27/11/2005
In the world of culinary arts, particularly within commercial kitchens and even at home, frying oil is an indispensable component. It’s the medium that transforms raw ingredients into crispy, golden delights. However, this seemingly simple liquid undergoes complex transformations during its use, fundamentally altering its properties and the quality of the food it cooks. These changes aren't merely cosmetic; they are the result of intricate chemical reactions occurring beneath the surface. Understanding these chemical processes is paramount for anyone involved in food preparation, not just for maintaining food quality and taste, but also for operational efficiency and safety. When frying oil is subjected to the high temperatures and continuous use typical of deep-frying, it inevitably degrades. This degradation isn't a single process but a confluence of three primary chemical reactions: oxidation, polymerisation, and hydrolysis. Each plays a distinct role in the oil's breakdown, contributing to undesirable flavours, textures, and even potentially harmful compounds. Let's delve into the science behind these transformations.
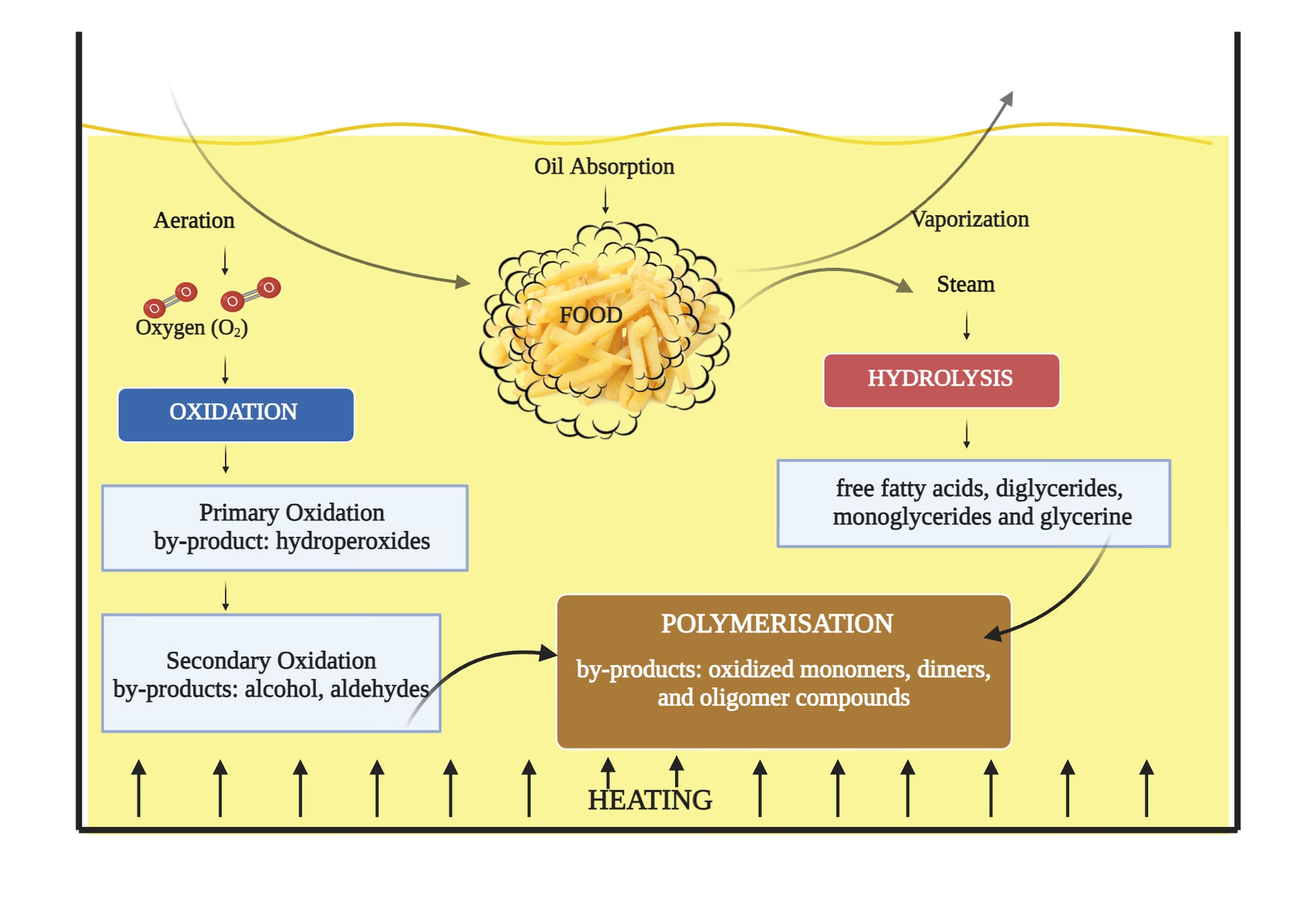
Oxidation: The Reaction with Air
Oxidation is arguably the most significant and often the initial chemical change that occurs in frying oil, particularly when exposed to heat and air. This reaction involves the oil's unsaturated fatty acids reacting with oxygen molecules present in the atmosphere. The process is accelerated significantly by high temperatures, light, and the presence of certain metal ions (like copper and iron), which act as catalysts. Think of it like rust forming on metal, but for your oil.
How Oxidation Manifests:
- Rancidity: The most noticeable effect of oxidation is the development of off-flavours and odours, often described as 'rancid' or 'stale'. This is due to the formation of volatile compounds like aldehydes and ketones.
- Colour Changes: Oxidised oil typically darkens in colour, often turning a brownish hue.
- Increased Viscosity: The oil may become thicker or more viscous.
- Foaming: As oxidation progresses, the oil's surface tension changes, leading to excessive foaming during frying.
- Reduced Smoke Point: The smoke point of the oil, which is the temperature at which it begins to produce a continuous bluish smoke, decreases significantly. This means the oil will smoke at lower temperatures, indicating severe degradation.
Mitigating Oxidation:
Minimising oxygen exposure is key. This includes keeping oil covered when not in use, ensuring proper filtration to remove food particles (which can also catalyse oxidation), and operating at optimal frying temperatures. The use of antioxidants in some commercial oils can also help delay this process.
Polymerisation: The Formation of Larger Molecules
Polymerisation is a chemical reaction where smaller molecules (monomers) combine to form larger, more complex molecules (polymers). In the context of frying oil, this occurs primarily at high temperatures, especially when the oil has already undergone some oxidation. The free radicals generated during oxidation can initiate the polymerisation process, causing fatty acid molecules to link together. These new, larger molecules are non-volatile and remain in the oil, accumulating over time.
How Polymerisation Manifests:
- Increased Viscosity: This is a hallmark of polymerisation. The oil becomes noticeably thicker and stickier, almost syrupy.
- Darkening: Similar to oxidation, polymerisation contributes to the oil turning much darker, often a deep brown or black.
- Gum and Varnish Formation: Polymers can deposit as sticky, gummy residues on frying equipment, heating elements, and even on the food itself. This 'varnish' can be difficult to remove and reduces the efficiency of equipment.
- Reduced Heat Transfer: The thickened oil and deposits on heating elements impede efficient heat transfer, leading to longer cooking times and unevenly cooked food.
- Foaming: Polymerised compounds can also contribute to excessive foaming.
Mitigating Polymerisation:
Controlling frying temperature is crucial, as is avoiding prolonged heating of the oil. Regular filtering helps remove some of the smaller polymerised products and food particles that can accelerate the process. Ultimately, regular oil replacement is the most effective way to combat severe polymerisation.
Hydrolysis: The Reaction with Water
Hydrolysis is a chemical reaction involving the breakdown of compounds through a reaction with water. In frying oil, triglycerides (the main components of oil) react with water molecules, leading to the release of free fatty acids (FFAs) and glycerol. This reaction is particularly prevalent when frying high-moisture foods, as the water from the food interacts directly with the hot oil. High temperatures accelerate this process considerably.
How Hydrolysis Manifests:
- Increased Free Fatty Acids (FFAs): This is the direct result of hydrolysis. A high FFA content indicates significant oil degradation.
- Reduced Smoke Point: Free fatty acids have a much lower smoke point than triglycerides. As their concentration increases, the oil's overall smoke point drops dramatically, leading to more smoke during frying.
- Foaming: FFAs can act as surfactants, reducing the surface tension of the oil and leading to excessive foaming.
- Off-Flavours: While not as pronounced as oxidative rancidity, the presence of high levels of FFAs can impart slightly sour or soapy off-flavours to food.
- Accelerated Degradation: FFAs can act as catalysts for further hydrolysis and oxidation, creating a vicious cycle of accelerated degradation.
Mitigating Hydrolysis:
The primary method to reduce hydrolysis is to minimise the introduction of water into the frying oil. This means ensuring food is as dry as possible before frying (e.g., patting chips dry before adding them to the fryer) and avoiding adding ice or frozen items directly into hot oil. Proper oil storage also prevents condensation from forming and dripping into the oil.
The Interplay of Degradation
It's important to understand that these three chemical changes rarely occur in isolation. They are interconnected and often accelerate one another. For instance, oxidation produces free radicals and breakdown products that can initiate polymerisation. Hydrolysis increases FFA levels, which can lower the smoke point and make the oil more susceptible to oxidation. This synergistic degradation means that once one process begins, it often creates conditions favourable for the others, leading to a rapid decline in oil quality.
Detecting Heat Abuse and Oil Degradation
Beyond sensory observation (smell, colour, viscosity, foaming), several methods are used to determine the extent of heat abuse and the formation of specific products in frying oils. In professional settings, these methods provide a more objective measure of oil quality:
- Total Polar Compounds (TPC): This is perhaps the most widely accepted and reliable method for assessing overall oil degradation. TPC measures the total amount of breakdown products formed from oxidation, polymerisation, and hydrolysis. Regulatory limits for TPC exist in many countries (e.g., 24-27% in Europe).
- Free Fatty Acid (FFA) Content: As discussed, FFAs are a direct indicator of hydrolysis. Higher FFA levels mean more water-induced breakdown.
- Peroxide Value (PV): This measures the amount of peroxides and hydroperoxides, which are primary products of oxidation. While useful, PV can decrease as oxidation progresses to secondary products, so it's best used in conjunction with other tests.
- Anisidine Value (AnV): This measures secondary oxidation products, such as aldehydes. Together, PV and AnV can give a more complete picture of oxidative degradation.
- Colour Comparators and Test Strips: Simpler, quicker methods for a visual or chemical estimation of oil quality, often used for daily checks.
Best Practices for Extending Frying Oil Life
Understanding the chemical changes allows for informed strategies to prolong oil life and ensure consistent food quality:
- Temperature Control: Never overheat oil. Maintain optimal frying temperatures (typically 170-180°C) and turn off fryers during idle periods.
- Filtration: Regularly filter oil to remove food particles, crumbs, and sediment. These act as catalysts for degradation and can absorb moisture.
- Proper Storage: Store fresh and used oil in a cool, dark place, in tightly sealed containers to minimise exposure to light, air, and moisture.
- Keep it Clean: Regularly clean the fryer, ensuring no residue builds up that could contaminate fresh oil.
- Dry Food Thoroughly: Remove as much surface moisture from food as possible before frying to reduce hydrolysis.
- Avoid Overloading: Overloading the fryer can cause a significant temperature drop, leading to longer cooking times and increased oil absorption by food.
- Top-Up vs. Replace: While topping up with fresh oil can dilute some degradation products, it's not a substitute for complete oil replacement when necessary.
Comparative Table of Frying Oil Degradation
| Chemical Change | Primary Cause | Key Effects on Oil & Food | Mitigation Strategies |
|---|---|---|---|
| Oxidation | Oxygen + Heat/Light/Metals | Rancid flavours, dark colour, increased viscosity, foaming, reduced smoke point. | Proper storage (sealed, dark, cool), filtration, optimal temperature, antioxidants. |
| Polymerisation | High Heat + Prolonged Heating + Free Radicals | Increased viscosity (syrupy), very dark colour, gum/varnish deposits, poor heat transfer. | Temperature control, regular oil replacement, good filtration. |
| Hydrolysis | Water + Heat | Increased Free Fatty Acids (FFAs), reduced smoke point, foaming, off-flavours (soapy/sour). | Drying food thoroughly, avoiding moisture introduction, proper oil storage. |
Frequently Asked Questions (FAQs)
How often should I change my frying oil?
There's no single answer, as it depends on the type of oil, the food being fried, the volume of frying, and the frying temperature. However, a good rule of thumb for commercial operations is to monitor oil daily using test strips or sensory evaluation. When the oil shows significant signs of degradation (dark colour, excessive foaming, low smoke point, off-flavours), or when TPC levels exceed acceptable limits, it's time for a change. For home use, if the oil smells stale, looks very dark, or smokes excessively at normal frying temperatures, it's time to replace it.
Can I mix new oil with old oil?
While topping up can extend the life of your oil, simply mixing old and new oil isn't ideal for long-term quality. The degradation products in the old oil will quickly accelerate the breakdown of the fresh oil. It's always best practice to filter old oil before topping up, and to ensure a complete oil change when the quality significantly deteriorates, rather than continuously adding new oil to a degraded batch.
What is 'smoke point' and why is it important?
The smoke point is the temperature at which cooking oil or fat begins to produce a continuous bluish smoke. This smoke indicates that the oil is breaking down, releasing volatile compounds. A lower smoke point means the oil is degrading faster and is less suitable for high-temperature cooking. It also affects the flavour of the food and can produce undesirable compounds. Maintaining a healthy smoke point is crucial for both food quality and safety.
Why does my frying oil foam excessively?
Excessive foaming is a common sign of oil degradation. It can be caused by an accumulation of free fatty acids (from hydrolysis), polar compounds (from oxidation and polymerisation), and tiny food particles. These substances reduce the oil's surface tension, allowing bubbles to persist and accumulate. It indicates the oil is nearing the end of its useful life.
Are degraded frying oils harmful?
While the occasional consumption of food fried in slightly degraded oil is unlikely to cause immediate harm, prolonged consumption of food fried in severely degraded oil is a concern. The breakdown products, such as aldehydes and polymers, can be associated with adverse health effects. Additionally, the nutritional quality of the food can be compromised. Therefore, maintaining good oil quality is important for both taste and health.
Conclusion
The degradation of frying oil is a complex chemical ballet, choreographed by oxidation, polymerisation, and hydrolysis. Each reaction contributes uniquely to the oil's decline, impacting its performance, the quality of fried foods, and ultimately, the profitability and reputation of any food establishment. By understanding these fundamental chemical changes, and implementing best practices for oil management, one can significantly extend the life of frying oil, ensure consistent product quality, and operate a more efficient and safer kitchen. It's not just about changing the oil when it looks bad; it's about understanding the science that dictates when and why it's truly time for a fresh batch.
If you want to read more articles similar to Frying Oil Degradation: The Chemical Trio, you can visit the Automotive category.
