28/01/2018
Fossil fuels – the very bedrock of modern civilisation. From the moment we wake up and switch on a light, to the fuel that powers our commutes and the energy that drives our industries, these ancient deposits are inextricably linked to our daily lives. But have you ever stopped to consider where these ubiquitous energy sources actually come from? The term 'fossil fuel' itself hints at a deep history, conjured from the remains of life that existed millions of years ago. Yet, the reality of their formation is a complex tale woven from biology, geology, and immense spans of time.

The Ancient Origins: More Than Just Dinosaur Bones
The name 'fossil fuel' can be a little misleading. While we often associate fossils with dinosaur skeletons, the primary building blocks of the fuels we rely on are not, for the most part, derived from these magnificent prehistoric reptiles. As geologist Eric Roberts from the Colorado School of Mines explains, while a dinosaur could theoretically become a fuel, their sheer numbers simply weren't sufficient to create the vast reserves we possess today. Instead, the energy locked within fossil fuels originates from far more abundant life forms: microscopic algae and terrestrial plants.
These organisms, having captured the sun's energy through photosynthesis, stored a remarkable amount of power within their chemical bonds. This stored energy is what we tap into when we burn coal, oil, and natural gas. Organisms higher up the food chain, including herbivorous and carnivorous dinosaurs, store significantly less energy. This is because energy is lost at each trophic level as organisms expend energy to consume and digest their prey. Therefore, the foundational energy providers – the plants and algae – are the true ancestors of our fossil fuel supply.
The Genesis of Fossil Fuels: A Million-Year Process
The formation of fossil fuels is a remarkable geological process that unfolds over millions of years, requiring a very specific set of conditions. It begins when dead algae sink to the bottom of the ocean, and terrestrial plants fall into the stagnant waters of swamps. In these environments, oxygen is scarce. This lack of oxygen is crucial, as it prevents rapid decomposition by aerobic bacteria. Instead, the organic matter is quickly buried under layers of sediment.
Over eons, these sediments accumulate, subjected to immense pressure from the weight of overlying rock and increasing temperatures from the Earth's internal heat. This high-pressure, high-temperature, low-oxygen environment is what transforms the decaying organic matter. The absence of oxygen allows for anaerobic decomposition to occur, preserving the carbon bonds that store energy. If oxygen were present, the carbon would readily combine with it to form carbon dioxide, much like when we burn fossil fuels today. This preservation of carbon bonds is the key to creating these potent energy sources.
The initial product of this decomposition is an insoluble organic material called kerogen. The type of kerogen formed depends on the original organisms. Lipids, commonly found in algae and plankton, are more 'oil-prone' and form Type I kerogen, which eventually becomes crude oil. Lignans, found in terrestrial plants, are more 'gas-prone' and form Type III kerogen, leading to natural gas. A mixture of both lipids and lignans results in Type II kerogen, capable of producing both oil and gas.

As the kerogen is pushed deeper into the Earth's crust, subjected to even greater heat and pressure, it undergoes further chemical changes. Hydrocarbons – molecules composed of hydrogen and carbon atoms arranged into long chains – begin to form. These hydrocarbons are the fundamental components of fossil fuels.
The Trio of Fossil Fuels: Coal, Oil, and Natural Gas
While all fossil fuels are hydrocarbons derived from kerogen, they differ in their formation processes and composition, leading to distinct properties:
Coal: The Swampy Legacy
Coal's journey begins in vast, swampy regions teeming with vegetation. When plants died, their remains accumulated in these waterlogged, oxygen-poor environments, forming peat – a partially decayed organic material. As sedimentation occurred, the peat was buried, subjected to increasing pressure and heat over millions of years. This process transforms peat through several stages:
- Peat: The initial stage, rich in moisture and carbon.
- Lignite: Often called brown coal, it has more carbon than peat but retains high moisture content.
- Bituminous Coal: More compressed and drier, this is the most commonly used coal for power generation and industry.
- Anthracite: The hardest and highest-grade coal, boasting the greatest carbon content and energy output.
Coal is the most carbon-rich fossil fuel, meaning its combustion releases the largest amount of carbon dioxide into the atmosphere.
Oil (Petroleum): The Marine Microbe's Gift
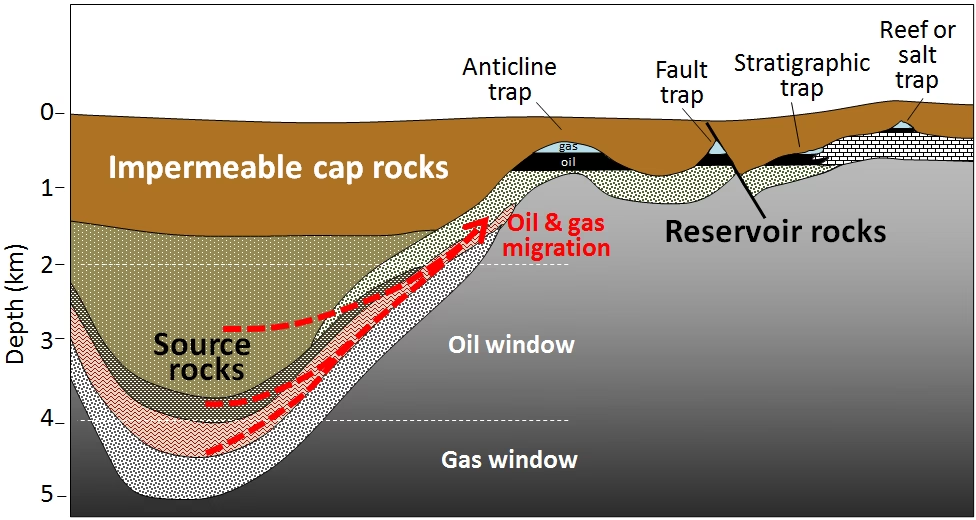
Oil originates from microscopic marine organisms like plankton. When these organisms died, their remains settled on the seabed, becoming mixed with sediment. Over millions of years, layers of silt and mud buried this organic material, creating the necessary pressure and heat. In a low-oxygen environment, this material transformed into kerogen. Further heat and pressure then converted the kerogen into liquid hydrocarbons – crude oil. This oil then migrated upwards through porous rock layers until it was trapped by impermeable rock, forming reservoirs that we can now tap through drilling.
Natural Gas: The Simpler Hydrocarbon
Natural gas, primarily composed of methane, often forms alongside oil. It can also be formed through the further decomposition of kerogen or crude oil under even higher temperatures. The heat breaks down larger hydrocarbon molecules into simpler ones. Methane is the least energy-rich fossil fuel. It is less dense than oil and rises more readily through rock formations. Natural gas can also be formed by microscopic organisms called methanogens in low-oxygen environments, creating biogenic methane, whereas gas formed from ancient plant matter is known as thermogenic methane.
The Energy Dilemma: Combustion and Consequences
Burning fossil fuels, a process known as combustion, releases the stored energy within their chemical bonds. When a fossil fuel is ignited, oxygen molecules replace the hydrogen atoms bonded to carbon. This rapid chemical reaction releases a significant amount of energy, producing heat, light, carbon dioxide, and water. However, this process is not without its drawbacks.

The combustion of fossil fuels releases not only energy but also harmful air pollutants, including carbon monoxide and sulfur dioxide. More significantly, it releases vast quantities of carbon dioxide (CO₂), a potent greenhouse gas that is a primary driver of global climate change. The continued reliance on fossil fuels for energy has profound implications for the planet's climate and ecosystems.
The Quest for Alternatives: A Post-Fossil Fuel Future
Recognising the environmental impact and finite nature of fossil fuels, scientists and engineers are actively pursuing solutions to reduce our dependence on them. This includes developing cleaner ways to burn existing fuels, such as using natural gas, which emits less CO₂ than coal, and exploring advanced technologies like carbon capture and sequestration. This process involves capturing CO₂ emissions from industrial sources and storing them underground to prevent them from entering the atmosphere.
Furthermore, research is underway to harness entirely new energy sources. Some scientists are even exploring innovative ways to create renewable materials from by-products of fossil fuel combustion, such as using carbon dioxide and sugar to produce renewable plastics. The challenge lies in transitioning to a sustainable energy future, one that can meet global energy demands without compromising the health of the planet.
Formation of Fossil Fuels: A Comparative Overview
| Fossil Fuel | Primary Organic Source | Dominant Process | Key Conditions |
|---|---|---|---|
| Coal | Terrestrial Plants | Accumulation and compression of plant debris in swamps | High pressure, moderate heat, low oxygen |
| Oil (Petroleum) | Marine Algae and Plankton | Burial and transformation of organic matter into kerogen, then hydrocarbons | High pressure, moderate to high heat, low oxygen |
| Natural Gas | Marine Organisms (plankton, bacteria) and some plant matter | Decomposition of organic matter, often at higher temperatures than oil formation | High pressure, high heat, low oxygen; can also be biogenic |
Frequently Asked Questions
Q1: Can fossil fuels be replaced?
Yes, the global effort is to replace fossil fuels with renewable energy sources such as solar, wind, hydro, and geothermal power. While fossil fuels are still essential in the immediate future, the long-term goal is a transition to a sustainable energy system.
Q2: Do fossil fuels really come from fossils like dinosaurs?
While the term 'fossil' is used, fossil fuels are primarily derived from the remains of ancient plants and microscopic organisms like algae and plankton, not typically from dinosaur bones. Dinosaur remains are too scarce to account for the vast reserves of fossil fuels.
Q3: How long does it take for fossil fuels to form?
The formation of fossil fuels is an incredibly slow process, taking millions of years. This geological timescale is why they are considered non-renewable resources; we consume them far faster than they can be naturally replenished.
Q4: What are the main environmental problems associated with fossil fuels?
The main environmental problems include the release of greenhouse gases (primarily carbon dioxide) during combustion, contributing to climate change, and the emission of air pollutants like sulfur dioxide and carbon monoxide, which can harm human health and the environment.
Q5: Is coal a fossil fuel?
Yes, coal is one of the primary types of fossil fuels, formed from the accumulated remains of ancient plants over millions of years under heat and pressure.
The journey from ancient organic matter to the fuels that power our modern world is a testament to the Earth's geological processes. Understanding this origin story is crucial as we navigate the challenges of energy consumption and strive for a more sustainable future. The better we understand these powerful, yet finite, resources, the more equipped we will be to transition to cleaner, more environmentally sound energy solutions.
If you want to read more articles similar to The Truth About Fossil Fuels, you can visit the Automotive category.
