01/03/2023
As dedicated professionals in vehicle maintenance, we spend a significant amount of time under the bonnet, diagnosing issues and ensuring our clients' cars run efficiently and cleanly. While much of our work focuses on mechanical components and electrical systems, there's a fascinating, often overlooked, microscopic world at play within our engines and exhaust systems: the realm of molecular chemistry. Understanding the fundamental behaviour of gases like carbon monoxide and nitrogen oxides, even at a theoretical level, can provide deeper insights into combustion efficiency, emission control, and the very air quality we strive to protect. It's about moving beyond simply 'fixing' a problem to truly comprehending its origins.
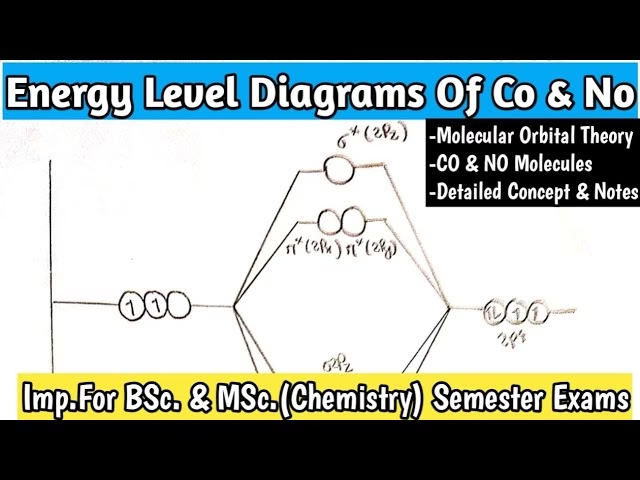
Carbon Monoxide (CO) – A Closer Look at a Common Culprit
Carbon monoxide (CO) is a notoriously dangerous gas, colourless, odourless, and a silent killer. In the automotive world, it's a direct byproduct of incomplete combustion – when there isn't enough oxygen to fully oxidise the fuel in your engine. High CO emissions often point to issues like a rich fuel mixture, faulty oxygen sensors, or a malfunctioning catalytic converter. But what makes CO behave the way it does? And why would something as complex as a Molecular Orbital (MO) diagram be relevant to this common exhaust gas?
For the everyday mechanic, directly interpreting a complex molecular orbital diagram of carbon monoxide isn't a routine task. In that immediate, practical sense, one might even say its direct importance is 'none'. However, this perspective would be incredibly short-sighted for anyone involved in the design, development, or deep diagnostic analysis of automotive systems. Molecular Orbital theory is a cornerstone of modern chemistry, providing a sophisticated framework for understanding how atoms bond together to form molecules and, crucially, predicting their properties and reactivity.
An MO diagram illustrates the energy levels of a molecule's orbitals and how electrons occupy them. For CO, such a diagram reveals the strong triple bond between carbon and oxygen, explaining its remarkable stability and the relatively high energy required to break it down. It also elucidates why CO has a strong affinity for haemoglobin in our blood, leading to its toxicity. While you, as a mechanic, won't be sketching MO diagrams during a service, the principles derived from them guide the engineers who design catalytic converters to efficiently break down CO into less harmful carbon dioxide. They inform the scientists developing new fuel additives or combustion strategies to minimise CO formation. So, while not a direct tool for a spanner-wielding professional, the underlying principles are foundational to the very technology we service.
Nitrogen Oxides (NOx) – Ions and Their Impact
Beyond carbon monoxide, another significant family of pollutants from vehicle exhausts are the Nitrogen Oxides, often collectively referred to as NOx. These gases, primarily nitric oxide (NO) and nitrogen dioxide (NO2), contribute to smog, acid rain, and respiratory problems. The formation of NOx occurs at high temperatures within the engine cylinders, where nitrogen and oxygen from the air react. But did you know that these molecules can exist in different ionic forms, such as NO+ and NO–, each with distinct properties? Understanding these subtle differences, again, delves into the molecular realm but has significant implications for how these pollutants behave and how we might mitigate them.
Unpacking NO+ and NO–: A Comparative Analysis
The question of 'What is the difference between NO+ and NO–?' might seem academic, but it touches upon the very nature of chemical bonding and molecular behaviour. Both NO+ and NO– are molecular ions derived from the neutral nitric oxide (NO) molecule, formed by either the loss or gain of electrons in their outermost atomic orbitals. These small changes in electron count lead to profound differences in their stability, reactivity, and magnetic properties.
Let's break down the key distinctions:
Formation and Electron Count:
- NO+ (Nitrosonium ion): Formed when the neutral NO molecule loses an electron. This loss occurs from one of its valence molecular orbitals. With fewer electrons, its electronic configuration changes, impacting its bonding.
- NO– (Nitroxyl anion): Formed when the neutral NO molecule gains an electron. This additional electron occupies a higher energy molecular orbital, also altering its electronic structure.
Bond Order:
The concept of bond order is crucial here. It's a measure of the number of chemical bonds between two atoms and indicates the stability of a molecule. A higher bond order generally means a stronger, shorter, and more stable bond.
- NO–: Has a bond order of 2. This suggests a double bond character, making it less stable compared to neutral NO.
- NO (Neutral): Has a bond order of 2.5. This fractional bond order is characteristic of molecules with unpaired electrons in bonding or anti-bonding orbitals.
- NO+: Boasts a bond order of 3. This indicates a very strong triple bond, making it the most stable of the three species.
This ascending pattern – NO– < NO < NO+ – in bond order (2, 2.5, and 3 respectively) directly correlates with their stability and reactivity. The more stable NO+ is less likely to react readily, while the less stable NO– might be more reactive.
Magnetic Properties: Paramagnetism vs. Diamagnetism
Another fascinating distinction lies in their magnetic properties, which are determined by the presence or absence of unpaired electrons in their molecular orbitals.
- NO– (Nitroxyl anion): This species is paramagnetic. This means it possesses unpaired electrons and is weakly attracted to an external magnetic field. The presence of these unpaired electrons often makes molecules more reactive.
- NO+ (Nitrosonium ion): In contrast, NO+ is a diamagnetic molecular species. All of its electrons are paired, meaning it is weakly repelled by a magnetic field. Diamagnetic molecules are generally more stable and less reactive than their paramagnetic counterparts.
Here's a quick summary of the differences:
| Property | NO– (Nitroxyl Anion) | NO (Neutral Nitric Oxide) | NO+ (Nitrosonium Ion) |
|---|---|---|---|
| Electron Change | Gains 1 electron | Neutral | Loses 1 electron |
| Bond Order | 2 (Double Bond Character) | 2.5 (Intermediate) | 3 (Triple Bond Character) |
| Stability Relative to Others | Least Stable | Intermediate Stability | Most Stable |
| Magnetic Property | Paramagnetic (Unpaired electrons) | Paramagnetic (Unpaired electron) | Diamagnetic (All electrons paired) |
| Reactivity | More Reactive | Intermediate Reactivity | Less Reactive |
Why Does This Molecular Detail Matter for Mechanics?
At first glance, delving into bond orders and magnetic properties might seem far removed from replacing a catalytic converter or tuning an engine. However, these microscopic insights are fundamental to the macro-level systems we work with daily. Engineers designing advanced engine control units (ECUs) and catalytic converters rely heavily on such chemical principles. For example:
Catalytic Converter Efficiency: The effectiveness of a catalytic converter in reducing NOx emissions depends on its ability to facilitate reactions that convert harmful nitrogen oxides into harmless nitrogen and oxygen. Understanding the reactivity of different NOx species (including transient ions like NO+ and NO– that might form during reaction pathways) is crucial for selecting the right catalyst materials and optimising their operating conditions. A catalyst must be designed to handle various forms of nitrogen oxides generated during combustion.
Sensor Development: Modern vehicles are equipped with sophisticated sensors that monitor exhaust gases. While direct detection of NO+ or NO– might not be common, the underlying principles of how these gases interact with sensor materials are rooted in their electronic and magnetic properties. Knowing whether a gas is paramagnetic or diamagnetic, or how strong its bonds are, influences how it might be adsorbed onto a sensor surface or how it might react to generate an electrical signal.
 NO + and NO – are molecular ions formed by the loss or gain of electrons in the valence shell atomic orbitals of constituent atoms. The bond order follows the ascending pattern: NO – < NO < NO + e., 2, 2.5, and 3 respectively. NO – is paramagnetic while NO + is a diamagnetic molecular species.[/caption]
NO + and NO – are molecular ions formed by the loss or gain of electrons in the valence shell atomic orbitals of constituent atoms. The bond order follows the ascending pattern: NO – < NO < NO + e., 2, 2.5, and 3 respectively. NO – is paramagnetic while NO + is a diamagnetic molecular species.[/caption]
Combustion Optimisation: Engine designers continuously strive for more efficient and cleaner combustion. This involves complex modelling of chemical reactions occurring within the cylinders. The stability and reactivity of various species, including the formation of different nitrogen oxides, are critical inputs for these models. By understanding the conditions under which NO, NO+, or NO– are formed and their subsequent fates, engineers can fine-tune parameters like fuel injection timing, ignition timing, and exhaust gas recirculation (EGR) to minimise the production of harmful pollutants.
Future Automotive Technologies: As the automotive industry moves towards stricter emission standards and alternative fuels, the need for deep chemical understanding will only intensify. Whether it's developing new selective catalytic reduction (SCR) systems or exploring novel combustion concepts, the fundamental properties of molecules and ions will remain at the forefront of innovation. For the forward-thinking mechanic, having even a conceptual grasp of these principles equips them to better understand the rationale behind new technologies and diagnose complex issues that transcend simple mechanical failures.
Frequently Asked Questions (FAQs)
Here are some common questions that might arise when considering the molecular side of automotive emissions:
Q: Are MO diagrams used in everyday car diagnostics?
A: No, not directly. MO diagrams are highly theoretical tools used by chemists and engineers during the research, development, and design phases of engines, fuels, and emission control systems. A mechanic's diagnostic tools focus on macroscopic measurements like sensor readings, fault codes, and exhaust gas analysis using specialised equipment.
Q: How do NO+ and NO– actually form in an engine?
A: While the primary nitrogen oxide formed in an engine is neutral NO (nitric oxide) and then NO2 (nitrogen dioxide), the extreme temperatures and pressures during combustion can lead to the transient formation of various ionic species. These ions are typically short-lived intermediates in complex reaction pathways. Their presence, even momentary, can influence the overall chemistry of the exhaust gases.
Q: Does the paramagnetism of NO– affect its detection?
A: Yes, in highly specialised laboratory settings, paramagnetism can be used to detect and quantify substances. In automotive applications, exhaust gas analysers typically use infrared (IR) or electrochemical sensors, which detect gases based on their absorption of specific wavelengths of light or their electrochemical reactivity, respectively. While the underlying molecular properties (like electron configuration) influence these detection methods, the paramagnetism itself isn't a direct measurement technique for a standard exhaust analyser.
Q: Why is carbon monoxide so dangerous, and how does its MO diagram explain this?
A: Carbon monoxide is dangerous because it binds to the haemoglobin in your blood much more readily and strongly than oxygen does, forming carboxyhaemoglobin. This prevents oxygen from being transported effectively throughout the body. While the MO diagram itself isn't a direct explanation for the mechanic, it reveals the electronic structure of CO, showing its lone pair of electrons on the carbon atom that makes it an excellent ligand for metal centres like the iron in haemoglobin. This strong binding capability is a direct consequence of its molecular orbital configuration.
Q: What is 'bond order' in simple terms?
A: In simple terms, bond order is a measure of how many chemical bonds exist between two atoms. For example, a single bond has a bond order of 1, a double bond has a bond order of 2, and a triple bond has a bond order of 3. A higher bond order generally means the bond is stronger and the molecule is more stable. In some cases, like neutral NO, molecules can have fractional bond orders, indicating a blend of bonding characteristics.
Conclusion
While our daily work as mechanics focuses on tangible components and measurable performance, appreciating the intricate molecular dance within an engine and its exhaust system adds a profound layer to our understanding. The theoretical concepts of molecular orbital diagrams, bond order, and magnetic properties, though seemingly abstract, are the very bedrock upon which modern automotive technology is built. By grasping these underlying principles, even conceptually, we not only enhance our professional knowledge but also contribute more effectively to the ongoing efforts for cleaner, more efficient, and environmentally responsible vehicle operation. It's about recognising that the smallest particles can have the biggest impact.
If you want to read more articles similar to Unravelling Exhaust Gases: A Molecular View, you can visit the Emissions category.
