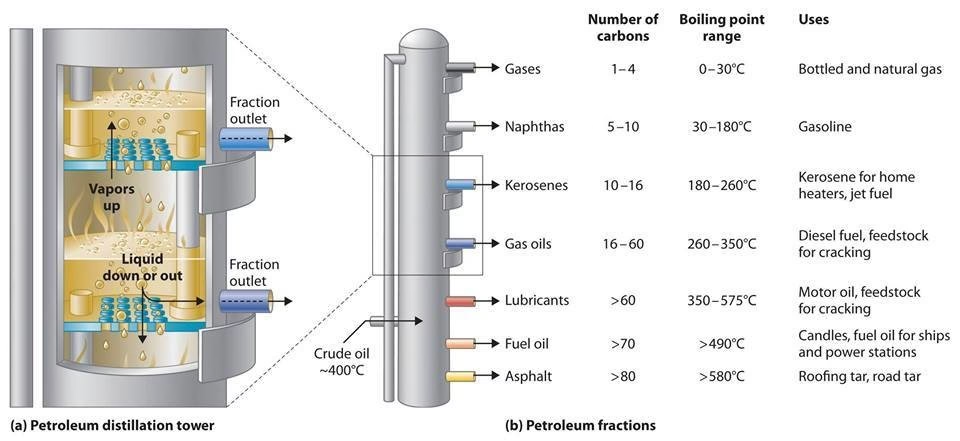
| Fraction | Boiling Point Range (°C) | Number of Carbon Atoms | Typical Uses | Viscosity | Volatility |
|---|---|---|---|---|---|
| Refinery Gases | < 40 | 1-4 | Bottled gas (LPG), chemical feedstock | Very low | Very high |
| Naphtha/Light Distillate | 40-180 | 5-12 | Petrol (gasoline) production, chemical feedstock | Low | High |
| Petrol (Gasoline) | 40-205 | 5-12 | Fuel for cars | Low | High |
| Kerosene | 175-325 | 12-16 | Jet fuel, paraffin for lamps | Medium | Medium |
| Diesel Oil | 250-350 | 15-18 | Fuel for diesel engines, heating oil | Medium-High | Medium-Low |
| Lubricating Oil | > 350 | 18-25 | Lubricants, waxes, polishes | High | Low |
| Bitumen/Asphalt | Residue | > 25 | Road surfacing, roofing | Very high | Very low |
It's crucial to understand the relationship between the size of a hydrocarbon molecule and its properties. As the size of the hydrocarbon molecule increases:
- The boiling point increases.
- The colour becomes darker.
- Flammability decreases.
- Usefulness as a direct fuel often decreases (though they are vital feedstocks).
- Viscosity (resistance to flow) increases; they become thicker and less fluid.
- Volatility (tendency to evaporate) decreases.
Why is Crude Distillation So Important?
The importance of crude oil distillation cannot be overstated. Without this initial refining step, the vast majority of the hydrocarbons within crude oil would be unusable. The process effectively unlocks the potential of crude oil, transforming it into the essential fuels that power our vehicles, heat our homes, and enable industries to operate. Furthermore, many of these fractions serve as crucial chemical feedstocks for the production of plastics, synthetic fibres, pharmaceuticals, and countless other materials that define modern life.
The composition of crude oil can vary significantly depending on its geological origin. Some crude oils are naturally lighter and more volatile, containing a higher proportion of smaller, more useful hydrocarbons. Others are heavier and more viscous, with a greater abundance of larger, less volatile molecules. This variability necessitates sophisticated control and optimisation within refinery operations to maximise profitability and meet market demands.
Challenges in Crude Distillation
While fractional distillation is a well-established and highly effective process, it is not without its challenges. Several factors can impact the efficiency and effectiveness of crude oil distillation:
- Crude Oil Variability: As mentioned, different crude oils have different compositions, requiring adjustments to the distillation process.
- Equipment Malfunctions: Issues with heaters, pumps, or the fractionating column itself can disrupt operations.
- Improper Pressure and Temperature Control: Maintaining the correct temperature and pressure gradients is critical for effective separation. Fluctuations can lead to incorrect fraction collection.
- Column Fouling: Over time, heavy residues can build up on the trays within the column, reducing efficiency and requiring cleaning.
- Corrosive Components: Crude oil can contain sulfur compounds and acids, which can be corrosive to equipment if not properly managed. Desalting processes are often employed to remove salts and water before distillation to mitigate these issues.
Addressing these challenges often involves rigorous monitoring, regular maintenance, and the implementation of advanced control systems. Specialized training for refinery personnel is also vital to ensure safe and efficient operation. Courses focusing on troubleshooting and optimisation of crude distillation units, covering aspects like feed preparation, column design, operational parameters, and incident analysis, are invaluable for industry professionals.
Beyond Distillation: Cracking and Further Processing
It's important to note that fractional distillation is often just the first stage in the complex refining process. While it separates crude oil into useful fractions, there can be an imbalance between the supply of certain fractions and market demand. For instance, there is often a higher demand for lighter fuels like petrol than for heavier fractions like lubricating oil or bitumen.
To address this, a process called cracking is employed. Cracking uses high temperatures and catalysts to break down larger, less useful hydrocarbon molecules into smaller, more valuable ones, such as those found in petrol. This allows refineries to adjust their product output to meet market needs more effectively.
Frequently Asked Questions
Q1: What is the primary purpose of fractional distillation of crude oil?
A1: The primary purpose is to separate crude oil into various fractions based on their different boiling points, making them more useful for different applications.
Q2: Why are the temperatures lower at the top of a fractionating column?
A2: The lower temperatures at the top allow hydrocarbons with lower boiling points to condense, while higher boiling point hydrocarbons remain as gases and condense at lower, hotter levels.

Q3: What happens to hydrocarbons with very high boiling points?
A3: Hydrocarbons with very high boiling points do not vaporise easily and remain as liquids at the bottom of the fractionating column. These are often referred to as the residue and include materials like bitumen.
Q4: Is petrol obtained directly from fractional distillation?
A4: Petrol, or gasoline, is a fraction obtained from fractional distillation, typically condensing in the middle sections of the column. However, further processing and blending may occur to meet specific octane ratings and performance requirements.
Q5: What is the difference between fractional distillation and simple distillation?
A5: Simple distillation is used to separate liquids with significantly different boiling points or to separate a liquid from a dissolved solid. Fractional distillation is used to separate mixtures of liquids with closer boiling points, using a fractionating column to achieve a more efficient separation through repeated vaporisation and condensation cycles.
In conclusion, the process of fractional distillation is a fundamental pillar of the oil refining industry, enabling the transformation of raw crude oil into the essential fuels and materials that underpin our modern way of life. Understanding this process provides valuable insight into the journey of hydrocarbons from the ground to our everyday products.
If you want to read more articles similar to Crude Oil Distillation: Separating the Essentials, you can visit the Automotive category.
