07/10/2024
In the intricate world of petroleum refining, understanding how crude oil is transformed into the fuels and products we rely on daily is fundamental. At the heart of this transformation lies distillation, a process that separates crude oil into various components based on their boiling points. Central to this separation are what we call 'distillate cut points' – precise temperature markers that define the boundaries of each valuable fraction. Without these meticulously determined points, the refining process would be haphazard, and the quality and quantity of products like petrol, diesel, and aviation fuel would be unpredictable. This article will delve into what distillate cut points are, how they are determined, their vital role in calculating product yields, and the fascinating challenges and innovations in characterising increasingly complex crude oil types, particularly the heavier varieties.
- What Exactly Are Distillate Cut Points?
- Calculating Distillate Yields: A Crucial Metric
- The Evolving Landscape: Characterising Heavy Oils
- Beyond Conventional Distillation: The Role of Physical Properties
- Specific Gravity, H/C Ratio, and the Limitations with Heavy Fractions
- The Breakthrough: Deep Vacuum Fractionation Apparatus (DVFA)
- Why Accurate Characterisation Matters for the UK Automotive and Energy Sectors
- Frequently Asked Questions (FAQs)
- Conclusion
What Exactly Are Distillate Cut Points?
At its core, a distillate cut point is a specific temperature that represents either the lower or upper limit of a particular distillate fraction obtained from crude oil. Think of crude oil as a complex mixture of thousands of different hydrocarbons, each with its own unique boiling point. When crude oil is heated in a distillation column, these hydrocarbons vaporise at different temperatures and then condense at various levels within the column, effectively separating into different groups or 'fractions'.
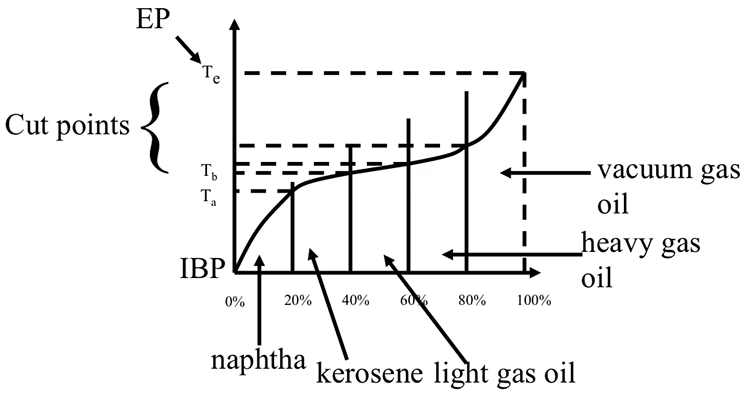
These fractions are not arbitrary; they are carefully defined by their boiling ranges. A TBP curve, or True Boiling Point curve, is an essential tool in this process. It plots the cumulative volume percentage of crude oil distilled against the corresponding boiling temperature. Using this curve, refiners can precisely identify the temperatures at which different fractions begin and end. For instance, consider kerosene: the TBP curve would show a specific temperature, say Ta, as its lower cut point, indicating when kerosene-range molecules begin to vaporise and condense. Similarly, a higher temperature, Tb, would represent its upper cut point, marking where the kerosene fraction ends and the next heavier fraction begins.
These boiling ranges between the cut points are what define commercially viable distillate products. Common examples include naphtha (used for petrol blending and petrochemicals), kerosene (for jet fuel and paraffin), light gas oil (for diesel and heating oil), and heavier fractions like fuel oil and asphalt. Each of these products has a specific set of desired properties, and their accurate separation relies entirely on the precise definition and control of these cut points.
Calculating Distillate Yields: A Crucial Metric
Beyond merely defining the fractions, distillate cut points are indispensable for calculating the 'yield' of each product. Yield refers to the volume percentage of a specific distillate fraction obtained from a given volume of crude oil. This calculation is straightforward yet incredibly significant for refinery economics and planning.
To determine the yield for a particular fraction, one simply subtracts the cumulative volume percentage at its lower cut point from the cumulative volume percentage at its upper cut point, as indicated on the TBP curve. Let's revisit our kerosene example from a hypothetical TBP curve. If the TBP curve shows that at the lower cut point (Ta), 20% of the crude oil by volume has distilled, and at the upper cut point (Tb), 40% of the crude oil by volume has distilled, then the yield for kerosene would be calculated as 40% (at Tb) - 20% (at Ta) = 20% by volume. This 20% represents the proportion of kerosene that can be extracted from that specific crude oil. Accurate yield calculations are vital for optimising refinery operations, forecasting product availability, and making informed decisions about crude oil purchasing.
The Evolving Landscape: Characterising Heavy Oils
The global demand for energy continues to rise steadily, while conventional, easily accessible crude oil reserves are depleting. This has driven a significant shift towards non-conventional oil sources, particularly heavy oil and oil sands. These resources represent a substantial extension of the world's energy reserves and are becoming increasingly important for energy security. However, their recovery and processing present unique and significant challenges compared to lighter, more conventional crude oils.
Traditionally, thermal production methods, such as steam injection, are employed to recover heavy oils from relatively shallow and cool deposits. While effective, these methods are highly energy-intensive and consume substantial amounts of water, raising both economic and environmental concerns. Consequently, there is a growing interest in using solvents, either alone or in conjunction with thermal methods, to recover heavy oils. Solvents can significantly reduce energy usage, water consumption, and the overall environmental impact. However, mixtures of solvent and heavy oil are notoriously challenging to model from a fluid dynamics perspective. They can form multiple liquid phases and are prone to asphaltene precipitation, which can cause severe operational issues.
To design and optimise these advanced recovery and processing methods, an exceptionally accurate phase behaviour model is required. The starting point for such a model is a far more rigorous characterisation of the heavy oil than is typically needed for conventional thermal processes. This brings us to the complexities of how specific gravity and other properties are used in characterisation.
Beyond Conventional Distillation: The Role of Physical Properties
The characterisation methodology for crude oils is commonly based on a distillation curve, obtained either through direct distillation in a laboratory or via simulated distillation techniques using chromatography. These distillation assays provide a boiling point distribution, which forms the bedrock for determining other crucial property distributions, such as molecular weight, density (from which specific gravity is derived), and critical properties. These additional properties are often determined through direct measurements or, more commonly, through empirical correlations.
Most conventional physical property correlations involve a combination of boiling point, specific gravity, and molecular weight. Often, one of these three properties can be estimated or determined from one or both of the others using these correlations. There are numerous such correlations, developed over decades, that have proven sufficient for predicting the phase behaviour of conventional oils in most petroleum-related applications.
Specific Gravity, H/C Ratio, and the Limitations with Heavy Fractions
While specific gravity is a vital input for many characterisation correlations, its direct relationship with the Hydrogen-to-Carbon (H/C) ratio of crude oil fractions is complex and indirect. The H/C ratio is fundamentally determined by the elemental composition and molecular structure of the hydrocarbons within a fraction. However, accurate specific gravity measurements are crucial for robust correlations that, in turn, help predict other properties, including those related to elemental composition.
For heavy oil and bitumen/solvent mixtures, applying these established characterisation approaches presents two significant challenges:
Limited Distillability: Only a small fraction of heavy oils is distillable using conventional methods. Most techniques can only fractionate up to about 30 weight percent (wt%) of a bitumen sample before reaching temperatures of 300 °C, even under vacuum conditions. This severely limits the ability to characterise the bulk of the fluid and introduces considerable uncertainty when attempting to extrapolate the distillation curve over the entire oil.

Cut points and definition of distillate products as boiling ranges. Boiling ranges between the cut points represent distillate products, such as naphtha, kerosene, light gas oil, etc. The difference between the cumulative volume percent at upper and lower cut points is reported as the yield (in volume %) for the particular distillate fraction. Lack of Data and Inadequate Correlations: Since conventional distillation methods struggle to collect heavy cuts, there is a severe scarcity of physical and thermal property data available for these heavier fractions. Currently, refiners are often forced to use correlations developed for conventional oils. The problem is, these correlations were developed from datasets primarily comprising light to medium oils and pure paraffinic components. For example, some widely used correlations were developed using North Sea oils and gas condensates with API gravities no lower than 25. Crucially, these correlations do not adequately account for the significantly higher aromaticity and heteroatom content characteristic of the heavier crude oil cuts. The H/C ratio is directly affected by aromaticity (more aromatic compounds tend to have lower H/C ratios). Therefore, correlations that don't account for this structural difference will be less accurate for heavy oils, even if they use specific gravity as an input.
The Breakthrough: Deep Vacuum Fractionation Apparatus (DVFA)
To overcome these formidable challenges, innovative technologies are being developed. One such advancement is the Deep Vacuum Fractionation Apparatus (DVFA). This apparatus, originally designed to measure the vapour pressure of compounds with very low volatility, has revolutionised the characterisation of heavy oils.
The DVFA operates at pressures significantly lower than most current vacuum techniques (as low as 1 × 10-9 kPa). A standardised procedure has been developed to batch distill heavy oils using this apparatus, extending the distillation range to approximately 50 wt% distilled, a substantial improvement over the typical 25 wt% achieved with conventional vacuum distillation. An interconversion method also allows for obtaining Atmospheric Equivalent Temperatures (AET) corresponding to the normal boiling point (NBP) of these distillation cuts.
Crucially, the DVFA provides physical samples of these heavy distillation fractions. This capability addresses the second major challenge: the dire lack of data and validated correlations for heavy fractions. With physical samples now available, researchers can directly measure key physical properties such as molecular weight and density (which directly relates to specific gravity). Furthermore, their elemental analysis (which includes the H/C ratio) can also be precisely measured. This new, high-quality data is invaluable for evaluating existing correlations and, more importantly, for modifying them or proposing entirely new correlations that accurately account for the unique properties of heavy fractions, including their distinct aromaticity and heteroatom content. This ensures that models using specific gravity, boiling point, and molecular weight are far more reliable for these challenging crude sources.
Why Accurate Characterisation Matters for the UK Automotive and Energy Sectors
For the UK, a nation heavily reliant on imported crude oil and with a sophisticated refining industry, accurate crude oil characterisation is paramount. Precise determination of distillate cut points and the rigorous characterisation of all crude fractions, especially the heavier ones, directly impacts the efficiency of refineries, the quality of refined products (like petrol and diesel for the automotive sector), and the overall profitability of the energy sector. By adopting advanced techniques like the DVFA, UK refiners can better process diverse crude feedstocks, maximise the yield of high-value products, and reduce waste, contributing to greater energy security and environmental responsibility.
Frequently Asked Questions (FAQs)
What is a TBP curve?
A True Boiling Point (TBP) curve is a laboratory distillation curve that plots the cumulative volume percentage of crude oil distilled against its corresponding boiling temperature. It's considered the most accurate representation of a crude oil's boiling range distribution and is fundamental for defining distillate cut points.
Why are distillate cut points important?
Cut points are crucial because they define the precise temperature ranges for different valuable product fractions (e.g., naphtha, kerosene, diesel) within crude oil. They enable refiners to separate crude oil efficiently, calculate the exact yield of each product, and ensure that products meet specific quality standards required for various applications.
What are common distillate fractions?
Common distillate fractions include naphtha (used for petrol and petrochemicals), kerosene (for jet fuel and heating), light gas oil (for diesel and heating oil), and heavier gas oils. Each has a distinct boiling range defined by cut points.
Why is characterising heavy oil so difficult?
Heavy oils are challenging to characterise due to their high viscosity, complex molecular structures (high aromaticity and heteroatom content), and the fact that only a small portion of them can be distilled using conventional laboratory methods. This makes it difficult to obtain representative samples for property measurements and to apply existing correlations accurately.
How does the Deep Vacuum Fractionation Apparatus (DVFA) help refine heavy oils?
The DVFA operates at extremely low pressures, allowing for the distillation of much heavier fractions of crude oil than previously possible. Crucially, it provides physical samples of these heavy fractions, enabling direct measurement of their molecular weight, density, and elemental composition (including H/C ratio). This data is vital for developing more accurate characterisation models and correlations for heavy oils.
Does specific gravity directly determine the H/C ratio?
No, specific gravity does not directly determine the H/C ratio. Specific gravity is a measure of density, while the H/C ratio is derived from the elemental composition of the hydrocarbons. However, both are crucial properties used in correlations to characterise crude oil fractions. The challenge with heavy oils is that conventional correlations (which use specific gravity) were developed for lighter oils and don't adequately account for the higher aromaticity (and thus, different H/C ratios) of heavier cuts. The DVFA helps by providing samples for direct elemental analysis, which in turn helps develop better correlations that incorporate these unique characteristics.
Conclusion
Distillate cut points are more than just temperatures on a graph; they are the fundamental building blocks of crude oil refining, dictating the separation, quality, and quantity of every valuable product derived from crude. As the world transitions towards more challenging crude oil sources like heavy oils, the precision demanded from characterisation techniques becomes even greater. Innovations like the Deep Vacuum Fractionation Apparatus are not just technological marvels; they are essential tools that enable the industry to unlock the full potential of these complex feedstocks, ensuring a more efficient, sustainable, and secure energy future for the UK and beyond. Understanding these intricate processes reinforces the sophistication and continuous evolution of the automotive and energy industries that power our lives.
If you want to read more articles similar to Crude Oil Distillation: Mastering Distillate Cut Points, you can visit the Automotive category.
