19/04/2006
In the vast and intricate landscape of biology, certain terms carry immense weight, hinting at complex phenomena and groundbreaking discoveries. One such term is 'phage'. While it might sound technical or even obscure to the uninitiated, 'phage' and its derivatives are at the heart of some of the most exciting advancements in medicine and biotechnology. From understanding the very essence of consumption to pioneering new treatments against formidable pathogens, the concept encapsulated by 'phage' is both ancient in its linguistic roots and utterly modern in its applications. This article delves deep into what 'phage' truly means, exploring its etymological journey, its role in sophisticated laboratory techniques, and its surprising resurgence as a powerful weapon in the fight against disease.
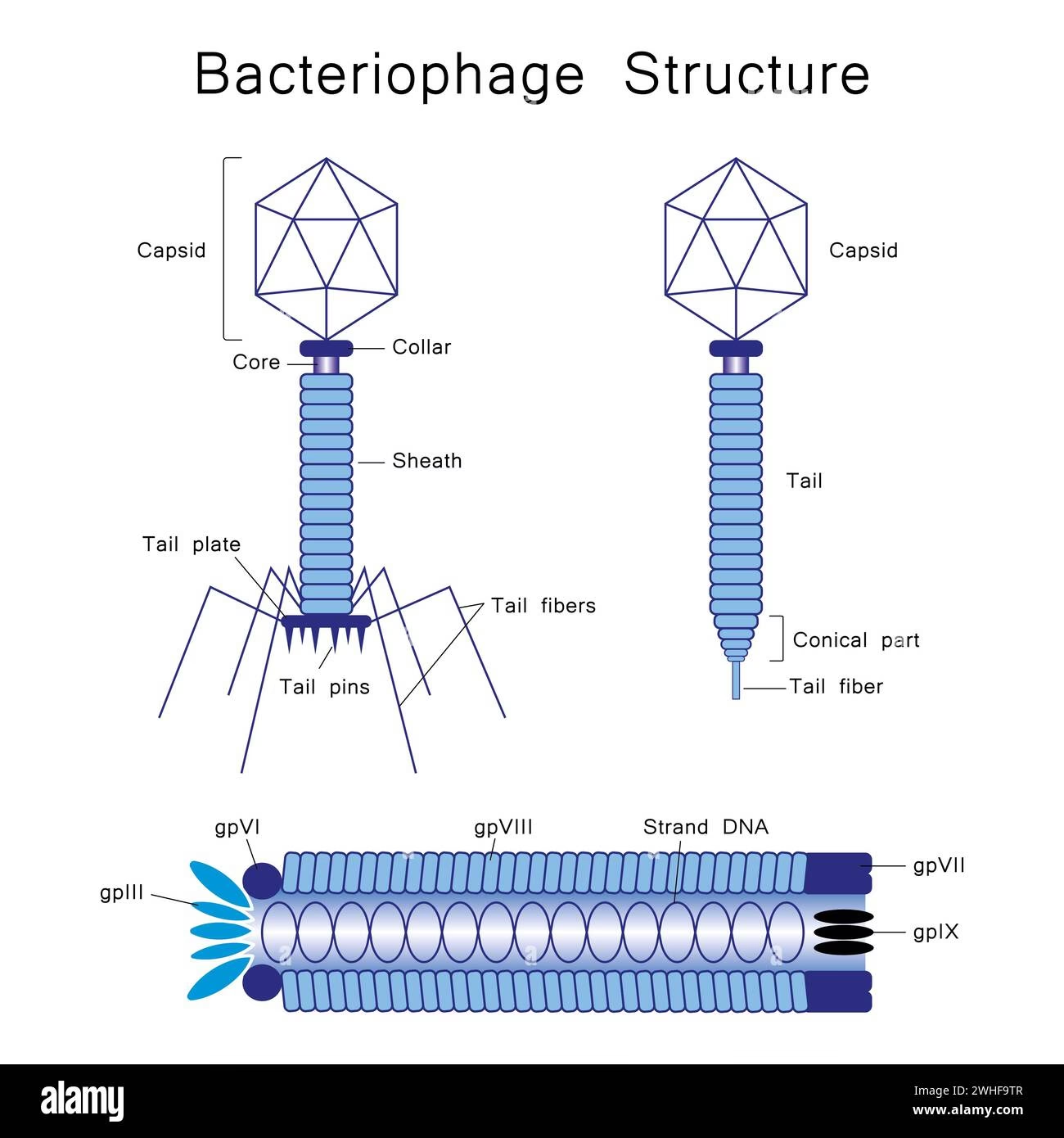
At its core, the word 'phage' carries a fundamental meaning that speaks to its biological functions. The suffix '-phage' originates from the ancient Greek noun phágos, which translates directly to 'eater' or 'devourer'. This simple yet profound root provides the key to understanding a multitude of scientific and everyday terms. In the majority of English words that incorporate this suffix, '-phage' signifies 'that which eats' or 'that which feeds on'. The specific object of this consumption is then indicated by the preceding part of the word, leading to a fascinating array of descriptive terms.
Consider, for instance, words like entomophage, which refers to an organism that feeds on insects. Similarly, anthropophage describes an entity that consumes human flesh, a term often associated with cannibalism. These examples vividly illustrate the direct meaning of 'eater'. However, the versatility of the suffix extends beyond mere consumption. It can also imply 'that which gnaws' or 'that which destroys'. This destructive aspect is evident in terms such as lithophage, which describes an organism that erodes or gnaws at stone, or xylophage, used for creatures that devour wood, a common concern for timber structures. Another interesting example is onychophage, a term less about external feeding and more about a self-destructive habit, specifically referring to someone who bites or gnaws their fingernails. Perhaps one of the most clinically relevant examples in this category is bacteriophage, which denotes a type of virus that infects and, crucially, destroys bacteria. This specific application of 'phage' will be explored in greater detail later, given its profound implications for human health.

Beyond the literal acts of eating or destroying, the suffix '-phage' can also take on a more figurative sense, indicating 'that which consumes' in a broader, often metaphorical, context. The most widely recognised example here is chronophage, a term used to describe something that consumes or takes up too much time. This concept of consumption extends playfully into words like bibliophage, which might describe an avid reader who 'devours' books, or cinéphage, referring to someone who consumes a great deal of cinema. Interestingly, even familiar anatomical terms like oesophagus (literally 'gullet-eater', referring to the tube through which food is 'eaten' or transported to the stomach) and the historical funerary container sarcophagus (literally 'flesh-eater', from the belief that certain types of stone would decompose the body within) are also formed using the '-phage' root, highlighting its deep integration into the lexicon.
Understanding Bacteriophages: Nature's Tiny Predators
While the suffix 'phage' is broadly applied, its most significant scientific application centres around bacteriophages, often simply called phages. These are viruses that specifically infect and replicate within bacteria and archaea. They are ubiquitous in every environment on Earth where bacteria exist, including soil, water, and the human gut. Bacteriophages play a crucial role in regulating bacterial populations and are among the most abundant biological entities on the planet. Their unique ability to target and destroy bacteria without harming human cells makes them subjects of intense scientific interest, particularly in an era grappling with antibiotic resistance.
Phage Display: A Powerful Biotechnological Tool
Beyond their natural role, bacteriophages have been ingeniously repurposed as powerful tools in biotechnology. One such groundbreaking technique is phage display. This method is primarily used to study the interactions between proteins displayed on the surface of a bacteriophage and other molecules, such as peptides, DNA, or other proteins. It has revolutionised the identification of high-affinity interactions, particularly between antibodies and antigens, which are absolutely critical in understanding viral pathogenesis, developing vaccines, and creating new therapeutic treatments.

The underlying principle of phage display involves genetically engineering bacteriophages to 'display' a particular protein or peptide on their outer coat. A library of phages, each displaying a different protein, can then be created. This massive diversity allows researchers to screen billions of potential binders against a target molecule of interest. The workflow for phage display typically involves several iterative steps, designed to enrich for phages that exhibit the strongest binding affinity:
The Phage Display Workflow
- Panning (Adherence to Plastic): This is the initial enrichment process. A large library of phages, potentially containing billions of different protein sequences, is exposed to a target antigen immobilised on a surface (often plastic). Phages that bind to the antigen are retained, while unbound phages are washed away. The bound phages are then 'eluted' (released) and amplified by infecting bacterial hosts, preparing them for the next round. This iterative process allows for the progressive enrichment of phages with higher binding affinities for the target.
- Colony Picking: After several rounds of panning, the selected bacteriophages, which now represent a population highly enriched for strong binders, are cloned. Individual bacterial colonies, each infected by a unique phage, are then picked and isolated. This step ensures the isolation of individual protein binders for further characterisation.
- Antigen-Antibody Interactions (Validation): While panning provides a qualitative selection for high-affinity binders, quantitative validation is essential. This step involves using more precise immunological assays to evaluate the specific antibody-antigen interactions. Common techniques include:
- ELISA (Enzyme-Linked Immunosorbent Assay): A plate-based assay designed to detect and quantify peptides, proteins, antibodies, and hormones.
- Immunofluorescence: A technique used to visualise the binding of antibodies to specific antigens within cells or tissues.
- HTRF (Homogeneous Time-Resolved Fluorescence): A robust technology for drug discovery and life science research, enabling high-throughput screening of molecular interactions.
- Other methods like complement fixation, agglutination, and precipitation may also be employed depending on the specific research question.
- Functional Screening: Following the detailed characterisation of antibody-antigen interactions, the most promising candidate molecules are put through functional screening. This often involves cell-based assays to determine their biological activity, such as their ability to neutralise a virus (e.g., viral neutralisation assays) or their efficacy as a vaccine component. This step confirms that the identified binders not only bind strongly but also perform the desired biological function.
The industrial applications of phage display, particularly in biotechnology and synthetic biology, are vast due to its high-throughput capabilities for protein interaction determination and protein engineering. Identifying high-affinity binders or ligands from massive phage libraries (ranging from 107 to 1012 potential candidates) requires efficient removal of non-binding phages and precise elution and harvesting of target-specific phages. This can be challenging with traditional manual methods, especially when screening multiple selections concurrently with different antigens. To accelerate phage display antibody screening, researchers are increasingly integrating automated solutions, such as the QPix™ system. Automated systems like QPix™ are ideal for high-speed plating and clone selection, capable of selecting up to 3,000 clones per hour with over 98% efficiency, allowing for the screening of up to 30,000 colonies per day with minimal human intervention. This significantly reduces manual effort and accelerates the discovery of promising target antibodies.
The Resurgence of Phage Therapy: A New Hope Against Superbugs
Perhaps the most compelling and historically rich application of bacteriophages is in phage therapy – the use of phages to treat bacterial infections. This concept is not new; it predates antibiotics and saw significant early success before largely falling out of favour in Western medicine.
A Brief History and Its Decline
The therapeutic potential of bacteriophages was first recognised by Félix D'Hérelle in the early 20th century. Alongside his Soviet counterparts, D'Hérelle pioneered phage therapy, with numerous reports of its efficacy in treating various bacterial infections, including cholera and dysentery. Major pharmaceutical companies in the US and Europe initially invested in producing phage cocktails. During World War II, both Allied and Axis forces, including Rommel's Afrika Korps and the Japanese, reportedly carried bacteriophage preparations for treating battlefield infections, and phages were successfully used to combat cholera outbreaks during the Battle of Stalingrad. However, despite these early successes, phage therapy gradually faded from prominence in the West after the 1940s. Several factors contributed to this decline.

Firstly, the advent of potent, broad-spectrum antibiotics, which were easier to standardise and administer, offered a seemingly superior alternative. Secondly, scientific controversies arose regarding the nature of phages (whether they were viruses or enzymes) and the lack of rigorous, placebo-controlled clinical trials by Western standards. Early phage preparations often suffered from poor quality control, containing inactive phages, bacterial debris, or harmful preservatives. Furthermore, a misunderstanding of what phages could treat led to their misapplication against viral diseases or non-infectious conditions. D'Hérelle's maverick personality and unconventional theories also generated friction within the scientific community. Finally, phage therapy became increasingly perceived as a 'Soviet medicine' during the Cold War, losing credibility in the West. While abandoned in Western countries, research and clinical application of phage therapy continued to flourish in parts of Eastern Europe, particularly in Georgia and Poland.
Challenges and a Renewed Interest
Today, with the escalating crisis of antibiotic resistance, there is a significant global resurgence of interest in phage therapy. The unique mechanisms of action of phages, their ability to evolve with bacteria, and their specificity make them a highly attractive alternative or adjunct to conventional antibiotics. However, significant challenges remain on the path to widespread adoption:
- Scientific Hurdles: There's a need for more robust, Western-standard clinical trials to demonstrate efficacy and safety. Understanding phage-host interactions and potential resistance mechanisms is also crucial.
- Regulatory Frameworks: Phages are living, replicating entities, which makes them inherently difficult to regulate under existing pharmaceutical frameworks designed for static chemical compounds. Their adaptability and multiplicity clash with regulations demanding stable, unique products.
- Financial Viability: Patenting living organisms, especially those naturally occurring, is complex, which discourages significant private investment from pharmaceutical companies. The development of 'phage cocktails' tailored to individual patients further complicates mass production and commercialisation.
- Educational and Public Perception: A lack of awareness among medical professionals and public apprehension towards using viruses for treatment are ongoing barriers.
Despite these challenges, innovative approaches are emerging. The concept of creating 'polyphagic cocktails' – formulations containing multiple phage types to ensure broad-spectrum activity against diverse bacterial strains – is gaining traction. Furthermore, advanced genetic engineering techniques, such as those derived from CRISPR technology, are being explored to modify phages for enhanced specificity or efficacy, particularly against antibiotic-resistant biofilms. In many Western countries, phage therapy is currently available primarily through 'compassionate use' or 'named patient' frameworks, allowing its application in severe, otherwise untreatable cases within a hospital setting, under strict medical supervision.
Effectiveness and Modern Evidence
While the historical evidence for phage therapy's effectiveness was often anecdotal or not up to modern clinical trial standards, contemporary practitioners and studies, particularly from countries where it was never abandoned, continue to report positive outcomes. For instance, in cases where patients face amputation or death due to untreatable infections, phage therapy has shown remarkable success. Recent scientific literature, including systematic reviews, generally supports the efficacy of phage therapy, particularly in localised infections. Studies also indicate that phages can be more effective against bacteria when they are colonising human cells compared to simple in vitro cultures, highlighting the importance of studying their effectiveness in relevant biological contexts.

Intriguingly, the concept of phages is now being explored in a novel way to combat not just bacteria, but also viruses like influenza and SARS-CoV-2. This innovative approach involves engineering bacteriophages to target viral components. For example, researchers have developed chemically modified phages that bind specifically to the haemagglutinins – the viral spikes that influenza uses to attach to and infect human cells. By essentially 'enveloping' the virus, these phages prevent it from binding to host cells, thereby neutralising its infectivity. This has been demonstrated in preclinical studies on human lung tissue, showing that phage-treated infected tissue could hardly infect new cells. This groundbreaking research, initially focused on influenza and avian flu, is now being applied to SARS-CoV-2, opening up a completely new avenue for phage-based antiviral therapies.
Other Applications of Phage Technology
The utility of phages extends beyond human medicine:
- Veterinary Medicine: Phage therapy is also being explored and used in veterinary medicine to treat bacterial infections in livestock, poultry, and aquaculture, offering a potential alternative to antibiotics in animal agriculture.
- Food Industry: Bacteriophages are used to destroy specific bacteria that can contaminate fresh food products. For instance, in 2006, the US Food and Drug Administration (FDA) approved the spraying of a cocktail of six non-genetically modified anti-Listeria phages on meat and poultry products. This application helps enhance food safety by reducing the risk of bacterial contamination, and current research suggests that oral exposure to active phages is generally considered safe for consumers.
Frequently Asked Questions About Phage
- What exactly is a 'phage'?
- The term 'phage' broadly refers to something that 'eats' or 'devours', derived from ancient Greek. In biology, it most commonly refers to bacteriophages, which are viruses that specifically infect and destroy bacteria.
- How do bacteriophages work?
- Bacteriophages operate by attaching to specific receptors on the surface of a bacterium, injecting their genetic material (DNA or RNA) into the host cell. They then hijack the bacterial machinery to replicate themselves, ultimately causing the bacterial cell to lyse or burst, releasing new phage particles.
- Is phage therapy safe for humans?
- Yes, bacteriophages are generally considered safe for humans because they are highly specific to bacterial cells and do not infect human cells. Any potential side effects are typically mild and related to the bacterial lysis itself rather than the phage. However, more robust clinical trials are needed to fully establish their safety and efficacy under various conditions.
- Why was phage therapy largely abandoned in Western medicine?
- Phage therapy was overshadowed by the discovery and mass production of antibiotics, which offered a more convenient and standardised treatment. Other reasons included a lack of rigorous clinical trials by modern Western standards, inconsistent quality of early phage preparations, and regulatory challenges for living biological agents.
- What is phage display used for?
- Phage display is a powerful laboratory technique that uses bacteriophages to display proteins or peptides on their surface. It's primarily used to identify and engineer high-affinity binders, such as antibodies, against specific targets. This is crucial for drug discovery, vaccine development, and understanding protein interactions.
In conclusion, the journey from the ancient Greek root 'phágos' to the cutting-edge applications of phage technology is a testament to the enduring power of fundamental biological principles. From the precise targeting of bacteria by bacteriophages to the sophisticated engineering of proteins via phage display, and the promising re-emergence of phage therapy, these microscopic entities are set to play an increasingly vital role in addressing some of humanity's most pressing health challenges, particularly the looming threat of antibiotic resistance. The versatility and adaptability of 'phage' concepts promise a future where innovative biological solutions continue to reshape our understanding and treatment of disease.
If you want to read more articles similar to Unravelling 'Phage': From Roots to Revolution, you can visit the Automotive category.

