24/01/2013
- The Curious Case of Group 7: More Reactive Than You Think!
- What Exactly Are Group 7 Elements?
- The Myth Busted: Group 7 and Metals
- Reactivity Trends Within Group 7
- Physical Properties of the Halogens
- How Halogens Behave in Reactions
- Displacement Reactions: A Halogen Hierarchy
- The Impact of Reactivity on Atomic Mass
- Frequently Asked Questions about Group 7
- Conclusion
The Curious Case of Group 7: More Reactive Than You Think!
The periodic table is a fascinating map of the elements, organised by their properties. Within this intricate arrangement, Group 7 holds a special place. Known as the halogens, these elements are defined by their high reactivity, particularly their tendency to interact with metals. Far from being inert, the halogens are the salt-producers of the elemental world, a characteristic hinted at by their very name, derived from Greek words meaning 'salt-producing'. This article delves into the intriguing world of Group 7, exploring their reactions, properties, and the underlying reasons for their chemical behaviour. We'll uncover why the statement that Group 7 elements won't react with metals is, in fact, entirely false, and explore the nuances of their interactions.
What Exactly Are Group 7 Elements?
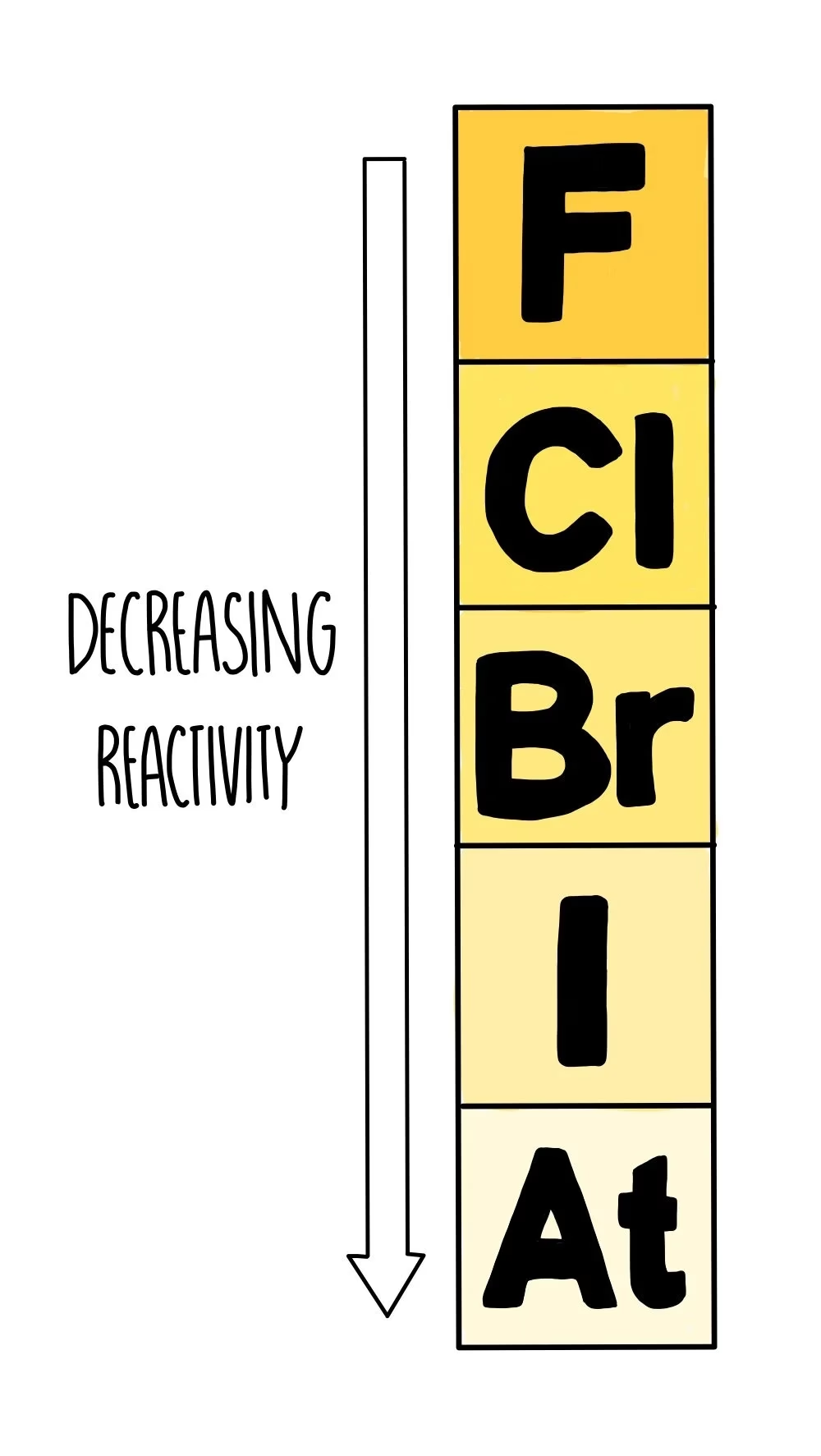
Group 7 of the periodic table is home to the halogens. This group includes fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). These elements are all non-metals, and they share a distinctive electronic configuration. Each halogen atom possesses seven electrons in its outermost shell. This incomplete outer shell is the key to their reactivity. To achieve a stable, full outer shell, similar to that of the noble gases, halogen atoms have a strong tendency to gain one electron. This drive to complete their outer shell makes them highly reactive and eager participants in chemical reactions.

The Myth Busted: Group 7 and Metals
It's a common misconception that certain elements shy away from reacting with metals. However, when it comes to Group 7, the opposite is true. The halogens are renowned for their vigorous reactions with metals. When a halogen atom encounters a metal atom, the halogen readily accepts an electron from the metal. This electron transfer results in the formation of an ionic bond, creating a metal halide salt. For instance, sodium (a metal from Group 1) reacts vigorously with chlorine (a halogen from Group 7) to form sodium chloride (NaCl), commonly known as table salt. This reaction is a prime example of the fundamental nature of halogen-metal interactions. The generalisation that Group 7 elements will not react with metals is demonstrably false.
Reactivity Trends Within Group 7
While all halogens are reactive, their reactivity is not uniform. There's a predictable trend as you move down Group 7: reactivity decreases. Fluorine is the most reactive halogen, followed by chlorine, then bromine, and finally iodine. Astatine is also in Group 7, but it's a radioactive element and less commonly studied in introductory chemistry. This decrease in reactivity is linked to the increasing atomic size and the distance of the outermost electron shell from the nucleus. As you go down the group, the nucleus's attraction to an incoming electron in the outer shell weakens, making it less likely for the atom to gain an electron. This is the opposite trend to that observed in Group 1, the alkali metals, where reactivity increases down the group.
Physical Properties of the Halogens
The halogens exhibit a range of physical properties that also change down the group. At room temperature, fluorine and chlorine are gases, bromine is a liquid, and iodine is a solid. Their colours also vary, from the pale yellow of chlorine gas to the reddish-brown of bromine liquid and the violet vapour of iodine. They all exist as diatomic molecules, meaning they exist as pairs of atoms bonded together (e.g., F₂, Cl₂, Br₂, I₂). This diatomic nature is crucial for understanding their reactions.
| Element | Symbol | State at Room Temp. | Colour | Melting Point (°C) | Boiling Point (°C) |
|---|---|---|---|---|---|
| Fluorine | F | Gas | Pale Yellow | -219.6 | -188.1 |
| Chlorine | Cl | Gas | Yellow-Green | -101.5 | -34.0 |
| Bromine | Br | Liquid | Red-Brown | -7.2 | 58.8 |
| Iodine | I | Solid | Grey/Purple (solid), Violet (vapour) | 113.7 | 184.3 |
How Halogens Behave in Reactions
As mentioned, the defining characteristic of Group 7 elements is their seven valence electrons. This means they are just one electron short of a full outer shell. In a chemical reaction, a halogen atom will readily accept an electron from another atom, typically a metal or a less reactive non-metal. This process is called reduction, as the halogen atom gains electrons. When a halogen gains an electron, it forms a halide ion with a charge of -1 (e.g., F⁻, Cl⁻, Br⁻, I⁻). These halide ions are stable because they now have a complete outer electron shell.

Displacement Reactions: A Halogen Hierarchy
The difference in reactivity among the halogens leads to interesting phenomena like displacement reactions. In a displacement reaction involving halogens, a more reactive halogen can displace a less reactive halogen from its salt solution. For example, if you add chlorine water (an aqueous solution of chlorine) to a solution of potassium bromide (KBr), the chlorine will displace the bromide ions. This happens because chlorine is more reactive than bromine and can readily accept an electron from the bromide ion, turning it into bromine. The reaction would look like this:
Cl₂ (aq) + 2KBr (aq) → 2KCl (aq) + Br₂ (aq)
Conversely, if you add bromine water to potassium chloride, no reaction would occur because bromine is less reactive than chlorine and cannot displace the chloride ions.
This hierarchy of reactivity allows us to predict the outcome of reactions between halogens and halide ions:
- Fluorine can displace chlorine, bromine, and iodine.
- Chlorine can displace bromine and iodine.
- Bromine can displace iodine.
- Iodine cannot displace chlorine or bromine.
The Impact of Reactivity on Atomic Mass
While the reactivity of Group 7 elements is primarily dictated by their electron configuration, there isn't a direct causal link between their reactivity and their atomic mass in a way that would be easily quantifiable in a simple formula. However, as we've seen, atomic mass does increase down the group, and this increase in atomic size correlates with a *decrease* in reactivity. So, while atomic mass itself doesn't *cause* reactivity, the trends in atomic mass and size within the group reflect the underlying electronic factors that determine reactivity.

Frequently Asked Questions about Group 7
- Q1: Are all elements in Group 7 non-metals?
- A1: Yes, the elements in Group 7, the halogens, are all non-metals.
- Q2: What makes Group 7 elements so reactive?
- A2: They have seven electrons in their outer shell and a strong tendency to gain one more electron to achieve a stable electron configuration.
- Q3: Which halogen is the most reactive?
- A3: Fluorine is the most reactive halogen.
- Q4: What happens when a halogen reacts with a metal?
- A4: They react to form a metal halide salt, typically through ionic bonding where the halogen gains an electron from the metal.
- Q5: Can a less reactive halogen displace a more reactive halogen?
- A5: No, a more reactive halogen can displace a less reactive halogen from its salt solution, but not the other way around.
Conclusion
Group 7, the halogens, are a cornerstone of understanding chemical reactivity. Their propensity to gain electrons makes them highly reactive, especially with metals, forming the salts that give them their name. From the vigorous reactions of fluorine to the displacement capabilities of chlorine and bromine, the halogens showcase a fascinating interplay of electronic structure and chemical behaviour. Understanding these trends is fundamental for anyone delving into the world of chemistry, whether in a classroom setting or through personal exploration.
If you want to read more articles similar to Group 7: The Reactive Halogens, you can visit the Automotive category.
