03/02/2008
The molecular structure of a chemical species is fundamental to understanding its properties, reactivity, and behaviour. For diatomic species, this often involves a deep dive into their electronic configurations, typically elucidated through molecular orbital (MO) theory. The cyanogen cation, CN+, is one such fascinating diatomic molecule whose structure and bonding can be effectively explained by constructing its molecular orbital diagram. Composed of a carbon atom and a nitrogen atom, with an overall positive charge, CN+ presents a compelling case study in chemical bonding.
Understanding the Building Blocks: Atomic Orbitals
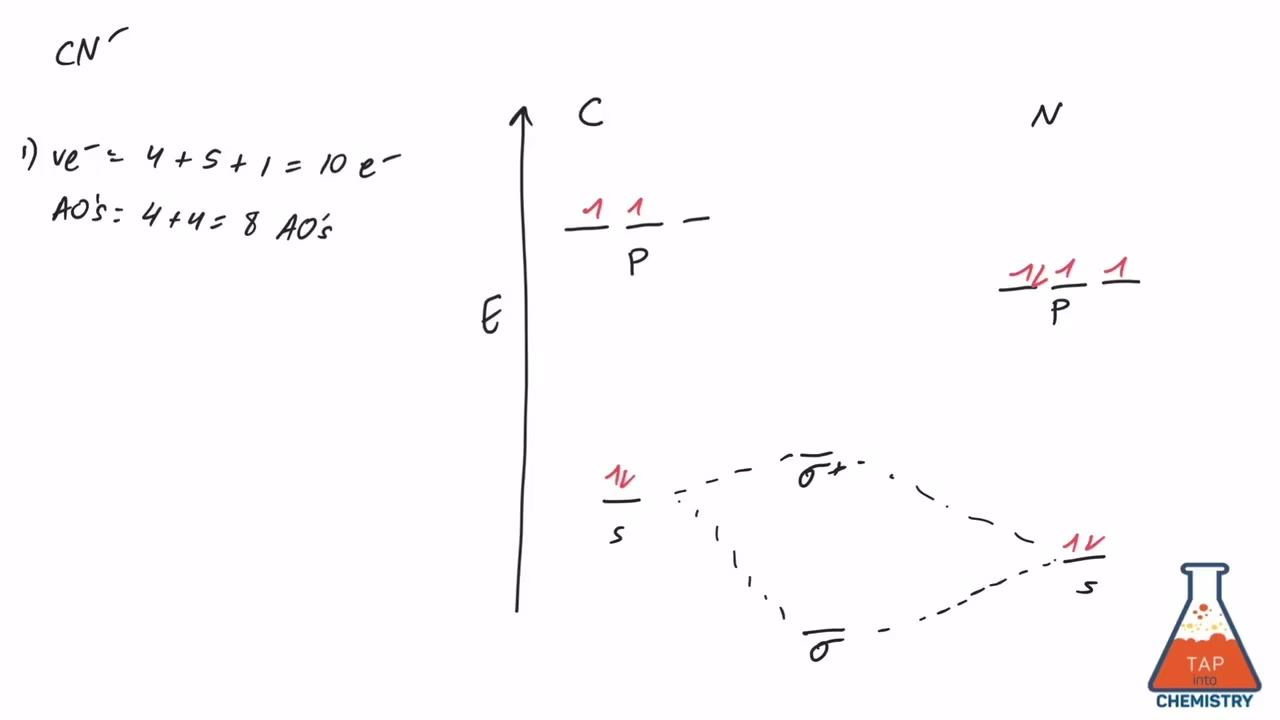
To construct the molecular orbital diagram for CN+, we first need to consider the atomic orbitals of the constituent atoms: carbon (C) and nitrogen (N). Both are second-period elements, meaning their valence electrons reside in the 2s and 2p atomic orbitals.
- Carbon (C): Atomic number 6. Electronic configuration: 1s²2s²2p². The valence orbitals are 2s and 2p.
- Nitrogen (N): Atomic number 7. Electronic configuration: 1s²2s²2p³. The valence orbitals are 2s and 2p.
Forming the Cation: The Charge Effect
The designation CN+ indicates that the molecule has lost one electron compared to a neutral cyanogen molecule (CN). This loss of an electron influences the overall electron count and, consequently, the filling of the molecular orbitals. A neutral CN molecule would have 6 (from C) + 7 (from N) = 13 valence electrons. Therefore, CN+ has 12 valence electrons.
Constructing the Molecular Orbital Diagram
Molecular orbital theory describes how atomic orbitals combine to form molecular orbitals, which are delocalised over the entire molecule. For diatomic molecules like CN+, the 2s and 2p atomic orbitals from each atom combine to form sigma (σ) and pi (π) molecular orbitals. The general order of filling for diatomic molecules of second-period elements (with the exception of O₂ and F₂) is:
σ2s, σ*2s, π2p, σ2p, π*2p, σ*2p
The combination of the 2s atomic orbitals from carbon and nitrogen leads to the formation of a bonding σ2s molecular orbital and an antibonding σ*2s molecular orbital. Similarly, the combination of the 2p atomic orbitals results in the formation of sigma (σ2p) and pi (π2p) bonding molecular orbitals, and their corresponding antibonding counterparts (σ*2p and π*2p).
Valence Electron Configuration of CN+
With 12 valence electrons to distribute, we fill the molecular orbitals in order of increasing energy:
- σ2s: 2 electrons
- σ*2s: 2 electrons
- π2p: 4 electrons (two degenerate π orbitals, each accommodating 2 electrons)
- σ2p: 2 electrons
- π*2p: 0 electrons
- σ*2p: 0 electrons
Thus, the valence electron configuration of CN+ is: (σ2s)²(σ*2s)²(π2p)⁴(σ2p)².
Bond Order and Stability
The bond order is a crucial indicator of the strength and stability of a chemical bond. It is calculated using the formula:
Bond Order = ½ (Number of electrons in bonding MOs - Number of electrons in antibonding MOs)
For CN+:
- Bonding electrons: 2 (in σ2s) + 4 (in π2p) + 2 (in σ2p) = 8
- Antibonding electrons: 2 (in σ*2s) + 0 (in π*2p) + 0 (in σ*2p) = 2
Bond Order = ½ (8 - 2) = ½ (6) = 3
A bond order of 3 suggests a triple bond between the carbon and nitrogen atoms. This high bond order indicates a very stable molecule, consistent with the presence of a positive charge which often stabilises species with higher bond orders.
Molecular Geometry: Linearity is Key
The molecular structure of CN+ is linear. This linearity can be inferred from the electronic configuration and the nature of the bonding molecular orbitals. In diatomic molecules, the concept of molecular geometry as understood in polyatomic molecules (like VSEPR theory) doesn't directly apply in the same way. However, the electron distribution in the molecular orbitals dictates the spatial arrangement. The formation of a triple bond, involving sigma and pi interactions, inherently leads to a linear arrangement of the atoms.
The σ2p molecular orbital is formed by the end-on overlap of the p orbitals, resulting in a cylindrical electron distribution along the internuclear axis. The π2p molecular orbitals are formed by the side-by-side overlap of the p orbitals, and these are oriented perpendicular to the internuclear axis. The strong triple bond character, with electron density concentrated along the internuclear axis, reinforces the linear geometry.
Comparing CN+ with related species like CN (cyanide radical) and CN⁻ (cyanide anion) can further illuminate its bonding and stability.
| Species | Valence Electrons | Electron Configuration (Valence) | Bond Order | Bond Length (pm) |
|---|---|---|---|---|
| CN⁻ | 14 | (σ2s)²(σ*2s)²(π2p)⁴(σ2p)²(π*2p)² | 3 | 119.7 |
| CN | 13 | (σ2s)²(σ*2s)²(π2p)⁴(σ2p)²(π*2p)¹ | 2.5 | 117.3 |
| CN⁺ | 12 | (σ2s)²(σ*2s)²(π2p)⁴(σ2p)² | 3 | 113.8 |
As seen from the table, CN+ has the highest bond order (3), which correlates with the shortest bond length. This observation further supports the stability of the CN+ cation due to its strong triple bond. The addition of an electron to form CN radical, and then another to form CN⁻, gradually weakens the bond and increases the bond length.
Key Takeaways
- CN+ is a diatomic cation with a linear structure.
- Its molecular orbital configuration is (σ2s)²(σ*2s)²(π2p)⁴(σ2p)².
- It possesses a bond order of 3, indicating a strong triple bond between carbon and nitrogen.
- The stability of CN+ is attributed to this strong triple bond.
Frequently Asked Questions (FAQs)
- Is CN+ paramagnetic or diamagnetic?
- CN+ is diamagnetic because all of its electrons are paired in the molecular orbitals.
- What is the role of the positive charge in CN+?
- The positive charge signifies the removal of an electron. In the case of CN+, removing an electron from the highest occupied molecular orbital (which would be π*2p if it were filled in neutral CN) leads to a more stable configuration with a higher bond order and no unpaired electrons, thus increasing stability.
- Does VSEPR theory apply to diatomic molecules?
- VSEPR theory is primarily used for polyatomic molecules to predict molecular geometry based on electron domain repulsion. For diatomic molecules, the geometry is inherently determined by the nature of the atomic orbital overlap and the resulting molecular orbitals, which in the case of CN+ leads to a linear structure due to the triple bond.
- What are the potential applications or significance of CN+?
- While perhaps not as commonly encountered as CN⁻, species like CN+ can be relevant in astrochemistry, mass spectrometry, and in understanding reactive intermediates in various chemical processes.
If you want to read more articles similar to Unraveling the CN+ Molecular Structure, you can visit the Automotive category.
