16/04/2015
Understanding the Atom: A Fundamental Exploration
The atom, a concept that has captivated scientists for centuries, represents the fundamental unit of a chemical element. It's the smallest particle of an element that retains its chemical identity. While the idea of indivisible particles dates back to ancient Greece, our modern understanding of the atom is a testament to scientific curiosity and relentless experimentation. This article will delve into how we represent an atom, its core components, and the historical evolution of our comprehension of these essential building blocks of the universe.
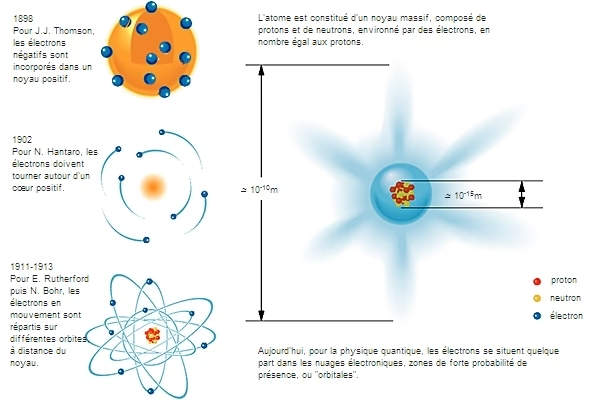
The Atomic Nucleus: The Heart of the Matter
While quantum mechanics provided rapid explanations for atomic and molecular spectroscopic characteristics, the core of the atom, its nucleus, proved more challenging to comprehend. The difficulties were twofold: firstly, the immense energy of probe particles required to reach dimensions on the order of a femtometre (10-15 m), and secondly, the necessity to invent at least one additional interaction to explain the stability of a nucleus composed of protons (which repel each other electrically) and neutrons. This understanding of nuclear cohesion also needed to explain phenomena like alpha, beta, and gamma radioactivity, first observed in the last decade of the 19th century.
The decade preceding the Second World War saw the discovery of the two key interactions governing the stability of the nucleus: the strong nuclear force and the weak nuclear force. The short range of these two interactions, 10-15 m and 10-18 m respectively, explains the experimental difficulties encountered. Theoretical challenges were also abundant; these were not physical laws as simple as those of electromagnetism, even when complicated by quantum mechanics, but rather a profound understanding of all elementary particles. The invention of quarks and gluons thus provides our current view of the interaction that holds nucleons together. This nuclear physics also leads to the explanation of nucleosynthesis, accounting for the nuclear aspects of the periodic table. We find ourselves here amidst the effervescence of the universe's birth and stellar dynamics.
Representing an Atom: Notation and Symbols
An atom is commonly designated by its chemical symbol. To provide more specific information, this symbol is often augmented with its mass number, denoted by 'A' (the total number of nucleons – protons and neutrons – in the atom), placed as a superscript to the left of the symbol. For instance, carbon-12, with a mass number of 12, is written as 12C.
It is also customary to include the atomic number, denoted by 'Z' (the number of protons in the nucleus, which defines the element), as a subscript to the left of the symbol. This is particularly useful when describing nuclear reactions involving isotopes. Therefore, carbon-12 is formally written as 612C. Using this notation, carbon-14 (614C) and carbon-12 (612C) are clearly identified as two different isotopes of carbon. Isotopes are atoms of the same element (meaning they have the same number of protons) but differ in their number of neutrons, and consequently, their mass number.
The Structure of an Atom: A Closer Look
At its simplest, an atom consists of a central nucleus surrounded by a cloud of electrons. The nucleus itself is composed of positively charged protons and electrically neutral neutrons, collectively known as nucleons. The number of protons (Z) dictates the element's identity. For example, every atom with one proton is hydrogen, and every atom with six protons is carbon.
The electrons, negatively charged, orbit the nucleus in specific energy levels or shells. The arrangement of these electrons, particularly the outermost ones (valence electrons), determines an atom's chemical behaviour and its ability to form bonds with other atoms. The electrostatic attraction between the positively charged protons in the nucleus and the negatively charged electrons holds the atom together.
Key Components:
- Protons: Positively charged particles found in the nucleus. They determine the atomic number (Z).
- Neutrons: Neutrally charged particles found in the nucleus. They contribute to the atom's mass number (A) along with protons.
- Electrons: Negatively charged particles that orbit the nucleus in specific energy levels.
A Brief History of Atomic Models
Our understanding of atomic structure has evolved significantly over time, with each model building upon the discoveries of its predecessors:
| Model | Scientist | Key Features | Analogy |
|---|---|---|---|
| Dalton's Atomic Theory | John Dalton | Atoms are indivisible, indestructible spheres. Atoms of the same element are identical. Atoms of different elements have different properties. | Billiard Ball |
| Plum Pudding Model | J.J. Thomson | A positively charged sphere with negatively charged electrons embedded within it. | Plum Pudding / Blueberry Muffin |
| Rutherford Model | Ernest Rutherford | A dense, positively charged nucleus at the centre, with electrons orbiting it. Most of the atom is empty space. | Nuclear Model / Solar System |
| Bohr Model | Niels Bohr | Electrons orbit the nucleus in specific, fixed energy levels or shells. Electrons can jump between these levels by absorbing or emitting energy. | Planetary Model with specific orbits |
| Quantum Mechanical Model | Schrödinger, Heisenberg, etc. | Electrons exist in probability clouds or orbitals, not fixed orbits. The exact position and momentum of an electron cannot be known simultaneously (Heisenberg's Uncertainty Principle). | Electron Cloud / Probability Distribution |
Properties and Classification of Atoms
The properties of an atom are primarily determined by its atomic number (number of protons) and the arrangement of its electrons. The atomic number (Z) classifies an element. For instance, all elements with Z=1 are hydrogen, Z=2 are helium, and so on. This forms the basis of the periodic table, where elements are arranged in order of increasing atomic number and grouped by similar chemical properties, which are largely dictated by their electron configurations.
The number of neutrons can vary, leading to isotopes. Isotopes of an element have the same chemical properties but may differ in physical properties such as mass and radioactivity. For example, Uranium-235 (235U) and Uranium-238 (238U) are isotopes of uranium. While chemically very similar, their nuclear stability and behaviour in nuclear reactions differ significantly.
Frequently Asked Questions (FAQs)
What is the smallest particle of an element?
The smallest particle of an element that retains the chemical properties of that element is an atom.
What are the main parts of an atom?
An atom consists of a central nucleus, containing protons and neutrons, and electrons that orbit the nucleus in specific energy levels.
What determines the element an atom belongs to?
The number of protons in the nucleus, known as the atomic number (Z), determines the element.
What is an isotope?
An isotope is an atom of an element that has the same number of protons but a different number of neutrons compared to other atoms of the same element.
Why is the nucleus stable despite having positively charged protons?
The nucleus is held together by the strong nuclear force, an incredibly powerful short-range attraction that overcomes the electrostatic repulsion between protons.
In conclusion, the atom, though incredibly small, is a complex entity whose structure and behaviour have been deciphered through centuries of scientific inquiry. From the early concept of indivisible spheres to the sophisticated quantum mechanical model, our understanding continues to evolve, revealing the intricate dance of particles that forms the very fabric of reality.
If you want to read more articles similar to The Atom: A Building Block Explained, you can visit the Automotive category.
