17/03/2004
Plastics have become an indispensable part of modern life, serving as essential components in everything from packaging to electronics. From the sleek dashboard of your car to the robust casing of your smartphone, these versatile materials are ubiquitous. Yet, many are unaware of the intricate process that transforms crude oil, a fossil fuel extracted from the depths of the Earth, into the polymers that comprise plastic. This article delves into the step-by-step journey of oil to plastic, shedding light on the complex chemical processes involved in this remarkable transformation, and exploring the environmental considerations that come with it.
The Refining Process: Unlocking Crude Oil's Potential
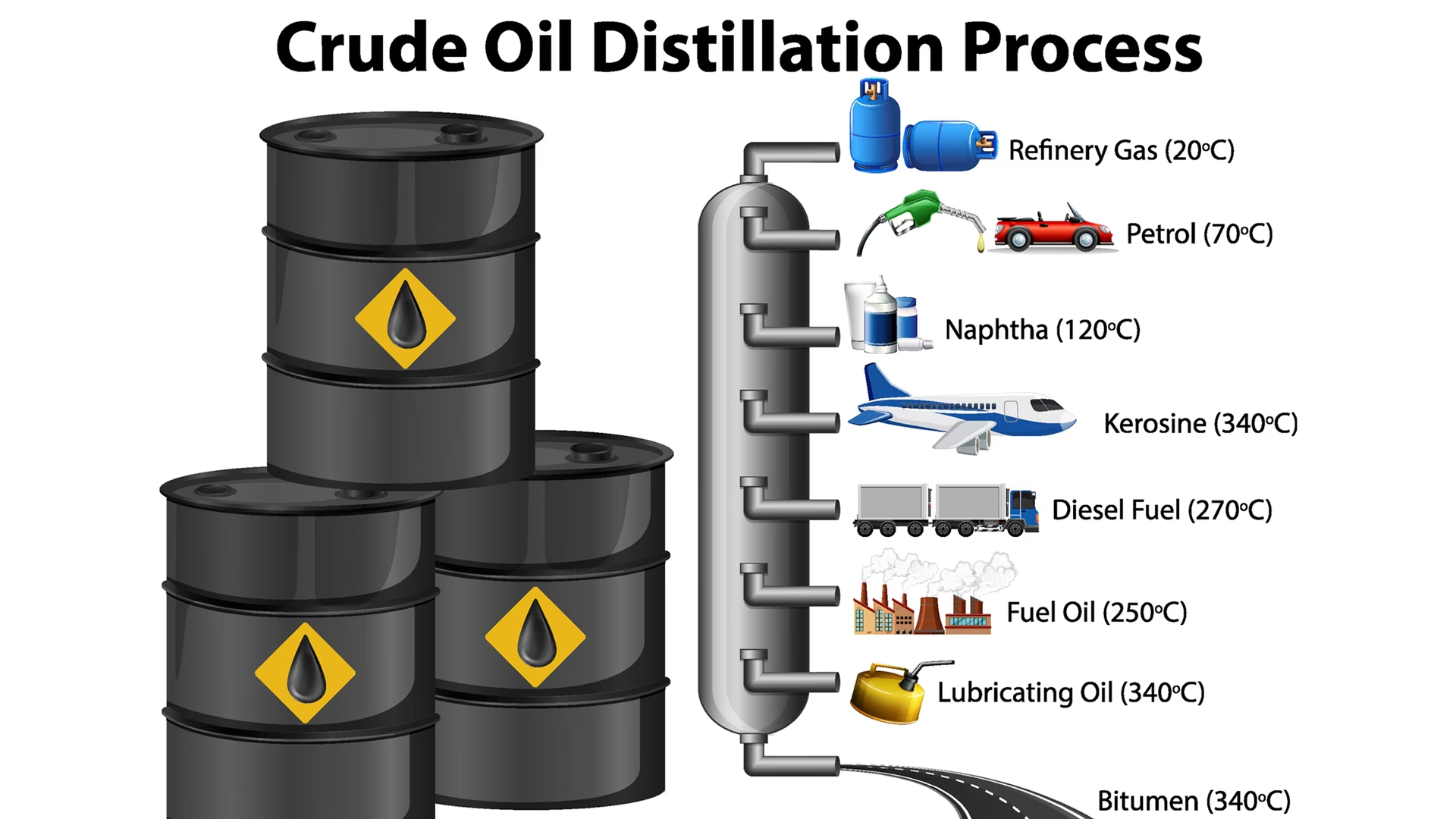
The journey of crude oil to plastic begins with its extraction from deep underground reservoirs. Once brought to the surface, this raw, dark, viscous liquid is transported to a refinery. Here, it undergoes a crucial initial transformation to separate its various components, preparing them for further processing. Think of crude oil as a complex cocktail of hydrocarbons, each with a different boiling point, and the refinery's job is to carefully distil these components apart.
Distillation: Separating the Blend
The primary method for separating crude oil is fractional distillation. Imagine a giant, towering pot where the crude oil is heated to extremely high temperatures, causing it to vaporise. This hot vapour then rises through a tall distillation column, gradually cooling as it ascends. As the vapours cool, different hydrocarbons condense back into liquid form at various levels, depending on their unique boiling points. Lighter fractions, such as gases and naphtha (a key precursor for plastics), rise higher before condensing, while heavier fractions like fuel oil and bitumen condense lower down. This careful separation is akin to sifting different-sized particles through a series of sieves, each layer collecting a specific component. The naphtha fraction is particularly vital for plastic production.
The Role of Catalysts: Speeding Up Reactions
During the refining process, and indeed throughout the entire transformation to plastic, catalysts play an absolutely critical role. These are substances that accelerate chemical reactions without being consumed in the process themselves. You can think of a catalyst as a skilled matchmaker, bringing reactive molecules together more efficiently and allowing reactions to occur at lower temperatures or pressures, saving immense amounts of energy. For instance, in crude oil refining, catalysts are used in processes like catalytic cracking to break down heavier, less useful hydrocarbons into lighter, more valuable ones, such as those needed for plastic monomers. Without catalysts, many of these transformations would be too slow or energy-intensive to be economically viable, making the entire process of plastic production far less efficient.
Monomer Production: Building Blocks of Plastic
Once the crude oil has been refined into its various fractions, the specific components destined for plastic production, primarily naphtha, undergo further processing to create the fundamental building blocks of plastic: monomers. Monomers are small molecules that can chemically bond together in long, repeating chains to form polymers, which are the essence of plastic.
Ethylene Formation: The Simplest Alkene
One of the most crucial monomers produced is ethylene, also known as ethene. This simple organic compound is a cornerstone of the plastics industry. Ethylene is typically produced from crude oil components through a process called steam cracking. Here, long hydrocarbon chains from the naphtha fraction are subjected to extremely high temperatures (around 800-900°C) in the presence of steam. This intense heat breaks the strong chemical bonds within the larger hydrocarbon molecules, shattering them into smaller, unsaturated molecules like ethylene. It’s a bit like taking a long, complex Lego structure and breaking it down into its simplest, most fundamental bricks. The steam helps to prevent undesirable side reactions and promotes the formation of these lighter olefins, ensuring a more efficient yield of the desired monomer.
Styrene Creation: Adding Versatility
Another important monomer derived from crude oil is styrene. While ethylene forms the backbone for many common plastics, styrene contributes its own unique properties, leading to materials like polystyrene. The production of styrene also involves cracking crude oil components, often ethylbenzene, but under different conditions than those used for ethylene. Typically, ethylbenzene is dehydrogenated (hydrogen atoms are removed) at high temperatures, often with the aid of specific catalysts, to form styrene. This process, while chemically distinct from steam cracking, similarly transforms a petroleum-derived compound into a versatile monomer. Together, ethylene and styrene, alongside other monomers like propylene, form the foundation of a vast array of plastic materials, essential components in our modern world.
Polymerization Reaction: Linking the Monomers
With the monomers successfully produced, the next critical stage is polymerization. This is the chemical process where individual monomer units are joined together to form long, repeating chains known as polymers. Imagine linking countless small paper clips together to form a long chain; each paper clip is a monomer, and the entire chain is a polymer. The method of polymerization significantly influences the final properties of the plastic, from its strength and flexibility to its transparency and heat resistance.
Suspension Polymerization: Tiny Plastic Beads
One common method is the suspension polymerization. In this technique, monomers are dispersed as tiny droplets in a liquid, usually water, with the help of a suspending agent to prevent them from clumping together. Think of it like creating a fine mist of monomer droplets in water. A small amount of a catalyst is then added to initiate the reaction within each individual droplet. As the polymerization proceeds, the monomers within each droplet link up, forming solid polymer particles. These particles remain suspended in the water, resembling tiny plastic beads or pearls. This method is particularly useful for producing polymers in a granular form, which are then easily separated from the water and can be processed further, for example, into PVC (polyvinyl chloride) or polystyrene.
Solution Polymerization: A Homogeneous Blend
In contrast to suspension polymerization, the solution polymerization process involves dissolving the monomers directly into a suitable solvent, creating a homogeneous mixture. Picture making a perfectly blended smoothie where all ingredients are uniformly mixed. A catalyst is then introduced to kick-start the polymerization. As the monomers react and form polymers, these new, larger polymer chains remain dissolved or dispersed within the solvent. This method often yields polymers with high molecular weights and uniform properties, making it ideal for producing certain types of plastics, such as poly(methyl methacrylate) (PMMA), used in acrylic glass. The choice between suspension, solution, and other polymerization methods depends on the specific plastic desired and its intended application.
| Polymerization Method | Monomer State | Solvent Used | Typical Products | Advantages |
|---|---|---|---|---|
| Suspension | Dispersed droplets in water | Water (as continuous phase) | PVC, Polystyrene (granular) | Easy heat removal, granular product, simple recovery |
| Solution | Dissolved in organic solvent | Organic solvent | PMMA, Polyethylene (some types) | Good temperature control, homogeneous product, high purity |
Plastic Types from Oil: A World of Versatility
The beauty of the oil-to-plastic transformation lies in its ability to create an incredible diversity of materials, each with unique properties tailored for specific applications. From the same crude oil, engineers can derive monomers that lead to vastly different plastics, shaping our world in countless ways.

Polyethylene (PE): The Ubiquitous Workhorse
Polyethylene, often abbreviated as PE, is perhaps the most widely used plastic globally. Its name, derived from "poly" (meaning many) and "ethylene" (its monomer), perfectly describes its structure: a long chain of interconnected ethylene molecules. This polymer's versatility is astounding; it can range from very flexible and transparent (low-density polyethylene, LDPE, used in plastic bags and films) to rigid and opaque (high-density polyethylene, HDPE, found in milk bottles, pipes, and car fuel tanks). The length and branching of the ethylene chains dictate its density and properties. Its chemical inertness, durability, and relatively low cost make it indispensable for packaging, construction, and consumer goods.
Polystyrene (PS): Lightweight and Insulating
Another common plastic derived from oil is polystyrene (PS). This polymer is formed from styrene monomers and is well-known for its lightweight, rigid, and insulating properties. You'll encounter polystyrene in foam cups, disposable food containers, CD cases, and insulation panels. When expanded, it forms expanded polystyrene (EPS), which is the material often used for protective packaging. Polystyrene can be clear and brittle, or it can be foamed to create a material that is excellent for thermal insulation. Its ease of moulding and relatively low cost make it a popular choice for a multitude of applications, though its brittleness can be a drawback in certain uses.
| Plastic Type | Monomer | Key Properties | Common Applications |
|---|---|---|---|
| Polyethylene (PE) | Ethylene | Flexible, durable, chemical resistant, variable density | Plastic bags, bottles, pipes, films, containers |
| Polystyrene (PS) | Styrene | Rigid, lightweight, good insulator, clear or opaque | Foam cups, food containers, CD cases, insulation |
| Polypropylene (PP) | Propylene | Tough, flexible, heat resistant, chemical resistant | Car parts, packaging, textiles, laboratory equipment |
| Polyvinyl Chloride (PVC) | Vinyl Chloride | Rigid or flexible, durable, weather resistant, flame retardant | Window frames, pipes, flooring, electrical cable insulation |
Environmental Impact: The Cost of Convenience
While the transformation of crude oil into plastic has revolutionised modern life, offering unparalleled convenience and innovation, it comes with significant environmental costs. Understanding these impacts is crucial for making informed choices and driving the push for more sustainable solutions.
Greenhouse Gas Emissions: Warming Our Planet
The entire lifecycle of plastic, from oil extraction to refining and manufacturing, is energy-intensive and releases substantial amounts of greenhouse gases (GHGs) into the atmosphere. The refining of crude oil alone is a major emitter, as it requires immense heat and pressure. Subsequent chemical processes, like steam cracking for ethylene production, consume vast amounts of energy, often derived from burning fossil fuels, which further contributes to GHG emissions. These gases, such as carbon dioxide and methane, act like an invisible blanket, trapping heat in our atmosphere and contributing to global warming and climate change. Methane, often released during oil and gas extraction, has a far greater warming potential than carbon dioxide over a shorter period, making its emissions particularly concerning.
Energy Consumption: A Resource-Intensive Process
Beyond direct GHG emissions, the sheer energy consumption required to transform crude oil into plastic is a major environmental concern. Every step, from drilling and transportation to refining and polymerization, demands significant amounts of power. Oil refineries are massive industrial complexes that run continuously, requiring a constant supply of electricity to operate their complex machinery, pumps, and heating systems. This energy typically comes from non-renewable sources, further depleting finite resources and exacerbating the environmental footprint. The energy embedded in plastic products is substantial, highlighting the importance of efficient production processes, recycling, and the development of alternative, less energy-intensive materials.

Frequently Asked Questions
What is the process of turning oil into plastic?
The transformation of crude oil into plastic is a multi-stage chemical engineering process. It begins with the extraction of crude oil, followed by its refining through fractional distillation to separate it into various components, including naphtha. This naphtha is then subjected to processes like steam cracking to produce smaller molecules called monomers, such as ethylene and propylene. Finally, these monomers undergo polymerization, where they chemically bond together to form long chains known as polymers, which are the fundamental substance of plastic. These polymers are then typically compounded with additives and processed into the final plastic products.
What is the journey of oil to plastic?
The journey from oil to plastic is intricate and involves several key stages. It starts with the extraction of crude oil from underground reservoirs. This crude oil is then transported to a refinery for refining via fractional distillation, separating it into different fractions like naphtha. The naphtha is then used in processes such as cracking and monomer production, where larger hydrocarbons are broken down into smaller, reactive monomer units (e.g., ethylene, styrene). These monomers then undergo polymerization, linking together to form long polymer chains. Subsequently, these polymers are blended with additives in a process called formulation and processing, before being shaped into final products through techniques like injection moulding or extrusion. Finally, these plastic products enter distribution and consumption, becoming part of our daily lives.
How do oil companies make plastics?
Oil companies play a foundational role in plastic production by primarily extracting and refining crude oil, which serves as the raw material. They don't typically produce the final plastic products themselves, but rather the essential precursors. After extracting crude oil, they refine it through distillation to obtain fractions like naphtha. This naphtha is then further processed, often through thermal cracking (like steam cracking), to produce individual monomer molecules such as ethylene, propylene, and butadiene. These monomers are the chemical building blocks. While some oil and gas companies might have petrochemical divisions that perform the subsequent polymerization steps to create raw plastic pellets (polymers), many sell these monomer feedstocks to dedicated chemical companies that specialise in polymerization and plastic manufacturing. Therefore, oil companies provide the fundamental chemical ingredients that make plastic production possible.
The journey from crude oil to plastic is a testament to human ingenuity and chemical engineering prowess. It transforms a raw, subterranean fossil fuel into an incredibly diverse range of materials that underpin much of our modern infrastructure and consumer goods. However, this convenience comes with a profound environmental footprint, from significant greenhouse gas emissions and energy consumption to the pervasive issue of plastic waste. As we continue to rely on plastics, understanding their origins and impacts becomes ever more critical, urging us towards greater innovation in sustainable practices, responsible consumption, and the development of alternative materials for a more environmentally conscious future.
If you want to read more articles similar to From Crude to Plastic: The Transformation Journey, you can visit the Automotive category.
