30/12/2023
The drive for enhanced fuel efficiency and reduced environmental impact in internal combustion engines has placed a significant spotlight on the intricacies of fuel injection. As engines become more sophisticated and operate under increasingly demanding conditions, understanding the behaviour of fuel sprays under supercritical pressure is paramount. This phenomenon, where fuel and oxidiser properties exceed their critical points, fundamentally alters the injection process, leading to unique mixing characteristics that can profoundly influence combustion efficiency and emission levels.
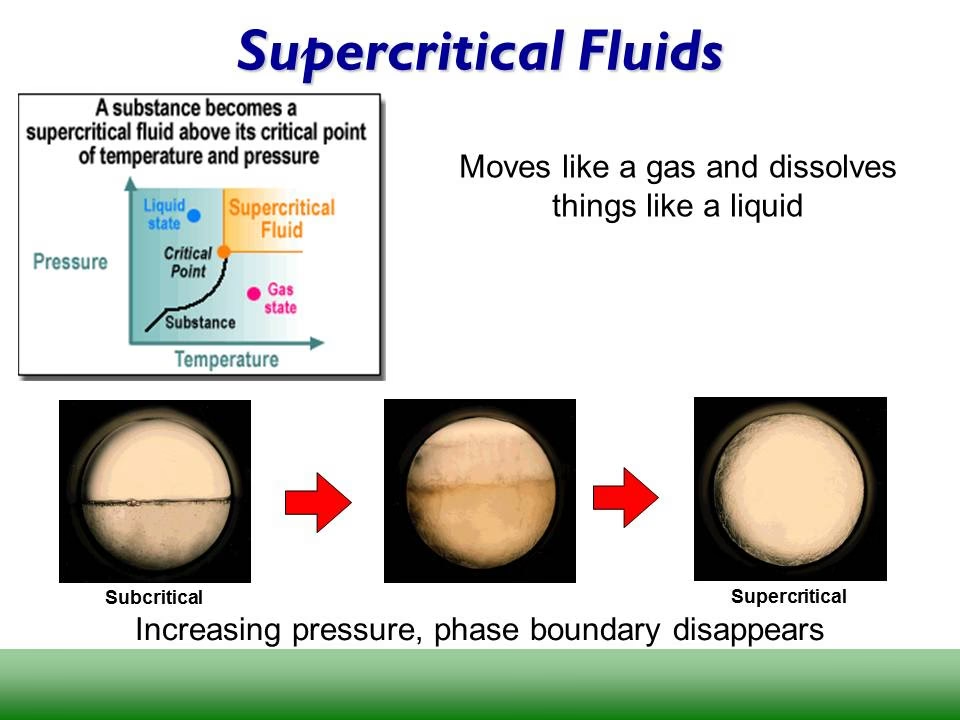
Understanding Supercritical Conditions
In the realm of fluid dynamics, the critical point represents a unique state where a substance can exist as either a liquid or a gas without a distinct phase boundary. When hydrocarbon fuels are injected into an environment with temperatures and pressures exceeding this critical point, they enter a supercritical state. Unlike traditional subcritical injection, where distinct liquid droplets exist and evaporation is a primary mechanism, supercritical injection presents a different scenario. The surface tension and latent heat of evaporation, crucial factors in subcritical sprays, become negligible. The clear interface between liquid and gas phases dissolves, giving way to a broad mixing layer. This transformation means that supercritical fuel sprays behave more like a gas jet, with their properties governed by factors like density gradients and intermolecular interactions.
The Science Behind Supercritical Injection
Research into supercritical fuel injection reveals several key characteristics that differentiate it from conventional methods:
- Phase Boundary Dissolution: The most striking feature is the absence of a distinct liquid-gas interface. This eliminates the phenomenon of boiling, with the fuel transitioning smoothly into a supercritical fluid.
- Enhanced Mixing: The lack of surface tension and the presence of a diffuse mixing layer promote rapid and efficient mixing between the fuel and the surrounding oxidiser (typically air). This improved mixing is a significant factor in achieving more complete combustion.
- Density Gradient Influence: As temperature and pressure increase, the density gradient within the jet diminishes. This causes the jet to behave more like a turbulent gas jet, influencing its penetration and spread characteristics.
- Ignition and Combustion: Supercritical sprays have demonstrated more uniform, faster, and cleaner combustion compared to traditional droplet sprays. This leads to higher combustion efficiency and a reduction in harmful emissions.
Experimental Investigations and Key Findings
Numerous experimental studies have shed light on the complex behaviour of supercritical fuel sprays:
Researchers like Branam et al. [15] have shown that temperature variations play a critical role in dictating jet dissipation and rupture characteristics. Wensing et al. [16] experimentally confirmed that fuel injected under high pressure and temperature conditions transitions to a supercritical state without boiling or significant density changes. Liu et al. [17], using advanced imaging techniques such as Backlit Shadowgraph and Schlieren, visualised these sprays, observing their resemblance to gas jets. Roy et al. [18] noted that as temperature and pressure rise, the density gradient decreases, further reinforcing the gas-like behaviour of the jet. Anitescu et al. [19] found that injecting heated diesel fuel under supercritical conditions improved mixing efficiency with air. Poursadegh et al. [20] specifically investigated conditions that either promote or inhibit fuel droplet formation in supercritical hydrocarbon fuel injection. Liu et al. [21] used Phase Doppler Particle Analysis (PDPA) to demonstrate that increasing fuel temperature in Gasoline Direct Injection (GDI) engines enhances mixing quality. Rehman et al. [26] provided a direct comparison, concluding that supercritical spray combustion is superior in uniformity, speed, and cleanliness, leading to significant improvements in combustion efficiency and emission reduction.
Numerical Modelling and Equations of State
While experimental studies provide valuable insights, numerical simulations are crucial for quantifying the complex multi-variable interactions present in supercritical sprays. The accuracy of these simulations heavily relies on the chosen equations of state (EOS), which describe the thermodynamic properties of fluids.
Early numerical analyses, such as those by Oefelein et al. [28, 29] on n-heptane, highlighted that the atomisation mechanisms in supercritical states differ significantly from conventional ones, necessitating the consideration of intermolecular forces. Zhang et al. [30] observed anisotropic velocity fluctuations in supercritical sprays across a wide density range. Wei et al. [31] identified the pseudo-boiling point as a region where local isobaric heat capacity peaks, correlating with minimum thermal conductivity and viscosity, which in turn influences the density gradient.
The choice of EOS is critical. Simple cubic EOS like Peng-Robinson (PR) and Soave-Redlich-Kwong (SRK) have been widely used. Schmitt et al. [32], using the PR EOS, observed a long, dense core region followed by a brief turbulent transition phase in cryogenic nitrogen injection. Petit et al. [33] validated simulation methods against experimental data, finding the SRK equation with the Smagorinsky subgrid-scale model to be more consistent. Park et al. [35] further emphasised that the selection of an appropriate real fluid model is often more critical than the turbulence model itself for supercritical flows. Zhu et al. [36] underscored the importance of EOS accuracy using the RK-PR equation for n-dodecane simulations. Garg et al. [37] demonstrated that the ideal gas law is inadequate for representing supercritical flow phenomena, with the SRK equation providing a better match to experimental combustion data.
Challenges and Future Directions
Despite significant progress, several challenges remain in fully understanding and harnessing supercritical fuel injection:
- Trade-off Phenomena: As observed by Xia et al. [12] and Zhang et al. [27], there are intricate trade-offs between various parameters like injection pressure, ambient pressure, fuel critical properties, and ambient temperature that influence spray behaviour. A deeper understanding of these trade-offs is needed to optimise engine design.
- Equation of State Accuracy: While PR and SRK equations are useful, they can lack accuracy, especially in the transcritical region. More precise EOS, such as the Modified Benedict-Webb-Rubin (BWRS) equation, are crucial for accurate modelling. However, the computational complexity of BWRS has limited its widespread adoption.
- Quantifying Mixing: The difficulty in precisely quantifying the mixing characteristics of multi-variable supercritical sprays through experiments necessitates more advanced and accurate numerical simulations.
Future research must focus on developing and applying higher-precision equations of state to accurately capture the behaviour of supercritical jets. Systematic studies across a wide range of pressures and temperatures, utilising advanced simulation techniques like Large Eddy Simulation (LES) combined with accurate EOS, are essential. This will not only provide a deeper explanation for the observed trade-off phenomena but also pave the way for the development of innovative multicomponent hybrid fuels and significantly improve the combustion efficiency of spray systems.
Frequently Asked Questions
What is the critical point of a fuel?
The critical point is a specific temperature and pressure at which a substance can no longer exist as a distinct liquid or gas. Above this point, the substance exists as a supercritical fluid.
How does supercritical injection differ from subcritical injection?
In supercritical injection, there is no distinct liquid-gas interface, and phenomena like surface tension and latent heat of evaporation are negligible. The fuel behaves more like a gas, leading to enhanced mixing compared to subcritical injection where distinct droplets evaporate.
Why is the equation of state important for supercritical simulations?
The equation of state defines the thermodynamic properties of the fluid. For supercritical fluids, where properties change dramatically, an accurate equation of state is crucial for reliably predicting the behaviour of the spray, including its mixing, penetration, and atomisation characteristics.
What are the benefits of supercritical fuel injection?
Supercritical injection offers improved mixing, leading to faster, more uniform, and cleaner combustion. This results in higher combustion efficiency and reduced harmful emissions, making it a promising technology for future engine development.
What are the main challenges in supercritical fuel research?
Key challenges include understanding complex trade-off phenomena between different operating parameters, the need for highly accurate equations of state for numerical simulations, and the difficulty in experimentally quantifying mixing characteristics due to the absence of clear phase boundaries.
If you want to read more articles similar to Supercritical Fuel Spray: A Deep Dive, you can visit the Automotive category.
